
Synopsis
Synopsis
0
KDMF
0
VMF
0
Canada
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
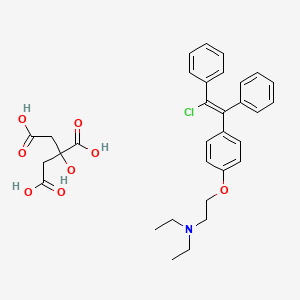

1. Chloramiphene
2. Citrate, Clomiphene
3. Clomid
4. Clomide
5. Clomifen
6. Clomifene
7. Clomiphene
8. Clomiphene Citrate
9. Clomiphene Hydrochloride
10. Clostilbegit
11. Dyneric
12. Gravosan
13. Hydrochloride, Clomiphene
14. Klostilbegit
15. Serophene
1. Clomiphene Citrate
2. 50-41-9
3. Zuclomiphene Citrate
4. Cis-clomiphene Citrate
5. Clomid
6. Zuclomid
7. Clomiphene A Citrate
8. 7619-53-6
9. Clomiphene Citrate Salt
10. Milophene
11. Serophene
12. (z)-clomiphene Citrate
13. Chloramiphene
14. Omifin
15. Rmi 16289
16. Clomivid
17. Clomphid
18. Dyneric
19. Genozym
20. Ikaclomin
21. Clomifene Citrate (serophene)
22. Zuclomiphene (citrate)
23. Uy5x264qzv
24. Mrl 41
25. Nsc-151466
26. 2-(4-(2-chloro-1,2-diphenylvinyl)phenoxy)-n,n-diethylethanamine 2-hydroxypropane-1,2,3-tricarboxylate
27. Nsc 35770
28. Rmi 16312
29. 2-[4-[(z)-2-chloro-1,2-diphenylethenyl]phenoxy]-n,n-diethylethanamine;2-hydroxypropane-1,2,3-tricarboxylic Acid
30. Ethanamine, 2-(4-((1z)-2-chloro-1,2-diphenylethenyl)phenoxy)-n,n-diethyl-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1)
31. Ethanamine, 2-[4-(2-chloro-1,2-diphenylethenyl)phenoxy]-n,n-diethyl-,2-hydroxy-1,2,3-propanetricarboxylate (1:1)
32. Mer-41
33. Mrl-41
34. Unii-uy5x264qzv
35. Nsc 151466
36. Nsc-35770
37. Cas-50-41-9
38. Clomiphene Citrate, Cis-
39. Triethylamine, 2-(p-(2-chloro-1,2-diphenylvinyl)phenoxy)-, Citrate (1:1), (z)-
40. Mls001332629
41. Mls002207099
42. Schembl223472
43. Spectrum1500196
44. Sgcut00109
45. Chembl1213137
46. Hms501h04
47. Hy-b1617a
48. Hms1920o19
49. Hms2091g06
50. Hms2232d12
51. Pharmakon1600-01500196
52. Amy33425
53. Bcp03057
54. To_000032
55. Ccg-39553
56. Nsc151466
57. Nsc756698
58. S2561
59. Akos005111131
60. Bcp9000541
61. Ds-6814
62. Fd12066
63. Nsc-756698
64. Ncgc00017008-01
65. Ncgc00017008-02
66. Ncgc00017008-03
67. Ncgc00094626-01
68. Ncgc00094626-02
69. Ncgc00164385-01
70. Ac-19685
71. Ac-32011
72. Ethanamine, 2-(4-(2-chloro-1,2-diphenylethenyl)phenoxy)-n,n-diethyl-, (z)-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1)
73. Bcp0726000214
74. Cs-0111663
75. Sw196898-3
76. C08161
77. Triethylamine,2-diphenylvinyl)phenoxy]-, Citrate
78. Sr-05000001591
79. Sr-05000001591-1
80. Q27291328
81. Triethylamine,2-diphenylvinyl)phenoxy]-, Citrate (1:1)
82. Triethylamine,2-diphenylvinyl)phenoxy]-, Citrate (1:1), (z)-
83. (2)2-[p-(2-chloro-1,2-diphenylvinyl)phenoxy]triethylamine Citrate (1:1)
84. 2-(4-[2-chloro-1,2-diphenylethenyl]phenoxy)-n,n-diethylethanamine Citrate Salt
85. {2-[4-(2-chloro-1,2-diphenylvinyl)phenoxy]ethyl}diethylamine 2-hydroxypropane-1,2,3-tricarboxylic Acid Salt
86. 2-({4-[(z)-2-chloro-1,2-diphenylethenyl]phenyl}oxy)-n,n-diethylethanamine 2-hydroxypropane-1,2,3-tricarboxylate (salt)
87. Ethanamine,2-diphenylethenyl)phenoxy]-n,n-diethyl-, (z)-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1)
88. Ethanamine,2-diphenylethenyl)phenoxy]-n,n-diethyl-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1)
| Molecular Weight | 598.1 g/mol |
|---|---|
| Molecular Formula | C32H36ClNO8 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 14 |
| Exact Mass | 597.2129448 g/mol |
| Monoisotopic Mass | 597.2129448 g/mol |
| Topological Polar Surface Area | 145 Ų |
| Heavy Atom Count | 42 |
| Formal Charge | 0 |
| Complexity | 708 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 6 | |
|---|---|
| Drug Name | Clomid |
| PubMed Health | Clomiphene (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Female Reproductive Agent |
| Drug Label | Clomiphene citrate tablets, USP is an orally administered, nonsteroidal, ovulatory stimulant designated chemically as 2-[p-(2chloro-1,2-diphenylvinyl)phenoxy] triethylamine citrate (1:1). It has the molecular formula of C26H28CINO C6H8O7 and a mo... |
| Active Ingredient | Clomiphene citrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Sanofi Aventis Us |
| 2 of 6 | |
|---|---|
| Drug Name | Clomiphene citrate |
| PubMed Health | Clomiphene (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Female Reproductive Agent |
| Drug Label | ClomiPHENE citrate is an orally administered, nonsteroidal, ovulatory stimulant designated chemically as 2-[p-(2-chloro-1,2-diphenylvinyl) phenoxy] triethylamine citrate (1:1). It has a molecular formula of C26H28CINO C6H8O7 and a molecular weig... |
| Active Ingredient | Clomiphene citrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Par Pharm |
| 3 of 6 | |
|---|---|
| Drug Name | Serophene |
| Active Ingredient | Clomiphene citrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Emd Serono |
| 4 of 6 | |
|---|---|
| Drug Name | Clomid |
| PubMed Health | Clomiphene (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Female Reproductive Agent |
| Drug Label | Clomiphene citrate tablets, USP is an orally administered, nonsteroidal, ovulatory stimulant designated chemically as 2-[p-(2chloro-1,2-diphenylvinyl)phenoxy] triethylamine citrate (1:1). It has the molecular formula of C26H28CINO C6H8O7 and a mo... |
| Active Ingredient | Clomiphene citrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Sanofi Aventis Us |
| 5 of 6 | |
|---|---|
| Drug Name | Clomiphene citrate |
| PubMed Health | Clomiphene (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Female Reproductive Agent |
| Drug Label | ClomiPHENE citrate is an orally administered, nonsteroidal, ovulatory stimulant designated chemically as 2-[p-(2-chloro-1,2-diphenylvinyl) phenoxy] triethylamine citrate (1:1). It has a molecular formula of C26H28CINO C6H8O7 and a molecular weig... |
| Active Ingredient | Clomiphene citrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Par Pharm |
| 6 of 6 | |
|---|---|
| Drug Name | Serophene |
| Active Ingredient | Clomiphene citrate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 50mg |
| Market Status | Prescription |
| Company | Emd Serono |
Fertility Agents, Female
Compounds which increase the capacity to conceive in females. (See all compounds classified as Fertility Agents, Female.)
Estrogen Antagonists
Compounds which inhibit or antagonize the action or biosynthesis of estrogenic compounds. (See all compounds classified as Estrogen Antagonists.)
Selective Estrogen Receptor Modulators
A structurally diverse group of compounds distinguished from ESTROGENS by their ability to bind and activate ESTROGEN RECEPTORS but act as either an agonist or antagonist depending on the tissue type and hormonal milieu. They are classified as either first generation because they demonstrate estrogen agonist properties in the ENDOMETRIUM or second generation based on their patterns of tissue specificity. (Horm Res 1997;48:155-63) (See all compounds classified as Selective Estrogen Receptor Modulators.)
 BioXera Pharma: A trusted partner for high-quality human and veterinary APIs and intermediates across global regulated markets.
BioXera Pharma: A trusted partner for high-quality human and veterinary APIs and intermediates across global regulated markets.

Date of Issue : 2025-12-02
Valid Till : 2028-12-01
Written Confirmation Number : WC-0248
Address of the Firm :

NDC Package Code : 49169-1050
Start Marketing Date : 2025-10-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.

GDUFA
DMF Review : Reviewed
Rev. Date : 2023-11-21
Pay. Date : 2023-11-17
DMF Number : 28947
Submission : 2015-01-20
Status : Active
Type : II

Certificate Number : R1-CEP 2003-251 - Rev 06
Issue Date : 2022-02-09
Type : Chemical
Substance Number : 997
Status : Valid

| Available Reg Filing : ASMF |

 Expand your portfolio with Egis- value-added generics, API capabilities, and reliable contract services to drive your growth.
Expand your portfolio with Egis- value-added generics, API capabilities, and reliable contract services to drive your growth.
 Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
 Willow Birch Pharma delivers trusted, high-quality APIs nationwide with unmatched service, compliance, and competitive value.
Willow Birch Pharma delivers trusted, high-quality APIs nationwide with unmatched service, compliance, and competitive value.

NDC Package Code : 10695-307
Start Marketing Date : 2025-05-07
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (35kg/35kg)
Marketing Category : BULK INGREDIENT FOR HUMAN PRESCRIPTION COMPOUNDING
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
About the Company : HRV Pharma is a global manufacturer, seller, and exporter of APIs, intermediates, pellets, food-grade chemicals, food additives, and food ingredients. The company provides sourcing...
 BioXera Pharma: A trusted partner for high-quality human and veterinary APIs and intermediates across global regulated markets.
BioXera Pharma: A trusted partner for high-quality human and veterinary APIs and intermediates across global regulated markets.
About the Company : BioXera Pharma is a quality-focused manufacturer of APIs, veterinary APIs, and intermediates, with expertise in innovative process development for domestic and regulated internatio...
 Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
About the Company : Founded in 2003, Seqens has evolved into a global leader in pharmaceutical solutions and specialty ingredients. The company supports customers in the development, scale-up, and man...
 Expand your portfolio with Egis- value-added generics, API capabilities, and reliable contract services to drive your growth.
Expand your portfolio with Egis- value-added generics, API capabilities, and reliable contract services to drive your growth.
About the Company : Egis is an EU-based, vertically integrated generic pharmaceutical company with over 110 years of experience across the entire value chain. Renowned for reliable supply, proprietary...
About the Company : Jai Radhe Sales, founded in 1999, is a global distributor specializing in high-quality pharmaceutical ingredients from India. It offers complete sourcing solutions, technical and r...
 Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
Octavius Pharma has been empowering lives since 1980 by providing quality products like DC granules, APIs and FDFs.
About the Company : Octavius Pharma is a global leader in Directly Compressible Granules with over 45 years of experience in formulation development, manufacturing, and commercialization. Its portfoli...
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
About the Company : Tenatra International was established as a proprietorship firm in 1999. It got off to a very good start, supporting clients in the United States, Mexico and Europe. As business opp...
About the Company : Maithri Drugs Pvt. Ltd. is a global supplier of Active Pharmaceutical Ingredients (APIs), serving pharmaceutical companies in 60+ countries. Its API portfolio spans antivirals, ant...
 Willow Birch Pharma delivers trusted, high-quality APIs nationwide with unmatched service, compliance, and competitive value.
Willow Birch Pharma delivers trusted, high-quality APIs nationwide with unmatched service, compliance, and competitive value.
About the Company : Willow Birch Pharma, Inc. is a premier supplier of bulk APIs to North American Compounding Pharmacies, sourcing from FDA registered and GMP manufacturers. With licenses in all 50 s...
About the Company : Public health is always an imperative that implies immense responsibility, not a mere re-adjustable option. As a company operating under world’s second largest industry of Pharma...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Androxal (Enclomiphene Citrate) is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Hypogonadism.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Endocrinology Brand Name: Androxal
Study Phase: Phase IProduct Type: Hormone
Sponsor: Celerion
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable October 24, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Endocrinology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Celerion
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Androxal (Enclomiphene Citrate) is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Hypogonadism.
Product Name : Androxal
Product Type : Hormone
Upfront Cash : Inapplicable
October 24, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Hormone
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable June 23, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
An Open-Label Study to Evaluate the Effect of Age Upon the Pharmacokinetics of Androxal
Details : Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Hormone
Upfront Cash : Inapplicable
June 23, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Androxal is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 23, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Study to Evaluate the Effect of Food Upon the Pharmacokinetics of Androxal
Details : Androxal is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
May 23, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Androxal is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 21, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
A Thorough QT/QTc Study to Assess the Effects of Androxal in Healthy Male Subjects
Details : Androxal is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 21, 2014
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Hypogonadism.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Endocrinology Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Hormone
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 25, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Endocrinology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Hypogonadism.
Product Name : Undisclosed
Product Type : Hormone
Upfront Cash : Inapplicable
November 25, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Androxal (Enclomiphene Citrate) is a Hormone drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Hypogonadism.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Endocrinology Brand Name: Androxal
Study Phase: Phase IIIProduct Type: Hormone
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 25, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Endocrinology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Androxal (Enclomiphene Citrate) is a Hormone drug candidate, which is currently being evaluated in phase III clinical studies for the treatment of Hypogonadism.
Product Name : Androxal
Product Type : Hormone
Upfront Cash : Inapplicable
November 25, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Hormone
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 25, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Assessment of Drug-Drug Interactions of Androxal With Cytochrome P450 Isoenyzmes in Healthy Males
Details : Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Hormone
Upfront Cash : Inapplicable
November 25, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Androxal is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable November 14, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Comparison of Two Formulations of Androxal
Details : Androxal is a Other Small Molecule drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
November 14, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Undisclosed Brand Name: Undisclosed
Study Phase: Phase IProduct Type: Hormone
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable October 10, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Undisclosed
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
A Dose Escalating Study to Determine the Tolerability and PK of a Single Dose of Androxal
Details : Enclomiphene Citrate is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of undefined medical condition.
Product Name : Undisclosed
Product Type : Hormone
Upfront Cash : Inapplicable
October 10, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Androxal (Enclomiphene Citrate) is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Hypogonadism.
Lead Product(s): Clomiphene Citrate,Inapplicable
Therapeutic Area: Endocrinology Brand Name: Androxal
Study Phase: Phase IProduct Type: Hormone
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable August 16, 2013
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Clomiphene Citrate,Inapplicable
Therapeutic Area : Endocrinology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Androxal (Enclomiphene Citrate) is a Hormone drug candidate, which is currently being evaluated in phase I clinical studies for the treatment of Hypogonadism.
Product Name : Androxal
Product Type : Hormone
Upfront Cash : Inapplicable
August 16, 2013
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
73
PharmaCompass offers a list of Clomiphene Citrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Clomiphene Citrate manufacturer or Clomiphene Citrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Clomiphene Citrate manufacturer or Clomiphene Citrate supplier.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate, including repackagers and relabelers. The FDA regulates 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate supplier is an individual or a company that provides 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate active pharmaceutical ingredient (API) or 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate finished formulations upon request. The 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate suppliers may include 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate API manufacturers, exporters, distributors and traders.
click here to find a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate DMF (Drug Master File) is a document detailing the whole manufacturing process of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate active pharmaceutical ingredient (API) in detail. Different forms of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate DMFs exist exist since differing nations have different regulations, such as 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate DMF submitted to regulatory agencies in the US is known as a USDMF. 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate USDMF includes data on 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate Drug Master File in Japan (1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate JDMF) empowers 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate JDMF during the approval evaluation for pharmaceutical products. At the time of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate suppliers with JDMF on PharmaCompass.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEP of the European Pharmacopoeia monograph is often referred to as a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate Certificate of Suitability (COS). The purpose of a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate to their clients by showing that a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEP has been issued for it. The manufacturer submits a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEP (COS) as part of the market authorization procedure, and it takes on the role of a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEP holder for the record. Additionally, the data presented in the 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate DMF.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate suppliers with CEP (COS) on PharmaCompass.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate written confirmation (1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate WC) is an official document issued by a regulatory agency to a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate manufacturer, verifying that the manufacturing facility of a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate APIs or 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate finished pharmaceutical products to another nation, regulatory agencies frequently require a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate WC (written confirmation) as part of the regulatory process.
click here to find a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate suppliers with NDC on PharmaCompass.
1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate GMP manufacturer or 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate GMP API supplier for your needs.
A 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CoA (Certificate of Analysis) is a formal document that attests to 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate's compliance with 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate specifications and serves as a tool for batch-level quality control.
1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CoA mostly includes findings from lab analyses of a specific batch. For each 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate may be tested according to a variety of international standards, such as European Pharmacopoeia (1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate EP), 1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (1-(p-(beta-Diethylaminoethoxy)phenyl)-1,2-diphenyl-2-chloroethylene citrate USP).

