
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
South Africa
0
Listed Dossiers
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. 9350, Gs
2. Gs 9350
3. Gs-9350
4. Gs9350
5. Tybost
1. 1004316-88-4
2. Cobicistat (gs-9350)
3. Gs-9350
4. Tybost
5. Gs 9350
6. Cobicistat,gs-9350
7. Unii-lw2e03m5pg
8. Chebi:72291
9. Lw2e03m5pg
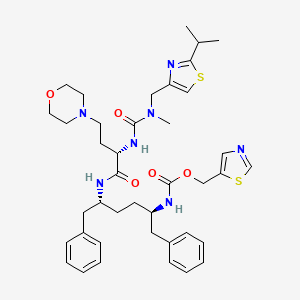
10. 1,3-thiazol-5-ylmethyl [(2r,5r)-5-{[(2s)-2-({[(2-isopropyl-1,3-thiazol-4-yl)methyl](methyl)carbamoyl}amino)-4-(morpholin-4-yl)butanoyl]amino}-1,6-diphenylhexan-2-yl]carbamate
11. 1,3-thiazol-5-ylmethyl N-[(2r,5r)-5-[[(2s)-2-[[methyl-[(2-propan-2-yl-1,3-thiazol-4-yl)methyl]carbamoyl]amino]-4-morpholin-4-ylbutanoyl]amino]-1,6-diphenylhexan-2-yl]carbamate
12. Thiazol-5-ylmethyl ((2r,5r)-5-((s)-2-(3-((2-isopropylthiazol-4-yl)methyl)-3-methylureido)-4-morpholinobutanamido)-1,6-diphenylhexan-2-yl)carbamate
13. 1,3-thiazol-5-ylmethyl N-[(2r,5r)-5-[(2s)-2-{[methyl({[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl})carbamoyl]amino}-4-(morpholin-4-yl)butanamido]-1,6-diphenylhexan-2-yl]carbamate
14. 2,7,10,12-tetraazatridecanoic Acid, 12-methyl-13-(2-(1-methylethyl)-4-thiazolyl)-9-(2-(4-morpholinyl)ethyl)-8,11-dioxo-3,6-bis(phenylmethyl)-, 5-thiazolylmethyl Ester, (3r,6r,9s)-
15. Cobicistat [usan]
16. Cobicistat [usan:inn]
17. Cobicistatum
18. 2,7,10,12-tetraazatridecanoic Acid, 12-methyl-13-[2-(1-methylethyl)-4-thiazolyl]-9-[2-(4-morpholinyl)ethyl]-8,11-dioxo-3,6-bis(phenylmethyl)-, 5-thiazolylmethyl Ester, (3r,6r,9s)-
19. Thiazol-5-ylmethyl (2r,5r)-5-((s)-2-(3-((2-isopropylthiazol-4-yl)methyl)-3-methylureido)-4-morpholinobutanamido)-1,6-diphenylhexan-2-ylcarbamate
20. Tybost (tn)
21. Cobicistat [mi]
22. Cobicistat [inn]
23. Cobicistat [jan]
24. Cobicistat; Gs-9350
25. Cobicistat [vandf]
26. Cobicistat [who-dd]
27. Cobicistat On Silicon Dioxide
28. Cobicistat (jan/usan/inn)
29. Gtpl7535
30. Schembl2736227
31. Chembl2095208
32. Cobicistat [orange Book]
33. Cobicistat, (r,r,s)-
34. Dtxsid00143269
35. Evotaz Component Cobicistat
36. Genvoya Component Cobicistat
37. Symtuza Component Cobicistat
38. Bcp06630
39. Ex-a2578
40. Rezolsta Component Cobicistat
41. Stribild Component Cobicistat
42. Bdbm50447471
43. Mfcd18251449
44. Prezcobix Component Cobicistat
45. S2900
46. Zinc85537014
47. Cobicistat Component Of Evotaz
48. Akos025404908
49. Cobicistat Component Of Genvoya
50. Cobicistat Component Of Symtuza
51. Ccg-270459
52. Cobicistat Component Of Stribild
53. Cs-0742
54. Db09065
55. Cobicistat Component Of Prezcobix
56. Ncgc00386235-04
57. Ncgc00386235-06
58. Ac-28961
59. As-17061
60. Hy-10493
61. Sw219553-1
62. D09881
63. Ab01566899_01
64. Q5138908
65. (1,3-thiazol-5-yl)methyl (5s,8r,11r)-8,11-dibenzyl-2-methyl-5-(2-(morpholin-4-yl)ethyl)-1-(2-(propan-2-yl)-1,3-thiazol-4-yl)-3,6-dioxo-2,4,7,12-tetraazatridecan-13-oate
66. 1,3-thiazol-5-ylmethyl ((2r,5r)-5-(((2s)-2-((methyl((2-(propan-2-yl)-1,3-thiazol-4-yl)methyl)carbamoyl)amino)-4-(morpholin-4-yl)butanoyl)amino)-1,6-diphenylhexan-2-yl)carbamate
67. Thiazol-5-ylmethyl ((1r,4r)-1-benzyl-4-({(2s)-2-((methyl{(2-(1-methylethyl)thiazol-4-yl)methyl}carbamoyl)amino)-4-(morpholin-4-yl)butanoyl}amino)-5-phenylpentyl)carbamate
68. Thiazol-5-ylmethyl N-[(1r,4r)-1-benzyl-4-[[(2s)-2-[[(2-isopropylthiazol-4-yl)methyl-methyl-carbamoyl]amino]-4-morpholino-butanoyl]amino]-5-phenyl-pentyl]carbamate
| Molecular Weight | 776.0 g/mol |
|---|---|
| Molecular Formula | C40H53N7O5S2 |
| XLogP3 | 5.7 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 20 |
| Exact Mass | 775.35496016 g/mol |
| Monoisotopic Mass | 775.35496016 g/mol |
| Topological Polar Surface Area | 195 Ų |
| Heavy Atom Count | 54 |
| Formal Charge | 0 |
| Complexity | 1120 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Cobicistat is a CYP3A inhibitor indicated to increase systemic exposure of atazanavir or darunavir (once daily dosing regimen) in combination with other antiretroviral agents in the treatment of HIV-1 infection. It is not interchangeable with ritonavir to increase systemic exposure of darunavir 600 mg twice daily, fosamprenavir, saquinavir, or tipranavir due to lack of exposure data. The use of cobicistat is not recommended with darunavir 600 mg twice daily, fosamprenavir, saquinavir or tipranavir. Complex or unknown mechanisms of drug interactions preclude extrapolation of ritonavir drug interactions to certain cobicistat interactions. Cobicistat and ritonavir when administered with either atazanavir or darunavir may result in different drug interactions when used with concomitant medications.
FDA Label
Tybost is indicated as a pharmacokinetic enhancer of atazanavir 300 mg once daily or darunavir 800 mg once daily as part of antiretroviral combination therapy in human immunodeficiency virus-1 (HIV-1) infected adults and adolescents aged 12 years and older:
- weighing at least 35 kg coadministered with atazanavir or
- weighing at least 40 kg coadministered with darunavir.
Anti-HIV Agents
Agents used to treat AIDS and/or stop the spread of the HIV infection. These do not include drugs used to treat symptoms or opportunistic infections associated with AIDS. (See all compounds classified as Anti-HIV Agents.)
Cytochrome P-450 CYP3A Inhibitors
Drugs and compounds which inhibit or antagonize the biosynthesis or actions of CYTOCHROME P-450 CYP3A. (See all compounds classified as Cytochrome P-450 CYP3A Inhibitors.)
V03AX03
J05AR14
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
V - Various
V03 - All other therapeutic products
V03A - All other therapeutic products
V03AX - Other therapeutic products
V03AX03 - Cobicistat
Absorption
Median peak plasma concentrations were observed at 3.5 hours post-dose.
Route of Elimination
With single dose administration of [14C] cobicistat after multiple dosing of cobicistat for six days, the mean percent of the administered dose excreted in feces and urine was 86.2% and 8.2%, respectively.
Cobicistat is metabolized by CYP3A and to a minor extent by CYP2D6 enzymes and does not undergo glucuronidation.
The terminal plasma half-life of cobicistat is approximately 3 to 4 hours.
Cobicistat is a mechanism-based inhibitor of cytochrome P450 3A (CYP3A) isoforms. Inhibition of CYP3A-mediated metabolism by cobicistat increases the systemic exposure of CYP3A substrates atazanavir and darunavir and therefore enables increased anti-viral activity at a lower dosage. Cobicistat does not have any anti-HIV activity on its own.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
63
PharmaCompass offers a list of Cobicistat API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cobicistat manufacturer or Cobicistat supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cobicistat manufacturer or Cobicistat supplier.
A Cobicistat manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Cobicistat, including repackagers and relabelers. The FDA regulates Cobicistat manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Cobicistat API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Cobicistat manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Cobicistat supplier is an individual or a company that provides Cobicistat active pharmaceutical ingredient (API) or Cobicistat finished formulations upon request. The Cobicistat suppliers may include Cobicistat API manufacturers, exporters, distributors and traders.
click here to find a list of Cobicistat suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Cobicistat DMF (Drug Master File) is a document detailing the whole manufacturing process of Cobicistat active pharmaceutical ingredient (API) in detail. Different forms of Cobicistat DMFs exist exist since differing nations have different regulations, such as Cobicistat USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Cobicistat DMF submitted to regulatory agencies in the US is known as a USDMF. Cobicistat USDMF includes data on Cobicistat's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Cobicistat USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Cobicistat suppliers with USDMF on PharmaCompass.
A Cobicistat written confirmation (Cobicistat WC) is an official document issued by a regulatory agency to a Cobicistat manufacturer, verifying that the manufacturing facility of a Cobicistat active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Cobicistat APIs or Cobicistat finished pharmaceutical products to another nation, regulatory agencies frequently require a Cobicistat WC (written confirmation) as part of the regulatory process.
click here to find a list of Cobicistat suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Cobicistat as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Cobicistat API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Cobicistat as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Cobicistat and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Cobicistat NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Cobicistat suppliers with NDC on PharmaCompass.
Cobicistat Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Cobicistat GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cobicistat GMP manufacturer or Cobicistat GMP API supplier for your needs.
A Cobicistat CoA (Certificate of Analysis) is a formal document that attests to Cobicistat's compliance with Cobicistat specifications and serves as a tool for batch-level quality control.
Cobicistat CoA mostly includes findings from lab analyses of a specific batch. For each Cobicistat CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Cobicistat may be tested according to a variety of international standards, such as European Pharmacopoeia (Cobicistat EP), Cobicistat JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Cobicistat USP).

