
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Canada
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
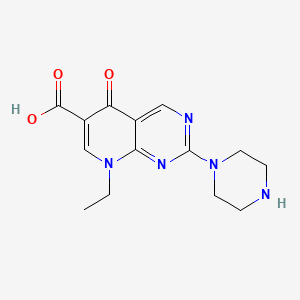

1. Acid, Pipemidic
2. Acid, Piperamic
3. Deblaston
4. Galusan
5. Nuril
6. Palin
7. Piperamic Acid
8. Pipram
9. Urisan
10. Uropipemid
1. 51940-44-4
2. Pipedac
3. Deblaston
4. Pipemid
5. Acido Pipemidico
6. Dolcol
7. Acide Pipemidique
8. Acidum Pipemidicum
9. Pipeacid
10. Pipram
11. Uropimid
12. Nuril
13. Pipemidate
14. Filtrax
15. Pipurin
16. Tractur
17. Urosten
18. Uroval
19. Pi-coli
20. 8-ethyl-5,8-dihydro-5-oxo-2-(1-piperazinyl)pyrido(2,3-d)pyrimidine-6-carboxylic Acid
21. Memento 400
22. Pipemidic Acid Anhydrous
23. Pipemidic Acid Hydrate
24. Nsc-758160
25. Lt12j5hvr8
26. 5,8-dihydro-8-ethyl-5-oxo-2-(1-piperazinyl)pyrido(2,3-d)pyrimidine-6-carboxylic Acid
27. Mls000028608
28. Pyrido(2,3-d)pyrimidine-6-carboxylic Acid, 8-ethyl-5,8-dihydro-5-oxo-2-(1-piperazinyl)-
29. Chebi:75250
30. Palin
31. 8-ethyl-5-oxo-2-(piperazin-1-yl)-5,8-dihydropyrido[2,3-d]pyrimidine-6-carboxylic Acid
32. Ncgc00016866-01
33. Pipemidic Acid (inn)
34. Smr000058711
35. Uromidin
36. Cas-51940-44-4
37. Dsstox_cid_3479
38. 8-ethyl-5-oxo-2-piperazin-1-yl-pyrido[2,3-d]pyrimidine-6-carboxylic Acid
39. Dsstox_rid_77045
40. Dsstox_gsid_23479
41. Pipemidic Acid [inn]
42. Pipemidicacid
43. 8-ethyl-5-oxo-2-piperazin-1-ylpyrido[2,3-d]pyrimidine-6-carboxylic Acid
44. Acido Pipemidico [spanish]
45. 1489 Rb
46. Rb 1489
47. Acide Pipemidique [inn-french]
48. Acido Pipemidico [inn-spanish]
49. Acidum Pipemidicum [inn-latin]
50. Sr-01000000245
51. Einecs 257-530-2
52. Unii-lt12j5hvr8
53. Brn 0626575
54. Pipemidic
55. Pipemidic-acid
56. Ccris 8517
57. Pipemidic Acid [inn:ban:dcf]
58. Nuril (tn)
59. Pipemidic Acid,(s)
60. Spectrum_001387
61. 8-ethyl-5,8-dihydro-5-oxo-2-(1-piperazinyl)pyrido[2,3-d]pyrimidine-6-carboxylicacidtrihydrate
62. Opera_id_1142
63. Prestwick0_000897
64. Prestwick1_000897
65. Prestwick2_000897
66. Prestwick3_000897
67. Spectrum2_001185
68. Spectrum3_001487
69. Spectrum4_000155
70. Spectrum5_001214
71. Acide Ethyl-8 Oxo-5 Piperazinyl-2 Dihydro-5,8 Pyrido(2,3-d)pyrimidine-6 Carboxylique [french]
72. Pipemidic Acid [mi]
73. Bspbio_000714
74. Bspbio_003073
75. Kbiogr_000630
76. Kbioss_001867
77. Pipemidic Acid [jan]
78. Bidd:gt0126
79. Chembl30116
80. Divk1c_000651
81. Schembl135557
82. Spectrum1502024
83. Spbio_001229
84. Spbio_002923
85. Bpbio1_000786
86. Pipemidic Acid [mart.]
87. Dtxsid3023479
88. Pipemidic Acid [who-dd]
89. Hms502a13
90. Kbio1_000651
91. Kbio2_001867
92. Kbio2_004435
93. Kbio2_007003
94. Kbio3_002573
95. Zinc57466
96. Ninds_000651
97. Hms1570d16
98. Hms1921f04
99. Hms2097d16
100. Hms3714d16
101. Pharmakon1600-01502024
102. Bcp13633
103. Hy-b1210
104. Tox21_110655
105. Nsc758160
106. S5051
107. Stk686262
108. Akos005599752
109. Tox21_110655_1
110. Ccg-220897
111. Cs-4838
112. Db13823
113. Nsc 758160
114. 8-ethyl-5-oxo-2-(piperazin-1-yl)-5h,8h-pyrido[2,3-d]pyrimidine-6-carboxylic Acid
115. 8-ethyl-5-oxo-2-piperazinyl-8-hydropyridino[2,3-d]pyrimidine-6-carboxylic Acid
116. Acide Ethyl-8 Oxo-5 Piperazinyl-2 Dihydro-5,8 Pyrido(2,3-d)pyrimidine-6 Carboxylique
117. Idi1_000651
118. Pyrido(2,3-d)pyrimidine-6-carboxylic Acid, 5,8-dihydro-8-ethyl-5-oxo-2-(1-piperazinyl)-
119. Pyrido[2,3-d]pyrimidine-6-carboxylicacid, 8-ethyl-5,8-dihydro-5-oxo-2-(1-piperazinyl)-
120. Smp1_000242
121. Ncgc00016866-02
122. Ncgc00016866-03
123. Ncgc00016866-04
124. Ncgc00016866-05
125. Ncgc00016866-06
126. Ncgc00016866-07
127. Ncgc00016866-08
128. Ncgc00016866-11
129. Ncgc00023999-03
130. Ncgc00023999-04
131. Ncgc00178314-01
132. Ncgc00178314-02
133. As-47690
134. Pipemidic Acid 100 Microg/ml In Methanol
135. Sbi-0051703.p002
136. Ab00490004
137. D08379
138. F15437
139. P-6835
140. Ab00052258_14
141. A828860
142. Q754986
143. Sr-01000000245-2
144. Sr-01000000245-3
145. Sr-01000000245-4
146. Brd-k08999871-001-05-5
147. Brd-k08999871-001-13-9
148. Pipemidic Acid, Antibiotic For Culture Media Use Only
149. Deblaston; Pipram; Uromidin; Rb 1489; Rb-1489; Rb1489
150. 2-(1-piperazinyl)-5,8-dihydro-8-ethyl-5-oxopyrido[2,3-d]pyrimidine-6-carboxylic Acid
151. 2-(1-piperazinyl)-8-ethyl-5,8-dihydro-5-oxopyrido[2,3-d]pyrimidine-6-carboxylic Acid
152. 5,8-dihydro-8-ethyl-2-(1-piperazinyl)-5-oxopyrido-[2,3-d]pyrimidine-6-carboxylic Acid
153. 5,8-dihydro-8-ethyl-2-(1-piperazinyl)-5-oxopyrido[2,3-d]pyrimidine-6-carboxylic Acid
154. 7-(1-piperazinyl)-1-ethyl-4-oxo-1,4-dihydro-6,8-diazaquinoline-3-carboxylic Acid
155. 8-ethyl-5-oxo-2-piperazin-1-yl-5,8-dihydropyrido[2,3-d]pyrimidine-6-carboxylic Acid
| Molecular Weight | 303.32 g/mol |
|---|---|
| Molecular Formula | C14H17N5O3 |
| XLogP3 | -2.1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 3 |
| Exact Mass | 303.13313942 g/mol |
| Monoisotopic Mass | 303.13313942 g/mol |
| Topological Polar Surface Area | 98.7 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 489 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
Anti-Infective Agents, Urinary
Substances capable of killing agents causing urinary tract infections or of preventing them from spreading. (See all compounds classified as Anti-Infective Agents, Urinary.)
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01M - Quinolone antibacterials
J01MB - Other quinolones
J01MB04 - Pipemidic acid
Global Sales Information

ABOUT THIS PAGE
60
PharmaCompass offers a list of Pipemidic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Pipemidic Acid manufacturer or Pipemidic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Pipemidic Acid manufacturer or Pipemidic Acid supplier.
A DSSTox_CID_3479 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of DSSTox_CID_3479, including repackagers and relabelers. The FDA regulates DSSTox_CID_3479 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. DSSTox_CID_3479 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of DSSTox_CID_3479 manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A DSSTox_CID_3479 supplier is an individual or a company that provides DSSTox_CID_3479 active pharmaceutical ingredient (API) or DSSTox_CID_3479 finished formulations upon request. The DSSTox_CID_3479 suppliers may include DSSTox_CID_3479 API manufacturers, exporters, distributors and traders.
click here to find a list of DSSTox_CID_3479 suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
DSSTox_CID_3479 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of DSSTox_CID_3479 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right DSSTox_CID_3479 GMP manufacturer or DSSTox_CID_3479 GMP API supplier for your needs.
A DSSTox_CID_3479 CoA (Certificate of Analysis) is a formal document that attests to DSSTox_CID_3479's compliance with DSSTox_CID_3479 specifications and serves as a tool for batch-level quality control.
DSSTox_CID_3479 CoA mostly includes findings from lab analyses of a specific batch. For each DSSTox_CID_3479 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
DSSTox_CID_3479 may be tested according to a variety of international standards, such as European Pharmacopoeia (DSSTox_CID_3479 EP), DSSTox_CID_3479 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (DSSTox_CID_3479 USP).

