
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Canada
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Ago 178
2. Ago-178
3. Ago178
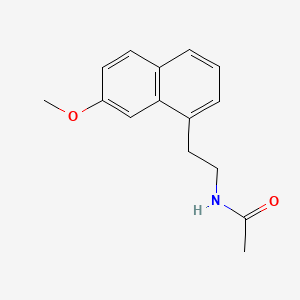
4. N-(2-(7-methoxy-1-naphthyl)ethyl)acetamide
5. S 20098
6. S-20098
7. S20098
8. Thymanax
9. Valdoxan
1. 138112-76-2
2. Thymanax
3. Valdoxan
4. N-[2-(7-methoxynaphthalen-1-yl)ethyl]acetamide
5. N-(2-(7-methoxynaphthalen-1-yl)ethyl)acetamide
6. S20098
7. S-20098
8. Agomelatine [inn]
9. Melitor
10. Ago-178
11. S 20098
12. Acetamide, N-[2-(7-methoxy-1-naphthalenyl)ethyl]-
13. N-(2-(7-methoxy-1-naphthyl)ethyl)acetamide
14. N-(2-(7-methoxynaphth-1-yl)ethyl)acetamide
15. N-(2-(7-methoxy-1-naphthalenyl)ethyl)acetamide
16. Chembl10878
17. 137r1n49ad
18. N-[2-(7-methoxy-1-naphthalenyl)ethyl]acetamide
19. Agomelatine (inn)
20. Ncgc00253646-01
21. N-[2-(7-methoxy-1-naphthyl)ethyl]acetamide
22. Acetamide, N-(2-(7-methoxy-1-naphthalenyl)ethyl)-
23. N-[2-(7-methoxy-naphthalen-1-yl)-ethyl]-acetamide
24. Ago 178
25. Valdoxan (tn)
26. Unii-137r1n49ad
27. Ago178
28. N-[2-(7-methoxynaphth-1-yl)ethyl]acetamide
29. Sr-01000944938
30. Agomelatine [inn:ban]
31. Ago178c
32. Agomelatine-[d4]
33. Awy
34. Agomelatine- Bio-x
35. Mfcd00916659
36. Agomelatine [mi]
37. Agomelatine [mart.]
38. Dsstox_cid_31431
39. Dsstox_rid_97317
40. Dsstox_gsid_57642
41. Agomelatine [who-dd]
42. Gtpl198
43. Mls006011913
44. Schembl114476
45. Agomelatine [ema Epar]
46. Zinc5608
47. Dtxsid3057642
48. Agomelatine, >=98% (hplc)
49. Chebi:134990
50. Bcpp000421
51. Hms3604n09
52. Hms3648g18
53. Hms3654b07
54. Hms3884a07
55. Bcp02084
56. Tox21_113772
57. Bbl029084
58. Bdbm50035179
59. Pdsp1_001305
60. Pdsp1_001784
61. Pdsp2_001289
62. Pdsp2_001767
63. S1243
64. Stl237728
65. Akos005145681
66. Ac-3395
67. Bcp9000250
68. Ccg-221950
69. Cs-0740
70. Db06594
71. Ks-1247
72. Sb19508
73. Ncgc00253646-10
74. Ba167079
75. Hy-17038
76. Smr002530056
77. Am20090763
78. B2262
79. Cas-138112-76-2
80. Ft-0657383
81. Sw219177-1
82. A19445
83. D02578
84. Ab01274769-01
85. Ab01274769_02
86. 112a762
87. L000528
88. Q395229
89. Q-102507
90. Sr-01000944938-1
91. Sr-01000944938-3
92. F0001-2383
93. (4-chlorophenyl)-[(9h-fluoren-9-ylmethoxycarbonylamino)]-aceticacid
94. N-[2-(7-methoxy-naphthalen-1-yl)-ethyl]-acetamide(agomelatine)
| Molecular Weight | 243.30 g/mol |
|---|---|
| Molecular Formula | C15H17NO2 |
| XLogP3 | 2.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 4 |
| Exact Mass | 243.125928785 g/mol |
| Monoisotopic Mass | 243.125928785 g/mol |
| Topological Polar Surface Area | 38.3 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 280 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Agomelatine is indicated to treat major depressive episodes in adults.
Treatment of major depressive episodes in adults.
Treatment of major depressive episodes in adults.
Treatment of major depressive episodes
Agomelatine resynchronises circadian rhythms in animal models of delayed sleep phase syndrome and other circadian rhythm disruptions. It increases noradrenaline and dopamine release specifically in the frontal cortex and has no influence on the extracellular levels of serotonin. Agomelatine has shown an antidepressant-like effect in animal depression models, (learned helplessness test, despair test, and chronic mild stress) circadian rhythm desynchronisation, and in stress and anxiety models. In humans, agomelatine has positive phase shifting properties; it induces a phase advance of sleep, body temperature decline and melatonin onset. Controlled studies in humans have shown that agomelatine is as effective as the SSRI antidepressants paroxetine and sertraline in the treatment of major depression
Hypnotics and Sedatives
Drugs used to induce drowsiness or sleep or to reduce psychological excitement or anxiety. (See all compounds classified as Hypnotics and Sedatives.)
N06AX22
N06AX22
N06AX22
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
N - Nervous system
N06 - Psychoanaleptics
N06A - Antidepressants
N06AX - Other antidepressants
N06AX22 - Agomelatine
Absorption
Bioavailability is less than 5%.
Hepatic (90% CYP1A2 and 10% CYP2C9).
<2 hours
The novel antidepressant agent, agomelatine, behaves as an agonist at melatonin receptors (MT1 and MT2) and as an antagonist at serotonin (5-HT)(2C) receptors.
 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38958
Submission : 2023-09-27
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26965
Submission : 2013-03-08
Status : Inactive
Type : II




NDC Package Code : 50370-0041
Start Marketing Date : 2013-06-21
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Aging Life Science Co., Ltd.
Registration Date : 2023-10-05
Registration Number : Su434-59-ND
Manufacturer Name : MSN Pharmachem Private Limited
Manufacturer Address : Plot. No. 182 to 186, 192-A, 193 to 197 & 212/A,B,C,D, Phase-II, IDA Pashamylaram, Pashamylaram (Village), Patancheru (Mandal), Sangareddy District, Telangana, Pincode-502 307, India.



Date of Issue : 2025-09-29
Valid Till : 2028-08-09
Written Confirmation Number : WC-0072
Address of the Firm :

Registrant Name : Pharmapia Co., Ltd.
Registration Date : 2021-04-16
Registration Number : Number 50-1-ND(1)
Manufacturer Name : Symed Labs Limited
Manufacturer Address : Plot No.25/B, Phase-III, IDA Jeedimetla Hyderabad-500 055, Telangana State, India


Date of Issue : 2023-11-15
Valid Till : 2026-05-11
Written Confirmation Number : WC-0273
Address of the Firm :

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26965
Submission : 2013-03-08
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38958
Submission : 2023-09-27
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27206
Submission : 2013-06-21
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Date of Issue : 2025-08-22
Valid Till : 2028-08-21
Written Confirmation Number : WC-0082
Address of the Firm : Karakhadi, Plot No. 842-843, At - Karakhdi, Tal. - Padra, Dist -Vadodara, Gujara...

Date of Issue : 2026-04-15
Valid Till : 2029-04-14
Written Confirmation Number : WC-0214
Address of the Firm : Gut No. 546,571,519 and 520, Village Kumbhavali, Boisar, Tal & Dist- Palghar, Pi...

Date of Issue : 2025-09-08
Valid Till : 2028-08-15
Written Confirmation Number : WC-0022
Address of the Firm : Plot No. 182 to 186, 192-A and 193 to 197 & 212/A,B,C,D Phase-ll, IDA, Pashamyla...

Date of Issue : 2023-11-15
Valid Till : 2026-05-11
Written Confirmation Number : WC-0273
Address of the Firm : Plot No: 34A, Road No: 1, Jawaharlal Nehru Pharma City, Thanam Village, Parawada...

Date of Issue : 2025-09-29
Valid Till : 2028-08-09
Written Confirmation Number : WC-0072
Address of the Firm : Unit-II, Plot No.: 25/B, Phase -III, Jeedimetla(V),Quthbullapur (M), Medchal -Ma...

Agomelatine citric acid co-crystal IH
Date of Issue : 2023-03-11
Valid Till : 2026-10-14
Written Confirmation Number : WC-0133
Address of the Firm : Plot No 3501 to 3515, 6301 to 6313 & 16 Meter Road/C, G.I.D.C Estate, Ankleshwar...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

ANALYTICAL

ABOUT THIS PAGE
21
PharmaCompass offers a list of Agomelatine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Agomelatine manufacturer or Agomelatine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Agomelatine manufacturer or Agomelatine supplier.
A Agomelatine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Agomelatine, including repackagers and relabelers. The FDA regulates Agomelatine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Agomelatine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Agomelatine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Agomelatine supplier is an individual or a company that provides Agomelatine active pharmaceutical ingredient (API) or Agomelatine finished formulations upon request. The Agomelatine suppliers may include Agomelatine API manufacturers, exporters, distributors and traders.
click here to find a list of Agomelatine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Agomelatine DMF (Drug Master File) is a document detailing the whole manufacturing process of Agomelatine active pharmaceutical ingredient (API) in detail. Different forms of Agomelatine DMFs exist exist since differing nations have different regulations, such as Agomelatine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Agomelatine DMF submitted to regulatory agencies in the US is known as a USDMF. Agomelatine USDMF includes data on Agomelatine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Agomelatine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Agomelatine suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Agomelatine Drug Master File in Korea (Agomelatine KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Agomelatine. The MFDS reviews the Agomelatine KDMF as part of the drug registration process and uses the information provided in the Agomelatine KDMF to evaluate the safety and efficacy of the drug.
After submitting a Agomelatine KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Agomelatine API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Agomelatine suppliers with KDMF on PharmaCompass.
A Agomelatine written confirmation (Agomelatine WC) is an official document issued by a regulatory agency to a Agomelatine manufacturer, verifying that the manufacturing facility of a Agomelatine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Agomelatine APIs or Agomelatine finished pharmaceutical products to another nation, regulatory agencies frequently require a Agomelatine WC (written confirmation) as part of the regulatory process.
click here to find a list of Agomelatine suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Agomelatine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Agomelatine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Agomelatine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Agomelatine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Agomelatine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Agomelatine suppliers with NDC on PharmaCompass.
Agomelatine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Agomelatine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Agomelatine GMP manufacturer or Agomelatine GMP API supplier for your needs.
A Agomelatine CoA (Certificate of Analysis) is a formal document that attests to Agomelatine's compliance with Agomelatine specifications and serves as a tool for batch-level quality control.
Agomelatine CoA mostly includes findings from lab analyses of a specific batch. For each Agomelatine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Agomelatine may be tested according to a variety of international standards, such as European Pharmacopoeia (Agomelatine EP), Agomelatine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Agomelatine USP).

