
Synopsis
Synopsis
0
JDMF
0
KDMF
0
VMF
0
South Africa
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
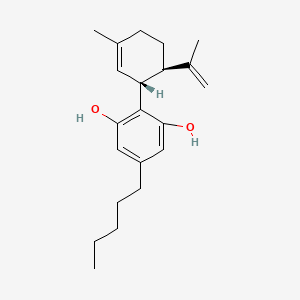

1. 1,3-benzenediol, 2-(3-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl)-5-pentyl-, (1r-trans)-
2. Epidiolex
1. 13956-29-1
2. (-)-cannabidiol
3. (-)-trans-cannabidiol
4. Epidiolex
5. Cbd
6. (-)-cbd
7. Gwp42003-p
8. Cannabidiol [usan]
9. (-)-trans-2-p-mentha-1,8-dien-3-yl-5-pentylresorcinol
10. Delta1(2)-trans-cannabidiol
11. 2-[(1r,6r)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol
12. 19gbj60sn5
13. Chembl190461
14. 3556-78-3
15. .delta.1(2)-trans-cannabidiol
16. Chebi:69478
17. Btx-1204
18. Btx-1503
19. Gwp42003
20. Gwp-42003-p
21. Gwp-42003
22. Resorcinol, 2-p-mentha-1,8-dien-3-yl-5-pentyl-, (-)-(e)-
23. 2-[1r-3-methyl-6r-(1-methylethenyl)-2-cyclohexen-1-yl]-5-pentyl-1,3-benzenediol
24. C21h30o2
25. 1,3-benzenediol, 2-(3-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl)-5-pentyl-, (1r-trans)-
26. 2-((1r,6r)-6-isopropenyl-3-methyl-cyclohex-2-enyl)-5-pentyl-benzene-1,3-diol
27. (1'r,2'r)-5'-methyl-4-pentyl-2'-(prop-1-en-2-yl)-1',2',3',4'-tetrahydrobiphenyl-2,6-diol
28. Cannabidiol Solution
29. 2-[(1r,6r)-3-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl]-5-pentyl-1,3-benzenediol
30. (-)-cannabidiol (cbd)
31. Cannabidiolum
32. Unii-19gbj60sn5
33. 1,3-benzenediol, 2-[(1r,6r)-3-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl]-5-pentyl-
34. 1,3-benzenediol, 2-[3-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl]-5-pentyl-, (1r-trans)-
35. Epidiolex (tn)
36. 1,3-benzenediol, 2-((1r,6r)-3-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl)-5-pentyl-
37. P0t
38. Cannabidiol (7ci)
39. Cardiolrx
40. (+/-)-cannabidiol
41. Cannabidiol [mi]
42. Cannabidiol [inn]
43. Cannabidiol (usan/inn)
44. D1(2)-trans-cannabidiol
45. Cannabidiol [inci]
46. Cannabidiol [mart.]
47. Cannabidiol [usp-rs]
48. Cannabidiol [who-dd]
49. (-)-cannabidiol (synthetic)
50. Schembl119679
51. Gtpl4150
52. Zyn002
53. 2-(6-isopropenyl-3-methyl-2-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol #
54. 2-(6-isopropenyl-3-methyl-cyclohex-2-enyl)-5-pentyl-benzene-1,3-diol
55. Delta(1(2))-trans-cannabidiol
56. Cannabidiol [orange Book]
57. Dtxsid00871959
58. Dtxsid301038839
59. Zinc4097406
60. Bdbm50121429
61. Bdbm50318484
62. Cannabidiol Solution [usp-rs]
63. Akos032948358
64. Db09061
65. Ncgc00386518-01
66. (-)-cannabidiol 1.0 Mg/ml In Methanol
67. Ac-34022
68. Db-093531
69. Sativex (cbd + Thc, Fixed-dose Oral Spray)
70. C07578
71. D10915
72. Nabiximols (cbd + Thc, Fixed-dose Oral Spray)
73. Q422917
74. (-)-cannabidiol (cbd) 100 Microg/ml In Methanol
75. (-)-cannabidiol (cbd) 1000 Microg/ml In Methanol
76. (-)-cannabidiol (cbd) 250 Microg/ml In Acetonitrile
77. (3r,4r)-2-p-mentha-1,8-dien-3-yl-5-pentylresorcinol
78. Resorcinol, 2-p-mentha-1,8-dien-3-yl-5-pentyl-, (+-)-
79. Resorcinol, 2-p-mentha-1,8-dien-3-yl-5-pentyl-, Trans-(-)- (8ci)
80. 1,3-benzenediol, 2-(4-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl)-5-pentyl-, Trans-
81. 2-((1r,6r)-3-methyl-6-(1-methylethenyl)cyclohex-2-enyl)-5-pentylbenzene-1,3-diol
82. 2-(3-methyl-6-prop-1-en-2-yl-1-cyclohex-2-enyl)-5-pentyl-benzene-1,3-diol
83. 2-(4-methyl-6-(1-methylethenyl)-2-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol Trans-
84. 2-(6-isopropenyl-3-methyl-cyclohex-2-enyl)-5-pentyl-benzene-1,3-diol (cannabidiol)
85. 2-[(1r,6r)-3-methyl-6-prop-1-en-2-yl-1-cyclohex-2-enyl]-5-pentylbenzene-1,3-diol
86. 2-[3-methyl-6-(methylethenyl)-2-cyclohexen-1-yl]-5-pentyl-1,3-benzenediol
87. Cannabidiol Solution, ~10 Mg/ml In Ethanol, Analytical Standard, For Drug Analysis
88. Cannabidiol Solution, 1.0 Mg/ml In Methanol, Ampule Of 1 Ml, Certified Reference Material
89. Cannabidiol Solution, 1.0 Mg/ml In Methanol, Analytical Standard, For Drug Analysis
90. (1'r,2'r)-5'-methyl-4-pentyl-2'-(prop-1-en-2-yl)-1',2',3',4'-tetrahydro-[1,1'-biphenyl]-2,6-diol
91. (1'r,2'r)-5'-methyl-4-pentyl-2'-(prop-1-en-2-yl)-1',2',3',4'-tetrahydro[1,1'-biphenyl]-2,6-diol
92. 2-((1r,6r)-3-methyl-6-(prop-1-en-2-yl)cyclohex-2-en-1-yl)-5-pentylbenzene-1,3-diol
| Molecular Weight | 314.5 g/mol |
|---|---|
| Molecular Formula | C21H30O2 |
| XLogP3 | 6.5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 6 |
| Exact Mass | 314.224580195 g/mol |
| Monoisotopic Mass | 314.224580195 g/mol |
| Topological Polar Surface Area | 40.5 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 414 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
When used in combination with delta-9-tetrahydrocannabinol as the product Sativex, cannabidiol was given a standard marketing authorization (ie. a Notice of Compliance (NOC)) by Health Canada for the following indications: 1) as adjunctive treatment for symptomatic relief of spasticity in adult patients with multiple sclerosis (MS) who have not responded adequately to other therapy and who demonstrate meaningful improvement during an initial trial of therapy; Due to the need for confirmatory studies to verify the clinical benefit coupled with the promising nature of the clinical evidence, Sativex was also given a Notice of Compliance with Conditions (NOC/c) by Health Canada for the following indications: 1) as adjunctive treatment for the symptomatic relief of neuropathic pain in adult patients with multiple sclerosis; 2) as adjunctive analgesic treatment in adult patients with advanced cancer who experience moderate to severe pain during the highest tolerated dose of strong opioid therapy for persistent background pain.
Epidyolex is indicated for use as adjunctive therapy of seizures associated with Lennox Gastaut syndrome (LGS) or Dravet syndrome (DS), in conjunction with clobazam, for patients 2 years of age and older.
Treatment of Rett syndrome
Treatment of seizures associated with Dravet Syndrome (DS), Treatment of seizures associated with infantile spasms (IS), Treatment of seizures associated with Lennox-Gastaut Syndrome (LGS), Treatment of seizures associated with Tuberous Sclerosis Complex (TSC)
Although the exact mechanism and magnitude of effects of THC and CBD are not fully understood, CBD has been shown to have analgesic, anticonvulsant, muscle relaxant, anxiolytic, neuroprotective, anti-oxidant, and anti-psychotic activity. This wide variety of effects is likely due to it's complex pharmacological mechanisms. In addition to binding to CB1 and CB2 receptors of the endocannabinoid system, there is evidence that CBD activates 5-HT1A serotonergic and TRPV12 vanilloid receptors, antagonizes alpha-1 adrenergic and -opioid receptors, inhibits synaptosomal uptake of noradrenaline, dopamine, serotonin and gaminobutyric acid and cellular uptake of anandamide, acts on mitochondria Ca2 stores, blocks low-voltage-activated (T-type) Ca2 channels, stimulates activity of the inhibitory glycine-receptor, and inhibits activity of fatty amide hydrolase (FAAH).
Anticonvulsants
Drugs used to prevent SEIZURES or reduce their severity. (See all compounds classified as Anticonvulsants.)
N03AX
N - Nervous system
N03 - Antiepileptics
N03A - Antiepileptics
N03AX - Other antiepileptics
N03AX24 - Cannabidiol
Absorption
Following a single buccal administration, maximum plasma concentrations of both CBD and THC typically occur within two to four hours. When administered buccally, blood levels of THC and other cannabinoids are lower compared with inhalation of smoked cannabis. The resultant concentrations in the blood are lower than those obtained by inhaling the same dose because absorption is slower, redistribution into fatty tissues is rapid and additionally some of the THC undergoes hepatic first pass metabolism to 11-OH-THC, a psycho-active metabolite. The CBD component of sublingual Sativex was found to have a Tmax of 1.63hr and a Cmax of 2.50ng/mL, while buccal Sativex was found to have a Tmax of 2.80hr and a Cmax of 3.02ng/mL.
Route of Elimination
Elimination from plasma is bi-exponential with an initial half-life of one to two hours. The terminal elimination half-lives are of the order of 24 to 36 hours or longer. Sativex is excreted in the urine and faeces.
Volume of Distribution
Cannabinoids are distributed throughout the body; they are highly lipid soluble and accumulate in fatty tissue. The release of cannabinoids from fatty tissue is responsible for the prolonged terminal elimination half-life.
THC and CBD are metabolized in the liver by a number of cytochrome P450 isoenzymes, including CYP2C9, CYP2C19, CYP2D6 and CYP3A4. They may be stored for as long as four weeks in the fatty tissues from which they are slowly released at sub-therapeutic levels back into the blood stream and metabolized via the renal and biliary systems. The main primary metabolite of CBD is 7-hydroxy-cannabidiol.
The CBD component of sublingual Sativex was found to have a half life (t1/2) of 1.44hr, while buccal Sativex was found to have a half life (t1/2) of 1.81hr.
The exact mechanism of action of CBD and THC is not currently fully understood. However, it is known that CBD acts on cannabinoid (CB) receptors of the endocannabinoid system, which are found in numerous areas of the body, including the peripheral and central nervous systems, including the brain. The endocannabinoid system regulates many physiological responses of the body including pain, memory, appetite, and mood. More specifically, CB1 receptors can be found within the pain pathways of the brain and spinal cord where they may affect CBD-induced analgesia and anxiolysis, and CB2 receptors have an effect on immune cells, where they may affect CBD-induced anti-inflammatory processes. CBD has been shown to act as a negative allosteric modulator of the cannabinoid CB1 receptor, the most abundant G-Protein Coupled Receptor (GPCR) in the body. Allosteric regulation of a receptor is achieved through the modulation of the activity of a receptor on a functionally distinct site from the agonist or antagonist binding site. The negative allosteric modulatory effects of CBD are therapeutically important as direct agonists are limited by their psychomimetic effects while direct antagonists are limited by their depressant effects.
 Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2022-01-03
Pay. Date : 2021-06-01
DMF Number : 35648
Submission : 2021-02-16
Status : Active
Type : II

Certificate Number : CEP 2024-055 - Rev 00
Issue Date : 2024-12-17
Type : Chemical
Substance Number : 3151
Status : Valid

NDC Package Code : 17359-1800
Start Marketing Date : 2019-05-24
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

 Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.
Biophore is a research-driven global pharmaceutical company focused on niche APIs for the generic industry.

GDUFA
DMF Review : Reviewed
Rev. Date : 2022-03-14
Pay. Date : 2022-02-22
DMF Number : 35992
Submission : 2021-06-03
Status : Active
Type : II

Date of Issue : 2025-02-27
Valid Till : 2028-02-26
Written Confirmation Number : WC-0511
Address of the Firm :

NDC Package Code : 73435-015
Start Marketing Date : 2021-10-05
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (10kg/10kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2019-08-20
Pay. Date : 2018-07-06
DMF Number : 32901
Submission : 2018-08-10
Status : Active
Type : II

 Malladi is a leader in Ephedrine, Pseudoephedrine Salts & Phenylephrine HCl // USFDA, EDQM, ANSM, KFDA, and TGA inspected.
Malladi is a leader in Ephedrine, Pseudoephedrine Salts & Phenylephrine HCl // USFDA, EDQM, ANSM, KFDA, and TGA inspected.
 Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.
Noramco is a partner to the pharmaceutical industry for controlled substance development & manufacturing.

GDUFA
DMF Review : Reviewed
Rev. Date : 2022-03-15
Pay. Date : 2022-03-10
DMF Number : 33223
Submission : 2019-02-22
Status : Active
Type : II

NDC Package Code : 73548-2155
Start Marketing Date : 2021-03-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.

GDUFA
DMF Review : Reviewed
Rev. Date : 2020-12-17
Pay. Date : 2020-09-29
DMF Number : 34635
Submission : 2020-09-29
Status : Active
Type : II

NDC Package Code : 68108-0881
Start Marketing Date : 2020-09-29
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 Lupin Manufacturing Solutions – delivering high-quality APIs & end-to-end CDMO services for faster, cost-effective drug development.
Lupin Manufacturing Solutions – delivering high-quality APIs & end-to-end CDMO services for faster, cost-effective drug development.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39675
Submission : 2024-04-11
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
39
PharmaCompass offers a list of Cannabidiol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cannabidiol manufacturer or Cannabidiol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cannabidiol manufacturer or Cannabidiol supplier.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol, including repackagers and relabelers. The FDA regulates 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol supplier is an individual or a company that provides 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol active pharmaceutical ingredient (API) or 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol finished formulations upon request. The 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol suppliers may include 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol API manufacturers, exporters, distributors and traders.
click here to find a list of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol DMF (Drug Master File) is a document detailing the whole manufacturing process of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol active pharmaceutical ingredient (API) in detail. Different forms of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol DMFs exist exist since differing nations have different regulations, such as 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol DMF submitted to regulatory agencies in the US is known as a USDMF. 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol USDMF includes data on 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol suppliers with USDMF on PharmaCompass.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEP of the European Pharmacopoeia monograph is often referred to as a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol Certificate of Suitability (COS). The purpose of a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol to their clients by showing that a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEP has been issued for it. The manufacturer submits a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEP (COS) as part of the market authorization procedure, and it takes on the role of a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEP holder for the record. Additionally, the data presented in the 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol DMF.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol suppliers with CEP (COS) on PharmaCompass.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol written confirmation (2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol WC) is an official document issued by a regulatory agency to a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol manufacturer, verifying that the manufacturing facility of a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol APIs or 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol finished pharmaceutical products to another nation, regulatory agencies frequently require a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol WC (written confirmation) as part of the regulatory process.
click here to find a list of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol suppliers with NDC on PharmaCompass.
2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol GMP manufacturer or 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol GMP API supplier for your needs.
A 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CoA (Certificate of Analysis) is a formal document that attests to 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol's compliance with 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol specifications and serves as a tool for batch-level quality control.
2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CoA mostly includes findings from lab analyses of a specific batch. For each 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol may be tested according to a variety of international standards, such as European Pharmacopoeia (2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol EP), 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol USP).

