
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
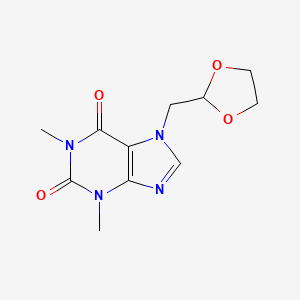

1. 2-(7'-theophyllinemethyl)1,3-dioxolane
2. Doxophylline
3. Tmdo
1. 69975-86-6
2. Doxophylline
3. Ansimar
4. Dioxyfilline
5. Ventax
6. Maxivent
7. Abc 12/3
8. Synasma
9. 7-(1,3-dioxolan-2-ylmethyl)theophylline
10. Mpm23gmo7z
11. Nsc-759645
12. Abc-12/3
13. 1h-purine-2,6-dione, 7-(1,3-dioxolan-2-ylmethyl)-3,7-dihydro-1,3-dimethyl-
14. 7-((1,3-dioxolan-2-yl)methyl)-1,3-dimethyl-1h-purine-2,6(3h,7h)-dione
15. Ncgc00159330-02
16. Dsstox_cid_2968
17. 7-(1,3-dioxolan-2-ylmethyl)-1,3-dimethyl-3,7-dihydro-1h-purine-2,6-dione
18. Dsstox_rid_76810
19. Dsstox_gsid_22968
20. Doxofilina
21. Doxofyllinum
22. Doxofylline [usan:inn]
23. Doxofilina [inn-spanish]
24. Doxofyllinum [inn-latin]
25. 7-((1,3-dioxolan-2-yl)methyl)-1,3-dimethyl-3,7-dihydro-1h-purine-2,6-dione
26. Cas-69975-86-6
27. Sr-01000789760
28. Einecs 274-239-6
29. Unii-mpm23gmo7z
30. Brn 0561195
31. 2-(7'-theophyllinemethyl)-1,3-dioxolane
32. Doxofylline,(s)
33. Maxivent (tn)
34. 2-(7'-teofillinmetil)-1,3-diossolano [italian]
35. Mfcd00865218
36. 2-(7'-teofillinmetil)-1,3-diossolano
37. Doxofylline [mi]
38. Doxofylline [inn]
39. Doxofylline (usan/inn)
40. Doxofylline [usan]
41. Theophylline, 7-(1,3-dioxolan-2-ylmethyl)-
42. 1h-purine-2,6-dione, 3,7-dihydro-7-(1,3-dioxolan-2-ylmethyl)-1,3-dimethyl-
43. 7-(1,3-dioxolon-2-ylmethyl)-1,2,3,6-tetrahydro-1,3-dimethyl-2,6-purindion
44. Doxofylline [mart.]
45. Schembl37963
46. Doxofylline [who-dd]
47. 5-26-14-00120 (beilstein Handbook Reference)
48. Mls001214637
49. Zinc3837
50. Chembl1527608
51. Dtxsid7022968
52. Chebi:94714
53. Doxofylline, >=98% (hplc)
54. 7-(1,3-dioxolan-2-ylmethyl)-1,3-dimethyl-purine-2,6-dione
55. Hms2090e04
56. Hms2877p10
57. Hms3652h03
58. Hms3714m21
59. Hms3885b09
60. Pharmakon1600-01502358
61. Abc-1213
62. Bcp12155
63. Hy-b0004
64. Tox21 111577
65. Tox21_111577
66. Bbl012263
67. Do-309
68. Nsc759645
69. S4164
70. Stk735429
71. 7-(1,3-dioxolan-2-ylmethyl)-3,7-dihydro-1,3-dimethyl-1h-purine-2,6-dione
72. Akos005535592
73. Tox21_111577_1
74. Ac-3492
75. Ccg-213050
76. Cs-8019
77. Db09273
78. Ds-7424
79. Nsc 759645
80. Ncgc00159330-03
81. Ncgc00159330-04
82. Ncgc00159330-10
83. Bd164389
84. Smr000543614
85. D4302
86. Ft-0630792
87. Sw199176-2
88. D03898
89. D90272
90. Ab00828111-06
91. Ab00828111_07
92. Ab00828111_08
93. 975d866
94. A836720
95. L001990
96. Q425887
97. 7-(1,3-dioxolan-2-ylmethyl)-1,3-dimethylxanthine
98. Sr-01000789760-2
99. Sr-01000789760-3
100. 7-(1,3-dioxolan-2-ylmethyl)-1,3-dimethyl-1h-purine-2,6-dione
101. Doxofylline, United States Pharmacopeia (usp) Reference Standard
102. 7-[(1,3-dioxolan-2-yl)methyl]-1,3-dimethyl-2,3,6,7-tetrahydro-1h-purine-2,6-dione
| Molecular Weight | 266.25 g/mol |
|---|---|
| Molecular Formula | C11H14N4O4 |
| XLogP3 | -0.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 266.10150494 g/mol |
| Monoisotopic Mass | 266.10150494 g/mol |
| Topological Polar Surface Area | 76.9 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 398 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Indicated for the treatment of chronic obstructive pulmonary disease (COPD), bronchial asthma and pulmonary disease with spastic bronchial component.
Doxofylline is a methylxanthine bronchodilator with potent bronchodilator activity comparable to that of theophylline. In animal studies, doxofylline demonstrated to attenuate bronchoconstriction, inflammatory actions and the release of thromboxane A2 (TXA2) when challenged with platelet-activating factor. Doxofylline does not demonstrate direct inhibition of any histone deacetylase (HDAC) enzymes or known PDE enzyme isoforms and did not act as an antagonist at A2 or A2 receptors. The affinity for adenosine A1, A2A and A2B receptors are reported to be all higher than 100 M. It only displays an inhibitory action against PDE2A1 and antagonism at adenosine A(2A) at high concentrations. A study demonstrated that doxofylline interacts with 2-adrenoceptors to induce blood vessel relaxation and airway smooth muscle relaxation. In dog studies, doxofylline decreased airway responsiveness at a dose that did not affect heart rate and respiratory rate.
Antitussive Agents
Agents that suppress cough. They act centrally on the medullary cough center. EXPECTORANTS, also used in the treatment of cough, act locally. (See all compounds classified as Antitussive Agents.)
Bronchodilator Agents
Agents that cause an increase in the expansion of a bronchus or bronchial tubes. (See all compounds classified as Bronchodilator Agents.)
Phosphodiesterase Inhibitors
Compounds which inhibit or antagonize the biosynthesis or actions of phosphodiesterases. (See all compounds classified as Phosphodiesterase Inhibitors.)
R - Respiratory system
R03 - Drugs for obstructive airway diseases
R03D - Other systemic drugs for obstructive airway diseases
R03DA - Xanthines
R03DA11 - Doxofylline
Absorption
After repeated administrations doxofylline reaches the steady-state in about 4 days. Following oral administration of 400 mg doxofylline twice daily for 5 days in adults with chronic bronchitis, the peak plasma concentrations (Cmax) at steady state ranged from 5.78 to 20.76 mcg/mL. The time to reach maximum concentration (Tmax) was 1.19 0.19 hours. The absolute bioavailability of doxofylline in healthy subjects was 63 25%.
Route of Elimination
Less than 4% of an orally administered dose is excreted unchanged in the urine due to extensive hepatic metabolism.
Volume of Distribution
Doxofylline demonstrates a short distribution phase following intravenous administration of 100 mg given in adults with chronic bronchitis. As methylxanthines are distributed to all body compartments, doxofylline may be detected in breast milk and placenta.
Clearance
Following oral administration of 400 mg doxofylline twice daily for 5 days, the total clearance was 555.2 180.6 mL/min.
Doxofylline is thought to undergo hepatic metabolism which accounts for 90% of total drug clearance. -hydroxymethyltheophylline was detected in the serum and urine after oral administration of 400 mg given in healthy subjects. The circulating metabolite was devoid of any significant pharmacological activity.
Following administration of a single intravenous dose of 100 mg over 10 minutes in adults with chronic bronchitis, the elimination half life of doxofylline was 1.83 0.37 hours. Following oral administration of 400 mg twice daily for 5 days in adults with chronic bronchitis, the mean elimination half life was 7.01 0.80 hours.
The main mechanism of action of doxofylline is unclear. One of the mechanisms of action of is thought to arise from the inhibition of phosphodiesterase activity thus increasing the levels of cAMP and promoting smooth muscle relaxation. The interaction of doxofylline with beta-2 adrenoceptors was demonstrated by a study using nonlinear chromatography, frontal analysis and molecular docking. Serine 169 and serine 173 residues in the receptor are thought to be critical binding sites for doxofylline where hydrogen bonds are formed. Via mediating the actions of beta-2 adrenoceptors, doxofylline induces blood vessel relaxation and airway smooth muscle relaxation. There is also evidence that doxofylline may exert anti-inflammatory actions by reducing the pleurisy induced by the inflammatory mediator platelet activating factor (PAF) according to a rat study. It is suggested that doxofylline may play an important role in attenuating leukocyte diapedesis, supported by mouse preclinical studies where doxofylline administration was associated with inhibited leukocyte migration across vascular endothelial cells in vivo and in vitro.Unlike theophylline, doxofylline does not inhibit tumor necrosis factor-induced interleukin (IL)-8 secretion in ASM cells.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26794
Submission : 2012-12-27
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 41746
Submission : 2025-04-10
Status : Active
Type : II

USDMF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7643
Submission : 1988-08-17
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Date of Issue : 2025-08-05
Valid Till : 2028-06-25
Written Confirmation Number : WC-0054
Address of the Firm : Block No. 82/B, ECP Road, At & Post Karakhadi, Tal - Padra, Dist. Baroda, Gujara...
Date of Issue : 2025-10-29
Valid Till : 2028-10-28
Written Confirmation Number : WC-0411
Address of the Firm : Gat No. 350, Wadiwarhe, Igatpuri, Nashik -422403, Taluka: Igatpuri, District: Na...

Date of Issue : 2022-08-16
Valid Till : 2025-07-15
Written Confirmation Number : WC-0042N
Address of the Firm : Plot Nos. 262 to 271, IDA, Pashamylaram, Sangareddy District- 502 307, Telangana...

Date of Issue : 2025-11-04
Valid Till : 2028-11-03
Written Confirmation Number : WC-0626
Address of the Firm : Plot No. 1, SR. No. 117,Behind Balaji Multiflex Pvt. Ltd., Village: - Amreli,Tal...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Registrant Name : Ace Biopharm Co., Ltd.
Registration Date : 2018-10-29
Registration Number : 20181029-209-J-98
Manufacturer Name : Ami Lifesciences Private Lim...
Manufacturer Address : Block No.82/B, ECP Road, At & Post. Karakhadi, Tal-Padra, City: Karakhadi-391 450, Di...
Registrant Name : Masung LS Co., Ltd.
Registration Date : 2022-05-03
Registration Number : 20181029-209-J-98(A)
Manufacturer Name : Ami Lifesciences Private Lim...
Manufacturer Address : Block No. 82/B, ECP Road At & Post. Karakhadi, Tal-Padra, City : Karakhadi-391450, Di...
Registrant Name : BT Pharm Co., Ltd.
Registration Date : 2020-12-24
Registration Number : 20201224-209-J-777
Manufacturer Name : Bajaj Healthcare Limited
Manufacturer Address : Block No. 588, Savli Karachia Road, At & Post – Gothada, Tal-Savli, Dist – Vadoda...

Registrant Name : Pampia Co., Ltd.
Registration Date : 2025-02-28
Registration Number : 20201224-209-J-777(1)
Manufacturer Name : Bajaj Healthcare Limited
Manufacturer Address : No. 588, Savli Karachia Road, At & Post – Gothada, Tal-Savli, Dist – Vadodara –...

Registrant Name : Daeshin Muyak Co., Ltd.
Registration Date : 2025-09-01
Registration Number : 20250901-209-J-1999
Manufacturer Name : Kores (India) Limited
Manufacturer Address : Plot No. 58/1, 58/2, 59A, 65A, 65B, 65C & 66A, MIDC Industrial Area, Dhatav, Tal - Ro...

Registrant Name : Masung LS Co., Ltd.
Registration Date : 2020-11-25
Registration Number : 20201125-209-J-437
Manufacturer Name : Cohance Lifesciences Limited...
Manufacturer Address : Plot Nos. 262 to 271, IDA, Pashamylaram(V), Patancheru(M), Sangareddy District Telang...

Registrant Name : Bukwang Pharmaceutical Co., Ltd.
Registration Date : 2021-02-25
Registration Number : 20201125-209-J-437(1)
Manufacturer Name : Cohance Lifesciences Limited...
Manufacturer Address : Plot Nos. 262 to 271, IDA, Pashamylaram (V), Patancheru (M), Sangareddy District Tela...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]NDC Package Code : 68022-7007
Start Marketing Date : 2016-02-02
End Marketing Date : 2027-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT FOR HUMAN P...

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]About the Company : Ami Lifesciences, established in 2006, is a rapidly growing API manufacturing company in India with strong capabilities in cardiovascular, anti-diabetic, CNS, and respiratory thera...
 ICE Pharma offers a unique range of bile acid products and best-in-class services for the pharmaceutical industry.
ICE Pharma offers a unique range of bile acid products and best-in-class services for the pharmaceutical industry.
About the Company : ICE Pharma manufactures active pharmaceutical ingredients, with a core focus on Ursodeoxycholic Acid (UDCA) for the treatment of liver diseases and gallstones. The company also dev...
About the Company : The Bakul Group of companies headquartered in Mumbai, India are engaged in the development and manufacture of high-quality Active Pharmaceutical Ingredients (APIs), Intermediates, ...

About the Company : Delta Finochem Pvt Ltd. is a privately owned and professionally managed company, accredited by WHO-GMP & ISO 9001:2015 (Year of establishment 1994) Delta Finochem is a leading manu...

About the Company : Guangzhou Tosun Pharmaceutical was founded in 1999, which mainly focuses on importation & exportation of Active Pharmaceutical Ingrediants, Chemical Raw Materials, Intermediate, Ex...

About the Company : Mangalmurti Bio-Chem Private Limited was established in 2010, initially focusing on the production of NPK fertilizer blends with a daily capacity of 200 tons. The company successfu...

About the Company : Public health is always an imperative that implies immense responsibility, not a mere re-adjustable option. As a company operating under world’s second largest industry of Pharma...

About the Company : Reine Lifescience is a highly acclaimed name among manufacturers and exporters of a wide variety of pharmaceuticals drugs.The Company Was Established in 1997 in GIDC, Ankleshwar In...

About the Company : S.S. Pharmachem is a professionally managed bulk drug and chemical manufacturing company led by Mr. Sagar S. Sangekar and Mr. Sanjay S. Sangekar. Established in 1987, the company i...

About the Company : SNEHA MEDICARE PVT LTD., one of the pioneers in API sourcing and supply in India, was established in 1985 by Shri. Kirti M Shah in the name of SNEHA CHEMICALS, which was later chan...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Bromo acetaldehyde dimethyl acetal
CAS Number : 7252-83-7
End Use API : Doxofylline
About The Company : The Bakul Group of companies headquartered in Mumbai, India are engaged in the development and manufacture of high-quality Active Pharmaceutical Ingredients (AP...

CAS Number : 4360-63-8
End Use API : Doxofylline
About The Company : The Bakul Group of companies headquartered in Mumbai, India are engaged in the development and manufacture of high-quality Active Pharmaceutical Ingredients (AP...

Bromoacetaldehyde dimethyl acetal
CAS Number : 7252-83-7
End Use API : Doxofylline
About The Company : Delta Finochem Pvt Ltd. is a privately owned and professionally managed company, accredited by WHO-GMP & ISO 9001:2015 (Year of establishment 1994) Delta Finoch...

Bromoacetaldehyde diethyl acetal
CAS Number : 2032-35-1
End Use API : Doxofylline
About The Company : Ocimum Labs was founded in 2018 and started its R&D activities with a cutting-edge facility. The Hyderabad-based firm focuses on process research and developmen...

CAS Number : CAS-4360-63-8
End Use API : Doxofylline
About The Company : Venkata Narayana Active Ingredients (Formerly Nutra Specialties Private Limited) Promoted by a well-known business house of India, Mr. Abhaya Kumar Jain who has...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Ansar
Dosage Form : Doxofylline 100Mg/10Ml 3 Units' Parenteral Use
Dosage Strength : 3 VIALS EV 100 mg 10 ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Doxofylline
Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral Use
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Ansar
Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral Use
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Ansar
Dosage Form : Doxofylline 400Mg 20 Joined' Oral Use
Dosage Strength : 20 CPR 400 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 400mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : India
Doxofylline; MONTELUKAST SODIUM
Brand Name :
Dosage Form : BILAYERED TABLET
Dosage Strength : 400MG; 10MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Granules
Dosage Strength : 400MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : China
Brand Name :
Dosage Form : Powder for Injection
Dosage Strength : 200mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : China

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : China
Brand Name :
Dosage Form : Injection
Dosage Strength :
Packaging : 100mL:300mg
Approval Date :
Application Number :
Regulatory Info :
Registration Country : China

Regulatory Info :
Registration Country : Italy
Brand Name : Doxovent
Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral Use
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Ansar
Dosage Form : Doxofylline 100Mg/10Ml 3 Units' Parenteral Use
Dosage Strength : 3 VIALS EV 100 mg 10 ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Doxofylline
Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral Use
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Ansar
Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral Use
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Ansar
Dosage Form : Doxofylline 400Mg 20 Joined' Oral Use
Dosage Strength : 20 CPR 400 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Regulatory Info :
Registration Country : Italy
Brand Name : Doxovent
Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral Use
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Italy
Brand Name : Doxovent
Dosage Form : Doxofylline 400Mg 20 Joined' Oral Use
Dosage Strength : 20 CPR 400 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : India
Doxofylline; MONTELUKAST SODIUM
Brand Name :
Dosage Form : BILAYERED TABLET
Dosage Strength : 400MG; 10MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info : Generic
Doxofylline; MONTELUKAST SODIUM
Dosage : BILAYERED TABLET
Dosage Strength : 400MG; 10MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Ampoule
Dosage Strength : 10MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Ampoule
Dosage Strength : 10MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Nepal
Brand Name : D-AASMA 200
Dosage Form : Tablet
Dosage Strength : 200MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Nepal

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 200MG
Brand Name : D-AASMA 200
Approval Date :
Application Number :
Registration Country : Nepal

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Granules
Dosage Strength : 400MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Granules
Dosage Strength : 400MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength :
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength :
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength :
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 400MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 400MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : China
Brand Name :
Dosage Form : Powder for Injection
Dosage Strength : 200mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : China

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Powder for Injection
Dosage Strength : 200mg
Brand Name :
Approval Date :
Application Number :
Registration Country : China

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 400mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 400mg
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 400MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Packaging :
Regulatory Info :
Dosage : Tablet
Dosage Strength : 400MG
Brand Name :
Approval Date :
Application Number :
Registration Country : India

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Dosage Form : Doxofylline 100Mg/10Ml 3 Units' ...
Dosage Strength : 3 VIALS EV 100 mg 10 ml
Price Per Pack (Euro) : 2.23
Published in :
Country : Italy
RX/OTC/DISCN : Class A

Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral U...
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Price Per Pack (Euro) : 4.22
Published in :
Country : Italy
RX/OTC/DISCN : Class A

Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral U...
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Price Per Pack (Euro) : 4.84
Published in :
Country : Italy
RX/OTC/DISCN : Class A

Dosage Form : Doxofylline 400Mg 20 Joined' Ora...
Dosage Strength : 20 CPR 400 mg
Price Per Pack (Euro) : 4.87
Published in :
Country : Italy
RX/OTC/DISCN : Class A

OTIFARMA Srl
Dosage Form : Doxofylline 20Mg/Ml 200Ml Oral U...
Dosage Strength : 1 Bottle 200 ml 20 mg/ml syrup
Price Per Pack (Euro) : 4.22
Published in :
Country : Italy
RX/OTC/DISCN : Class A

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
OTIFARMA Srl
Dosage Form : Doxofylline 400Mg 20 Joined' Ora...
Dosage Strength : 20 CPR 400 mg
Price Per Pack (Euro) : 4.05
Published in :
Country : Italy
RX/OTC/DISCN : Class A

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

REF. STANDARDS & IMPURITIES

CAS Number : 1429636-74-7
Quantity Per Vial :
Sale Unit :
Price :
Details : In stock
Monograph :
Storage :
Code/Batch No : D0033.02

CAS Number : 519-37-9
Quantity Per Vial :
Sale Unit :
Price :
Details : In stock
Monograph :
Storage :
Code/Batch No : D0033.10

CAS Number :
Quantity Per Vial :
Sale Unit :
Price :
Details : In stock
Monograph :
Storage :
Code/Batch No : D0033.04

CAS Number : 1174289-18-9
Quantity Per Vial :
Sale Unit :
Price :
Details : In stock
Monograph :
Storage :
Code/Batch No : D0033.05

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE
83
PharmaCompass offers a list of Doxofylline API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Doxofylline manufacturer or Doxofylline supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Doxofylline manufacturer or Doxofylline supplier.
A Ansimar manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ansimar, including repackagers and relabelers. The FDA regulates Ansimar manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ansimar API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ansimar manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ansimar supplier is an individual or a company that provides Ansimar active pharmaceutical ingredient (API) or Ansimar finished formulations upon request. The Ansimar suppliers may include Ansimar API manufacturers, exporters, distributors and traders.
click here to find a list of Ansimar suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Ansimar DMF (Drug Master File) is a document detailing the whole manufacturing process of Ansimar active pharmaceutical ingredient (API) in detail. Different forms of Ansimar DMFs exist exist since differing nations have different regulations, such as Ansimar USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ansimar DMF submitted to regulatory agencies in the US is known as a USDMF. Ansimar USDMF includes data on Ansimar's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ansimar USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ansimar suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Ansimar Drug Master File in Korea (Ansimar KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Ansimar. The MFDS reviews the Ansimar KDMF as part of the drug registration process and uses the information provided in the Ansimar KDMF to evaluate the safety and efficacy of the drug.
After submitting a Ansimar KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Ansimar API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Ansimar suppliers with KDMF on PharmaCompass.
A Ansimar written confirmation (Ansimar WC) is an official document issued by a regulatory agency to a Ansimar manufacturer, verifying that the manufacturing facility of a Ansimar active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Ansimar APIs or Ansimar finished pharmaceutical products to another nation, regulatory agencies frequently require a Ansimar WC (written confirmation) as part of the regulatory process.
click here to find a list of Ansimar suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Ansimar as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Ansimar API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Ansimar as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Ansimar and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Ansimar NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Ansimar suppliers with NDC on PharmaCompass.
Ansimar Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ansimar GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ansimar GMP manufacturer or Ansimar GMP API supplier for your needs.
A Ansimar CoA (Certificate of Analysis) is a formal document that attests to Ansimar's compliance with Ansimar specifications and serves as a tool for batch-level quality control.
Ansimar CoA mostly includes findings from lab analyses of a specific batch. For each Ansimar CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ansimar may be tested according to a variety of international standards, such as European Pharmacopoeia (Ansimar EP), Ansimar JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ansimar USP).

