Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
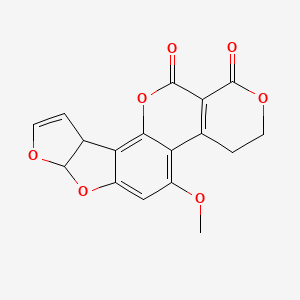

1. Aflatoxin G(1)
1. Aflatoxin
2. 1165-39-5
3. Aflatoxin G
4. Aflatoxin, Crude
5. 1402-68-2
6. 11-methoxy-6,8,16,20-tetraoxapentacyclo[10.8.0.02,9.03,7.013,18]icosa-1,4,9,11,13(18)-pentaene-17,19-dione
7. 5-(methyloxy)-3,4,7a,10a-tetrahydro-1h,12h-furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c]chromene-1,12-dione
8. Aflatoxing
9. Hsdb 3411
10. 1h,12h-furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione, 3,4,7a,10a-tetrahydro-5-methoxy-, (7ar-cis)-
11. Aflatoxin G1 2 Microg/ml In Acetonitrile
12. Schembl50772
13. Aflatoxin G1 In Acetonitrile
14. Aflatoxin G1-13c17 Solution
15. Aflatoxin G1, Reference Material
16. Chebi:190729
17. Mfcd28143303
18. Aflatoxin G1, From Aspergillus Flavus
19. Akos015896946
20. Ft-0621934
21. A937071
22. Q59260340
23. Aflatoxin G1 Solution, 2 Mug/ml In Acetonitrile, Analytical Standard
24. Aflatoxin G1 Solution, 3.78 Mug/g In Acetonitrile, Erm(r) Certified Reference Material
25. (7ar,10as)-5-methoxy-3,4,7a,10a-tetrahydro-1h,12h-furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c]chromene-1,12-dione
26. 11-methoxy-6,8,16,20-tetraoxapentacyclo[10.8.0.0^{2,9}.0^{3,7}.0^{13,18}]icosa-1,4,9,11,13(18)-pentaene-17,19-dione
27. 1h,12h-furo[3',2':4,5]furo[2,3-h]pyrano[3,4-c][1]benzopyran-1,12-dione,3,4,7a,10a-tetrahydro-5-methoxy-, (7ar,10as)-
28. Aflatoxin G1 Solution, Certified Reference Material, 3 Mug/ml In Benzene:acetonitrile (98:2), Ampule Of 1 Ml
| Molecular Weight | 328.27 g/mol |
|---|---|
| Molecular Formula | C17H12O7 |
| XLogP3 | 1.8 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 1 |
| Exact Mass | 328.05830272 g/mol |
| Monoisotopic Mass | 328.05830272 g/mol |
| Topological Polar Surface Area | 80.3 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 666 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 2 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Aflatoxins B1 & G1 & their metabolites exist in systemic blood as protein conjugates. This conjugation is specific to plasma albumin & proceeds enzymatically by liver & kidney cells. The albumin-aflatoxin conjugate is permanent & conjugation is an irreversible one.
PMID:6811900 Nassar AY et al; Mycopathologia 79 (1): 35 (1982)
Three groups of four Large White sows were fed diets containing either 800 ppb purified aflatoxin B1 (group 1), 800 ppb purified aflatoxin G1 (group 2) or 400 ppb B1 and 400 ppb G1 (group 3) throughout gestation and lactation. A control group of four sows was fed a diet free of aflatoxins. Aflatoxins B1 and M1 were found in milk samples taken five and 25 days after parturition from the sows of group 1, aflatoxin G1 was present in the milk of the sows of group 2 and all three aflatoxins were present in samples from the sows of group 3. The concentration of aflatoxin in the milk was about 1000-fold lower than that in the feed, but increased over the 25 days after parturition.
PMID:9392065 Silvotti L et al; Vet Rec 141(18): 469-472 (1997)
Yields aflatoxin B3 in rhizopus ... yields aflatoxin gm1 in rat. From table/
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. A-27
Aflatoxin B1, aflatoxin B2, & aflatoxin G1 admin iv to rats were rapidly metabolized to 7 groups of metabolites each, 6 of which were excreted in the bile. All 3 toxins were hydroxylated at the 2- & 4-positions. Bile from the rats that had received aflatoxin G1 contained glucuronide.
PMID:5034521 Dann RE et al; Res Commun Chem Pathol Pharmacol 3 (3): 667 (1972)
...The incubation of human liver microsomes with aflatoxin B1 /or/ aflatoxin G1 ...yielded genotoxic metabolites that induced umuC gene expression in Salmonella typhimurium (TA-1535/psK1002). The rank order of genotoxic potency was ...aflatoxin B1>aflatoxin G1. Microsomal activation of the ...aflatoxins was completely inhibited upon incubation with polyclonal antibodies against p450NF, and immunochemical determinations of p450NF /(nifedipine oxidase)/ in the liver microsomal preparations were correlated with the microsomal activation of ...aflatoxin G1 and aflatoxin B1. P450NF converted the ...aflatoxins to genotoxic metabolites in a reconstituted monooxygenase system containing the purified enzyme and an NADPH generating system. ...
PMID:2492107 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC286490 Shimada T, Guengerich FP; Proceedings of the National Academy of Sciences of the United States of America 86 (2): 462-5 (1989)
Ability of aflatoxin B1, aflatoxin B2, & aflatoxin G1 to inhibit RNA polymerase activity & decr RNA content in rat hepatocyte nuclei was qualitatively similar to the carcinogenic & acute & subacute toxic actions of these compounds. Aflatoxin g1 induced a rapid macrosegregation of the fibrillar & granular portions of the hepatocyte nucleolus.
PMID:5120291 Edwards GS et al; Cancer Res 31 (12): 1943 (1971)
In vitro studies with human liver indicate that the major catalyst involved in the bioactivation of the hepato-carcinogen aflatoxin B1 to its genotoxic 2,3-epoxide derivative is cytochrome P-450NF, a previously characterized protein that also catalyzes the oxidation of nifedipine and other dihydropyridines, quinidine, macrolide antibiotics, various steroids, and other compounds. ...Cytochrome p-450NF or a closely related protein also appears to be the major catalyst involved in the activation of aflatoxin G1 and sterigmatocystin, the latter compound being more genotoxic than aflatoxin B1 in these systems. Several drugs and conditions are known to influence the levels and activity of cytochrome p-450NF in human liver, and the activity of the enzyme can be estimated by noninvasive assays. These findings provide a test system for the hypothesis that a specific human disease state (liver cancer) is linked to the level of oxidative metabolism in populations in which aflatoxin ingestion is high.
PMID:2492107 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC286490 Shimada T, Guengerich FP; Proc Natl Acad Sci U S A 86 (2): 462-5 (1989)
Aflatoxin B1, aflatoxin G1 & aflatoxin G2 inhibited incorporation of (14)carbon labeled orotic acid into the RNA of rat liver slices at toxin concentrations of 100 umole/3 mL. Respective percent inhibitions were approx 90, 40, & 20. Aflatoxin B1, 20 umole/3 mL, aflatoxin G1, 150 umole/3 mL, & aflatoxin G2, 230 umole/3 mL inhibited the incorporation of (14)carbon labeled dl-leucine into proteins of rat liver slices by 32%, 35%, & 38%, respectively.
PMID:6033768 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1270395 Clifford JI et al; Biochem J 103: 258 (1967)
Phagocytosis, intracellular killing of Candida albicans, and superoxide production by rat peritoneal macrophages exposed to aflatoxins B1, B2, G1, G2, B2a, and M1 at several times and concn were analyzed to evaluate the intensity of a depressive effect for each mycotoxin. All aflatoxins used at very low concn had a depressive effect on the functions of macrophages. The biggest impairment of phagocytosis, intracellular killing, and spontaneous superoxide production was observed in macrophages exposed to aflatoxins B1 and M1.
PMID:2176448 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC184993 Cusumano V et al; Appl Environ Microbiol 56 (11): 3482-4 (1990)
Among the toxic aflatoxins, aflatoxins B1 and G1 are the most biologically active, but other derivatives also exhibit carcinogenicity. Aflatoxin B1 requires metabolic activation by the cytochrome p450 dependent mixed-function oxidase to be converted to the reactive 2,3-epoxide, the ultimate carcinogen. The aflatoxins, eg, aflatoxin B1, are genotoxic carcinogens and the reactive metabolites react with DNA. The major adduct formed with DNA in intracellular reactions is formed from the 2-position of aflatoxin B1 and the N-7 position of guanine in DNA.
Haddad, L.M., Clinical Management of Poisoning and Drug Overdose. 2nd ed. Philadelphia, PA: W.B. Saunders Co., 1990., p. 515
ABOUT THIS PAGE
77
PharmaCompass offers a list of Aflatoxin G1 API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Aflatoxin G1 manufacturer or Aflatoxin G1 supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Aflatoxin G1 manufacturer or Aflatoxin G1 supplier.
A Aflatoxin G1 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Aflatoxin G1, including repackagers and relabelers. The FDA regulates Aflatoxin G1 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Aflatoxin G1 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Aflatoxin G1 supplier is an individual or a company that provides Aflatoxin G1 active pharmaceutical ingredient (API) or Aflatoxin G1 finished formulations upon request. The Aflatoxin G1 suppliers may include Aflatoxin G1 API manufacturers, exporters, distributors and traders.
Aflatoxin G1 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Aflatoxin G1 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Aflatoxin G1 GMP manufacturer or Aflatoxin G1 GMP API supplier for your needs.
A Aflatoxin G1 CoA (Certificate of Analysis) is a formal document that attests to Aflatoxin G1's compliance with Aflatoxin G1 specifications and serves as a tool for batch-level quality control.
Aflatoxin G1 CoA mostly includes findings from lab analyses of a specific batch. For each Aflatoxin G1 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Aflatoxin G1 may be tested according to a variety of international standards, such as European Pharmacopoeia (Aflatoxin G1 EP), Aflatoxin G1 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Aflatoxin G1 USP).

