
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
Canada
0
Australia
0
South Africa
Annual Reports
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Calcort
2. Dezacor
3. Emflaza
4. Zamene
1. 14484-47-0
2. Azacort
3. Calcort
4. Oxazacort
5. Flantadin
6. Emflaza
7. Cortax
8. Deflan
9. Mdl 458
10. Mdl-458
11. L-5458
12. Kr5yz6ae4b
13. Mdl458
14. Dezacor
15. Lantadin
16. Dsstox_cid_378
17. Dsstox_rid_75552
18. Dsstox_gsid_20378
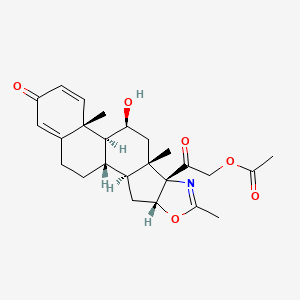
19. 2-((6ar,6bs,7s,8as,8bs,11ar,12as,12bs)-7-hydroxy-6a,8a,10-trimethyl-4-oxo-1,2,4,6a,6b,7,8,8a,11a,12,12a,12b-dodecahydro-8bh-naphtho[2',1':4,5]indeno[1,2-d]oxazol-8b-yl)-2-oxoethyl Acetate
20. Deflazacortum
21. Decortil
22. Deflanil
23. Enzocort
24. Deflazacort (calcort)
25. 2-[(4ar,4bs,5s,6as,6bs,9ar,10as,10bs)-5-hydroxy-4a,6a,8-trimethyl-2-oxo-2,4a,4b,5,6,6a,9a,10,10a,10b,11,12-dodecahydro-6bh-naphtho[2',1':4,5]indeno[1,2-d][1,3]oxazol-6b-yl]-2-oxoethyl Acetate
26. Cas-14484-47-0
27. Dl-458-it
28. Unii-kr5yz6ae4b
29. Deflazacortum [inn-latin]
30. Deflazacort [usan:inn:ban]
31. Einecs 238-483-7
32. Emflaza (tn)
33. Mfcd00866106
34. Deflazacort [mi]
35. Deflazacort [inn]
36. Deflazacort (usan/inn)
37. Deflazacort [usan]
38. Schembl4018
39. Deflazacort [mart.]
40. Deflazacort [usp-rs]
41. Deflazacort [who-dd]
42. Dl-458it
43. Gtpl9477
44. Chembl1201891
45. Dtxsid9020378
46. Deflazacort, >=98% (hplc)
47. Deflazacort [orange Book]
48. Chebi:135720
49. Hms3714d15
50. Bcp08474
51. Zinc4212809
52. Tox21_112506
53. Tox21_301415
54. Bbl036672
55. S1888
56. Stl559051
57. Akos015895199
58. Tox21_112506_1
59. Ccg-220817
60. Db11921
61. Ks-1158
62. Ncgc00255189-01
63. Ncgc00263521-01
64. 11beta,21-dihydroxy-2'-methyl-5'betah-pregna-1,4-dieno(17,16-d)oxazole-3,20-dione 21-acetate
65. Hy-13609
66. Deflazacort 100 Microg/ml In Acetonitrile
67. D4523
68. D03671
69. T70289
70. Ab01274724-01
71. Ab01274724_02
72. 484d470
73. Q779118
74. Q-101371
75. 3-amino-3-(4-chloro-3-nitro-phenyl)-propionicacid
76. Deflazacort, United States Pharmacopeia (usp) Reference Standard
77. 11b,21-dihydroxy-2'-methyl-5'bh-pregna-1,4-dieno[17,16-d]oxazole-3,20-dione 21-acetate
78. (11beta,16beta)-21-(acetyloxy)-11-hydroxy-2'-methyl-5'h-pregna-1,4-dieno(17,16-d)oxazole-3,20-dione
79. [2-[(1s,2s,4r,8s,9s,11s,12s,13r)-11-hydroxy-6,9,13-trimethyl-16-oxo-5-oxa-7-azapentacyclo[10.8.0.02,9.04,8.013,18]icosa-6,14,17-trien-8-yl]-2-oxoethyl] Acetate
80. 11.beta.,21-dihydroxy-2'-methyl-5'.beta.h-pregna-1,4-dieno(17,16-d)oxazole-3,20-dione 21-acetate
81. 2-((6ar,6bs,7s,8as,8bs,11ar,12as,12bs)-7-hydroxy-6a,8a,10-trimethyl-4-oxo-2,4,6a,6b,7,8,8a,8b,11a,12,12a,12b-dodecahydro-1h-naphtho[2',1':4,5]indeno[1,2-d]oxazol-8b-yl)-2-oxoethyl Acetate
82. 5'-beta-h-pregna-1,4-dieno(17,16-d)oxazole-3,20-dione, 11-beta,21-dihydroxy-2'-methyl-, 21-acetate
83. 5'h-pregna-1,4-dieno(17,16-d)oxazole-3,20-dione, 21-(acetyloxy)-11-hydroxy-2'-methyl-, (11.beta.,16.beta.)-
84. 5'h-pregna-1,4-dieno(17,16-d)oxazole-3,20-dione, 21-(acetyloxy)-11-hydroxy-2'-methyl-, (11beta,16beta)-
| Molecular Weight | 441.5 g/mol |
|---|---|
| Molecular Formula | C25H31NO6 |
| XLogP3 | 2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 4 |
| Exact Mass | 441.21513771 g/mol |
| Monoisotopic Mass | 441.21513771 g/mol |
| Topological Polar Surface Area | 102 Ų |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 996 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 8 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Deflazacort is indicated for the treatment of Duchenne Muscular Dystrophy (DMD) in patients 2 years of age and older.
Deflazacort exerts anti-inflammatory activity in DMD, likely improving various symptoms, including muscle weakness and cardiorespiratory symptoms in addition to delaying their onset. This allows for an increased quality of life and prevents the necessity for surgical procedures, such as those for scoliosis, which is associated with DMD. Studies showed significant preservation of muscle mass in patients generally treated with 0.9 mg/kg/day of deflazacort compared to a control group. The following findings are based on clinical studies using deflazacort on a long term basis: **Effects on muscle strength** At age 16, individuals treated with long-term deflazacort had 63 4% score in muscle strength compared to a mean muscle strength score of 31 3% for control patients. Significant improvements in climbing stairs and rising from a supine position were also seen in patients taking deflazacort. **Effects on ambulation** Ambulation was significantly higher by 12 years of age and 18 years of age in patients taking deflazacort when compared with the control group. The control group showed a mean loss of ambulation of 2 years sooner than with deflazacort treatment. **Effects on cardiac function** Mean left ventricular ejection fraction (a measure of cardiac function) was higher in patients treated with deflazacort over the long term. Preservation of cardiac function was demonstrated by a mean difference in ejection fraction of about 7%, favoring study groups taking deflazacort over control groups. **Effects on spinal alignment** Children treated with deflazacort also significantly lowered the rate and severity of scoliosis and eliminated the need for scoliosis surgery after long-term treatment.
Anti-Inflammatory Agents
Substances that reduce or suppress INFLAMMATION. (See all compounds classified as Anti-Inflammatory Agents.)
Immunosuppressive Agents
Agents that suppress immune function by one of several mechanisms of action. Classical cytotoxic immunosuppressants act by inhibiting DNA synthesis. Others may act through activation of T-CELLS or by inhibiting the activation of HELPER CELLS. While immunosuppression has been brought about in the past primarily to prevent rejection of transplanted organs, new applications involving mediation of the effects of INTERLEUKINS and other CYTOKINES are emerging. (See all compounds classified as Immunosuppressive Agents.)
H02AB13
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
H - Systemic hormonal preparations, excl. sex hormones and insulins
H02 - Corticosteroids for systemic use
H02A - Corticosteroids for systemic use, plain
H02AB - Glucocorticoids
H02AB13 - Deflazacort
Absorption
Deflazacort is rapidly absorbed after oral administration with peak concentration occurring within 1-2 hours. One pharmacokinetic study determined an AUC (area under the curve) of 280 ng/ml h. The bioavailability of both the oral suspension and tablet are similar. In clinical studies, coadministration of deflazacort crushed with food or applesauce did not affect absorption or bioavailability.
Route of Elimination
Urinary excretion is the major route of deflazacort elimination, accounting for about about 70% of the excreted dose. The remainder of the dose (about 30%) is excreted in the feces. Elimination is almost completed by 24 hours post-dose. 21-deflazacort makes up about 18% of the eliminated drug in the urine.
Volume of Distribution
One study determined the volume of distribution to be 204 84 L.
Clearance
114 27 L/h, according to one noncompartmental pharmacokinetic study. The clearance of corticosteroids is enhanced in hypothyroid patients and increased in patients with hyperthyroidism. Dosing adjustments may be considered according to thyroid status. A study of corticosteroid clearance was performed in patients with a creatinine clearance of 15 mL/min or less, and determined that the active metabolite of deflazacort, 21-deflazacort was similar to that in patients with normal renal clearance.
After oral ingestion, deflazacort is deacetylated at position 21 by plasma esterases, producing the active metabolite 21-deflazacort. 21-deflazacort is then further metabolized by CYP3A4 to inactive metabolite products. Deflazacort 21-OH metabolism is extensive. The metabolite of deflazacort-21-OH is deflazacort 6-beta-OH.
The half-life of deflazacort ranges from 1.1 to 1.9 h
Deflazacort is a corticosteroid prodrug with an active metabolite, 21-deflazacort, which binds to the glucocorticoid receptor to exert anti-inflammatory and immunosuppressive effects on the body. The exact mechanism by which deflazacort exerts its therapeutic effects in patients with DMD is unknown but likely occurs via its anti-inflammatory activities.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
39
PharmaCompass offers a list of Deflazacort API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Deflazacort manufacturer or Deflazacort supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Deflazacort manufacturer or Deflazacort supplier.
A Deflazacortum manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Deflazacortum, including repackagers and relabelers. The FDA regulates Deflazacortum manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Deflazacortum API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Deflazacortum manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Deflazacortum supplier is an individual or a company that provides Deflazacortum active pharmaceutical ingredient (API) or Deflazacortum finished formulations upon request. The Deflazacortum suppliers may include Deflazacortum API manufacturers, exporters, distributors and traders.
click here to find a list of Deflazacortum suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Deflazacortum DMF (Drug Master File) is a document detailing the whole manufacturing process of Deflazacortum active pharmaceutical ingredient (API) in detail. Different forms of Deflazacortum DMFs exist exist since differing nations have different regulations, such as Deflazacortum USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Deflazacortum DMF submitted to regulatory agencies in the US is known as a USDMF. Deflazacortum USDMF includes data on Deflazacortum's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Deflazacortum USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Deflazacortum suppliers with USDMF on PharmaCompass.
A Deflazacortum written confirmation (Deflazacortum WC) is an official document issued by a regulatory agency to a Deflazacortum manufacturer, verifying that the manufacturing facility of a Deflazacortum active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Deflazacortum APIs or Deflazacortum finished pharmaceutical products to another nation, regulatory agencies frequently require a Deflazacortum WC (written confirmation) as part of the regulatory process.
click here to find a list of Deflazacortum suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Deflazacortum as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Deflazacortum API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Deflazacortum as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Deflazacortum and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Deflazacortum NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Deflazacortum suppliers with NDC on PharmaCompass.
Deflazacortum Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Deflazacortum GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Deflazacortum GMP manufacturer or Deflazacortum GMP API supplier for your needs.
A Deflazacortum CoA (Certificate of Analysis) is a formal document that attests to Deflazacortum's compliance with Deflazacortum specifications and serves as a tool for batch-level quality control.
Deflazacortum CoA mostly includes findings from lab analyses of a specific batch. For each Deflazacortum CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Deflazacortum may be tested according to a variety of international standards, such as European Pharmacopoeia (Deflazacortum EP), Deflazacortum JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Deflazacortum USP).

