
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Listed Suppliers
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
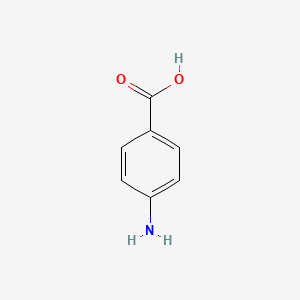

1. 4 Aminobenzoic Acid
2. 4 Aminobenzoic Acid, Potassium Salt
3. 4-aminobenzoate, Potassium
4. 4-aminobenzoic Acid, Potassium Salt
5. Aminobenzoate, Potassium
6. Aminobenzoic Acid (usp)
7. Epit Vit
8. Epitelplast
9. Hachemina
10. Magnesium Para-aminobenzoate
11. P Aminobenzoic Acid
12. P-aminobenzoic Acid
13. Paba
14. Pabasan
15. Para Aminobenzoic Acid
16. Para-aminobenzoate, Magnesium
17. Para-aminobenzoic Acid
18. Paraminan
19. Paraminol
20. Potaba
21. Potassium 4 Aminobenzoate
22. Potassium 4-aminobenzoate
23. Potassium Aminobenzoate
1. 150-13-0
2. P-aminobenzoic Acid
3. Paba
4. Para-aminobenzoic Acid
5. Vitamin Bx
6. Aminobenzoic Acid
7. P-carboxyaniline
8. 4-carboxyaniline
9. Sunbrella
10. P-carboxyphenylamine
11. Hachemina
12. Paraminol
13. Benzoic Acid, 4-amino-
14. 1-amino-4-carboxybenzene
15. Pabacyd
16. Pabafilm
17. Pabamine
18. Paranate
19. Amben
20. Potaba
21. Romavit
22. Bacterial Vitamin H1
23. Vitamin H'
24. Anticanitic Vitamin
25. Chromotrichia Factor
26. Rvpaba Lipstick
27. Papacidum
28. Rvpaba
29. Trichochromogenic Factor
30. Anti-chromotrichia Factor
31. Super Shade By Coppertone
32. Benzoic Acid, P-amino-
33. Acidum Paraminobenzoicum
34. Benzoic Acid, 4-amino
35. Vitamin H1
36. Abee
37. Pabagel
38. Pabanol
39. 4-amino-benzoic Acid
40. Caswell No. 033b
41. Kyselina P-aminobenzoova
42. Mfcd00007894
43. P-aminobenzoesaeure
44. 4-aminobenzoesaeure
45. 4-carboxyphenylamine
46. Acido P-aminobenzoico
47. Pab
48. Gamma-aminobenzoic Acid
49. Aminobenzoate
50. Acido P-aminobenzoico [italian]
51. Kyselina P-aminobenzoova [czech]
52. Para-aminobenzoate
53. Anticantic Vitamin
54. 4-aminobenzoic-acid
55. Epa Pesticide Chemical Code 233300
56. Brn 0471605
57. P-amino Benzoic Acid
58. P-amino-benzoic Acid
59. Chebi:30753
60. Ai3-02436
61. Tl2tje8qtx
62. Nsc-7627
63. Aminobenzoic Acid (usp)
64. Aminobenzoic Acid [usp]
65. .gamma.-aminobenzoic Acid
66. Aniline-4-carboxylic Acid
67. Chembl542
68. Aminobenzoic Acid, Para
69. Benzoic Acid, 4-amino-, Homopolymer
70. Ncgc00091051-01
71. Dsstox_cid_4466
72. Dsstox_rid_77411
73. Dsstox_gsid_24466
74. 4-azaniumylbenzoate
75. 25136-77-0
76. Cas-150-13-0
77. Smr000471833
78. Ccris 6209
79. Hsdb 6840
80. Unii-tl2tje8qtx
81. Nsc 7627
82. Einecs 205-753-0
83. Actipol
84. P-carboxy Aniline
85. Amino Benzoic Acid
86. P-aminobezoic Acid
87. Gamma-aminobenzoate
88. 4-aminobezoic Acid
89. Trochromogenic Factor
90. Aniline-4-carboxylate
91. 4-amino Benzoic Acid
92. 4-(amino)benzoic Acid
93. Antichromotrichia Factor
94. Spectrum_000036
95. Rvpaba Lipstick (tn)
96. P-amino-benzoate
97. Spectrum2_000123
98. Spectrum3_000294
99. Spectrum4_000142
100. Spectrum5_000778
101. Paba [inci]
102. Tetracaine Ep Impurity A
103. P-aminobenzoic Acid,(s)
104. Bmse000066
105. Bmse000887
106. Bmse000916
107. Epitope Id:115017
108. Ec 205-753-0
109. Schembl8249
110. 4-aminobenzoic Acid, 99%
111. Oprea1_221096
112. Bspbio_001828
113. Kbiogr_000584
114. Kbioss_000396
115. Zinc920
116. Mls001066325
117. Mls001335919
118. Mls001335920
119. Para Amino Benzoic Acid Usp
120. Bidd:er0375
121. Divk1c_000783
122. Spbio_000166
123. 4-aminobenzoic Acid-[13c6]
124. P-aminobenzoic Acid, Free Acid
125. Component Of Presun (salt/mix)
126. Dtxsid6024466
127. Schembl13232108
128. Kbio1_000783
129. Kbio2_000396
130. Kbio2_002964
131. Kbio2_005532
132. Kbio3_001328
133. Nsc7627
134. P-aminobenzoic Acid [mi]
135. Aminobenzoic Acid [mart.]
136. Ninds_000783
137. P-aminobenzoic Acid 100g
138. Hms2269e10
139. Hms3870e13
140. Aminobenzoic Acid [usp-rs]
141. Aminobenzoic Acid [who-dd]
142. 4-aminobenzoic Acid [hsdb]
143. Act09225
144. Hy-b1008
145. Tox21_111069
146. Tox21_201702
147. Tox21_300087
148. Bdbm50145829
149. S4510
150. Stk397441
151. Para-aminobenzoic Acid [iarc]
152. Akos000118983
153. Para-aminobenzoic Acid [vandf]
154. Tox21_111069_1
155. Ac-8107
156. Am86626
157. Ccg-266139
158. Cs-4505
159. Db02362
160. Idi1_000783
161. 4-aminobenzoic Acid, Analytical Standard
162. Aminobenzoic Acid [usp Monograph]
163. Ncgc00091051-02
164. Ncgc00091051-03
165. Ncgc00091051-04
166. Ncgc00253923-01
167. Ncgc00259251-01
168. As-12493
169. Nci60_041683
170. Sy003749
171. 4-aminobenzoic Acid [ep Impurity]
172. Sbi-0051277.p003
173. 4-aminobenzoic Acid [ep Monograph]
174. Db-028695
175. 4-aminobenzoic Acid, Reagentplus(r), 99%
176. A0269
177. Ft-0617557
178. 4-aminobenzoic Acid, >=98.0% (hplc/nt)
179. 4-aminobenzoic Acid, Reagentplus(r), >=99%
180. C00568
181. D02456
182. A809010
183. Q284959
184. 4-aminobenzoic Acid, Vetec(tm) Reagent Grade, 98%
185. Q-200432
186. 4-aminobenzoic Acid, Purified By Sublimation, >=99%
187. Z57127446
188. 4-aminobenzoic Acid, 98.5-100.2%, Saj First Grade
189. 4-aminobenzoic Acid, Saj Special Grade, 99.5-100.2%
190. F2191-0259
191. Tetracaine Hydrochloride Impurity A [ep Impurity]
192. 4a5e7dd8-8a22-4642-86bf-05b778c0c78e
193. Procaine Benzylpenicillin Impurity A [ep Impurity]
194. 4-aminobenzoic Acid, European Pharmacopoeia (ep) Reference Standard
195. Aminobenzoic Acid, United States Pharmacopeia (usp) Reference Standard
196. Aminobenzoic Acid, Pharmaceutical Secondary Standard; Certified Reference Material
| Molecular Weight | 137.14 g/mol |
|---|---|
| Molecular Formula | C7H7NO2 |
| XLogP3 | 0.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Exact Mass | 137.047678466 g/mol |
| Monoisotopic Mass | 137.047678466 g/mol |
| Topological Polar Surface Area | 63.3 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 128 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Sunscreening Agent
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Used orally in the treatment of conditions such as scleroderma, dermatomyositis, and Peyronie's disease, and topically as a sunscreen and protectant.
Hussar, D.A. (ed.). Modell's Drugs in Current Use and New Drugs. 38th ed. New York, NY: Springer Publishing Co., 1992., p. 91
Daily use of a sunscreen with a high SPF (greater than 15) on usually exposed skin is recommended for residents of areas of high ... /solar radiation/ who work outdoors or ... /enjoy/ regular outdoor recreation. Daily use of a sunscreen can reduce the cumulative ... /solar/ exposure that causes actinic keratoses and squamous-cell carcinoma.
IARC Working Group on the Evaluation of Cancer-Preventive Agents (2001) Sunscreens (IARC Handbooks of Cancer Prevention, Vol. 5), Lyon, IARC; Unit of Chemoprevention: Cancer-Preventive Effects of Sunscreens.
PABA has long been an accepted objective marker to verify completeness of 24 hour urine sampling as PABA is rapidly and almost completely eliminated with the urine. For this reason PABA has been used clinically for long as the indicator substance in pancreas and liver function tests.
European Commission Scientific Committeee on Consumer Products; Opinion on 4-Aminobenzoic acid (PABA), Available from, as of October 18, 2013: https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_058.pdf
Sunscreen preparations should be applied uniformly and generously to all exposed skin surfaces, including lips, before exposure to UVB radiation. Two applications of the sunscreen may be needed for maximum protection. PABA-containing sunscreens are most effective when applied 1-2 hours before exposure to sunlight. Sunscreen products that are not water resistant should be reapplied after swimming, towel-drying, or profuse sweating and, because most sunscreens are easily removed from the skin, reapplication every 1-2 hours or according to the manufacturer's directions usually is required to provide adequate protection from UVB light. /Sunscreens/
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013
PABA has been shown in vitro to displace methotrexate from plasma protein binding, thus increasing the free methotrexate concentrations.
Hansten P.D. Drug Interactions. 5th ed. Philadelphia: Lea and Febiger, 1985., p. 266
PABA derivatives reportedly have weak sensitization potential, but the incidence of allergic and photoallergic contact dermatitis associated with their use is increasing. Contamination of PABA derivatives with benzocaine which may cause allergic reactions has been reported. In patients allergic to compounds that are structurally similar to PABA (e.g., ester-type anesthetics, aniline dyes, thiazides, sulfonylurea and paraphenylenediamine drugs), cross-sensitivity to PABA derivatives has been reported occasionally; therefore, sunscreens containing PABA derivatives may be contraindicated in patients with a history of hypersensitivity to these chemicals.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013
The manufacturers of sunscreen preparations with propellants warn that concentrating and subsequently inhaling the fumes from these preparations may be harmful or fatal. /Propellants/
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013
Because the absorptive characteristics of skin of children younger than 6 months of age may differ from those of adults and because the immaturity of metabolic and excretory pathways of these children may limit their ability to eliminate any percutaneously absorbed sunscreen agent, sunscreen products should be used in children younger than 6 months of age only as directed by a clinician. It is possible that the characteristics of geriatric skin also differ from those of skin in younger adults, but these characteristics and the need for special considerations regarding use of sunscreen preparations in this age group are poorly understood. /Sunscreens/
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013
For more Drug Warnings (Complete) data for 4-AMINOBENZOIC ACID (13 total), please visit the HSDB record page.
D - Dermatologicals
D02 - Emollients and protectives
D02B - Protectives against uv-radiation
D02BA - Protectives against uv-radiation for topical use
D02BA01 - Aminobenzoic acid
The toxicokinetics of PABA is characterized by fast oral absorption, biotransformation by the major routes acetylation and glycine conjugation, the minor route by glucuronidation in the liver and kidney, and a fast and almost complete elimination via the urine within 24 hrs. PABA is extensively acetylated during percutaneous absorption in humans. Studies have shown that PABA can cross the placenta rapidly. Furthermore, the results of one study indicate that the human placenta has a significant capacity for acetylation of PABA.
European Commission Scientific Committeee on Consumer Products; Opinion on 4-Aminobenzoic acid (PABA), Available from, as of October 18, 2013: https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_058.pdf
Of an oral dose of 1 g para-aminobenzoic acid, 82% was excreted in the urine of 3 male volunteers within 4 hr; para-aminohippuric acid and acetyl-para- aminohippuric acid were the principal metabolites. Concurrent administration of sodium benzoate totally abolished the excretion of these glycine conjugates.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V16 256 (1978)
The percutaneous absorption and metabolism of three structurally related compounds, benzoic acid, p-aminobenzoic acid (PABA), and ethyl aminobenzoate (benzocaine), were determined in vitro through hairless guinea pig skin. Benzocaine was also studied in human skin. Absorption of benzocaine was rapid and similar through both viable and nonviable skin. The absorption of the two acidic compounds, benzoic acid and PABA, was greater through nonviable skin. A small portion (6.9%) of absorbed benzoic acid was conjugated with glycine to form hippuric acid. Although N-acetyl-benzocaine had not been observed as a metabolite of benzocaine when studied by other routes of administration, both PABA and benzocaine were extensively N-acetylated during percutaneous absorption. Thus, the metabolism of these compounds should be considered in an accurate assessment of absorption after topical application.
PMID:2293213 Nathan D et al; Pharm Res 7(11): 1147-51(1990)
Skin absorption of PABA corresponding to 1.6 to 9.6% of the applied amount of PABA was measured in the urine of six male volunteers after application of PABA in three different preparations. No significant difference where observed between the three preparations.
European Commission Scientific Committeee on Consumer Products; Opinion on 4-Aminobenzoic acid (PABA), Available from, as of October 18, 2013: https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_058.pdf
For more Absorption, Distribution and Excretion (Complete) data for 4-AMINOBENZOIC ACID (10 total), please visit the HSDB record page.
The two major metabolic pathways in a variety of species (guinea-pigs, rabbits and rats, but not dogs) are acetylation of the amino group and conjugation of the carboxy group, either with glycine or with glucuronic acid. Acetylation occurs in liver, heart, lung, blood and kidneys of rats and in the mucous membranes of the gastrointestinal tract of cattle. N-Acetyl-transferase activity in the presence of para-aminobenzoic acid is similar in liver and lung tissue of rabbits. Acetylation not only of para-aminobenzoic acid (30-40%) but also of para-aminohippuric acid (70%) takes place in the kidney of rabbits; acetylation of para-aminohippurate also occurs in the kidney of guinea-pigs.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V16 255 (1978)
Acetylation is dose-dependent. In rats given up to 5 mg/kg bw, 75% of the metabolites were acetylated; with higher doses, the extent of acetylation decreased down to 40%. An inverse relationship exists between acetylation and glycine conjugation: when acetylation decreases, glycine conjugation increases; such a decrease is seen in pantothenic acid-deficient rats. . Male rats excreted a larger amount of acetylated conjugates in the urine than females.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V16 255 (1978)
Following ingestion /in man/ of 1.0 g, ...main metabolites were p-aminohippuric acid and acetyl-p-aminohippuric acid.
Sheftel, V.O.; Indirect Food Additives and Polymers. Migration and Toxicology. Lewis Publishers, Boca Raton, FL. 2000., p. 823
... PABA is predominantly metabolized by acetylation and glycine conjugation to form p-acetamidobenzoic acid (PAABA), p-aminohippuric acid (PAHA), and p-acetamidohippuric acid (PAAHA). ... The half-lives of PABA were 7.01 +/- 0.32 min in rapid acetylation rabbits and 7.08 +/- 0.78 min in slow acetylation rabbits. Significant differences were obtained in formation of PAABA and PAHA formed from PABA in both acetylation phenotype rabbits.
PMID:10594871 Song DJ et al; Biopharm Drug Dispos 20(5): 263-270 (1999)
For more Metabolism/Metabolites (Complete) data for 4-AMINOBENZOIC ACID (9 total), please visit the HSDB record page.
... The half-lives of PABA were 7.01 +/- 0.32 min in rapid acetylation rabbits and 7.08+/-0.78 min in slow acetylation rabbits.
PMID:10594871 Song DJ et al; Biopharm Drug Dispos 20(5): 263-270 (1999)
Radiation is absorbed by chemical sunscreens when the electron energy level of the drug is raised from its ground state to a higher energy level or excited state. Chromophore groups (C=C, C=O, O-N=O) with loosely held electrons are easily excited by radiation. Compounds which have several chromophore groups in optimal positions have high absorbance over a broad range of wavelengths. Chemical sunscreens are usually agents that absorb not less than 85% of UVB radiation (thus preventing burning) but may permit transmission of UVA radiation (thus allowing tanning). Some sunscreens may absorb wavelengths over a range that is slightly wider or narrower than that of UVB. All PABA derivatives absorb wavelengths of approximately 290-320 nm, benzophenone derivatives absorb wavelengths of approximately 250-360 nm, cinnamic acid derivatives absorb wavelengths of 280-320 nm, and salicylate derivatives and other miscellaneous chemical sunscreens absorb wavelengths of about 270-320 nm.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013
The wavelength to which the skin is maximally sensitive had been accepted for many years to be 296.7 nm; however, recent evidence suggests that the most erythemogenic UVB wavelength may be slightly lower (e.g., somewhere in the range of 292-295 nm). In addition, of the stronger burning wavelengths that reach the earth's surface, most are approximately 310 nm. Therefore, sunscreens that maximally absorb UVB radiation near either of these wavelengths are particularly effective at preventing sunburn. Maximum absorbance occurs at about 290 nm for PABA, at about 295 nm for glyceryl-p-aminobenzoate, and at about 310 nm for the remaining PABA derivatives. Maximum absorbance occurs at 280-290 nm for benzophenone derivatives, at 310 nm for cinnamic acid derivatives with the exception of diethanolamine-p-methoxycinnamate which has its maximum absorbance at 290 nm, and at 300-305 nm for salicylate derivatives and other miscellaneous sunscreens.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013
Diminish the penetration of ultraviolet (UV) light through the epidermis by absorbing UV radiation within a specific wavelength range. The amount and wavelength of UV radiation absorbed are affected by the molecular structure of the sunscreen agent. /Sunscreen agents, topical/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
Diminish the penetration of ultraviolet (UV) light through the epidermis by absorbing UV radiation within a specific wavelength range. The amount and wavelength of UV radiation absorbed are affected by the molecular structure of the sunscreen agent. /Sunscreen agents, topical/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
29
PharmaCompass offers a list of 4-Aminobenzoic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right 4-Aminobenzoic Acid manufacturer or 4-Aminobenzoic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred 4-Aminobenzoic Acid manufacturer or 4-Aminobenzoic Acid supplier.
PharmaCompass also assists you with knowing the 4-Aminobenzoic Acid API Price utilized in the formulation of products. 4-Aminobenzoic Acid API Price is not always fixed or binding as the 4-Aminobenzoic Acid Price is obtained through a variety of data sources. The 4-Aminobenzoic Acid Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A 06940_FLUKA manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of 06940_FLUKA, including repackagers and relabelers. The FDA regulates 06940_FLUKA manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. 06940_FLUKA API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A 06940_FLUKA supplier is an individual or a company that provides 06940_FLUKA active pharmaceutical ingredient (API) or 06940_FLUKA finished formulations upon request. The 06940_FLUKA suppliers may include 06940_FLUKA API manufacturers, exporters, distributors and traders.
click here to find a list of 06940_FLUKA suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing 06940_FLUKA as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for 06940_FLUKA API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture 06940_FLUKA as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain 06940_FLUKA and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a 06940_FLUKA NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of 06940_FLUKA suppliers with NDC on PharmaCompass.
06940_FLUKA Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of 06940_FLUKA GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right 06940_FLUKA GMP manufacturer or 06940_FLUKA GMP API supplier for your needs.
A 06940_FLUKA CoA (Certificate of Analysis) is a formal document that attests to 06940_FLUKA's compliance with 06940_FLUKA specifications and serves as a tool for batch-level quality control.
06940_FLUKA CoA mostly includes findings from lab analyses of a specific batch. For each 06940_FLUKA CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
06940_FLUKA may be tested according to a variety of international standards, such as European Pharmacopoeia (06940_FLUKA EP), 06940_FLUKA JP (Japanese Pharmacopeia) and the US Pharmacopoeia (06940_FLUKA USP).

