
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
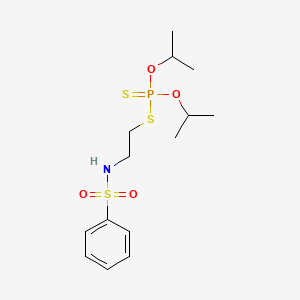

1. 741-58-2
2. Betasan
3. Benzulfide
4. Prefar
5. Disan
6. Sap (herbicide)
7. Disan (pesticide)
8. R 4461
9. N-[2-di(propan-2-yloxy)phosphinothioylsulfanylethyl]benzenesulfonamide
10. O,o-diisopropyl 2-(benzenesulfonamido)ethyl Dithiophosphate
11. Chebi:81899
12. O,o-diisopropyl S-(2-benzenesulfonylaminoethyl) Phosphorodithioate
13. 9882bw2q2s
14. N-(2-mercaptoethyl)benzenesulfonamide S-(o,o-diisopropyl Phosphorodithioate)
15. Phosphorodithioic Acid, O,o-diisopropyl Ester, S-ester With N-(2-mercaptoethyl)benzenesulfonamide
16. Sap
17. Kayaphenone
18. Betamec
19. Exporsan
20. Prefer
21. N-(2-(o,o-diisopropyldithiophosphoryl)ethyl)benzenesulfonamide
22. Betasan E
23. Betasan G
24. Prefar E
25. Pre-san
26. Phosphorodithioic Acid, O,o-bis(1-methylethyl) S-(2-((phenylsulfonyl)amino)ethyl) Ester
27. Phosphorodithioic Acid, O,o-bis(1-methylethyl) S-[2-[(phenylsulfonyl)amino]ethyl] Ester
28. Caswell No. 357
29. Betasan [obsolete]
30. Bensumec
31. Bensulide [bsi:iso]
32. Hsdb 393
33. Einecs 212-010-4
34. Epa Pesticide Chemical Code 009801
35. Brn 2164989
36. (n-(2-mercaptoethyl)benzenesulfonamide)
37. Bensulfide
38. N-[2-(o,o-diisopropyldithiophosphoryl)ethyl]benzenesulfonamide
39. Unii-9882bw2q2s
40. Ccris 9246
41. O,o-diisopropyl S-{2-[(phenylsulfonyl)amino]ethyl} Dithiophosphate
42. R-4461
43. Bensulide [iso]
44. N-(beta-o,o-diisopropyldithiophosphorylethyl)benzenesulfonamide
45. S-2-benzenesulfonamidoethyl O,o-di-isopropyl Phosphorodithioate
46. S-2-benzenesulphonamidoethyl O,o-diisopropyl Phosphorodithioate
47. S-beta-(benzenesulfonamido)ethyl O,o-diisopropyl Dithiophosphate
48. Bensulide [hsdb]
49. O,o-di-isopropyl S-2-phenylsulphonylaminoethyl Phosphorodithioate
50. N-(2-ethylthio)benzene Sulphonamide-s,o,o-diisopropylphosphorodithioate
51. N-(2-mercaptoethylbenzene)sulfonamide S-(o,o-diisopropylphosphorodithioate)
52. O,o-bis(1-methylethyl) S-(2-((phenylsulfonyl)amino)ethyl)pheosphorodithioate
53. O,o-bis(1-methylethyl)-s-(2-((phenylsulfonyl)amino)ethyl) Phosphorodithioate
54. S-(o,o-diisopropylphosphorodithioate) Of N-(2-mercaptoethyl)benzenesulfonamide
55. Benzenesulfonamide, N-(2-mercaptoethyl)-, S-ester With O,o-diisopropylphosphorodithioate
56. Dsstox_cid_12329
57. Dsstox_rid_78915
58. O,o-diisopropyl Dithiophosphate S-ester With N-(2-mercaptoethyl)benzenesulphonamide
59. O,o-diisopropyl Phosphorodithioate S-ester With N-(2-mercaptoethyl)benzenesulfonamide
60. O,o-diisopropyl Phosphorothioate S-ester With N-(2-mercaptoethyl)benzenesulfonamide
61. S-(o,o-diisopropyl Phosphorodithioate) Ester Of N-(2-mercaptoethyl)benzenesulfonamide
62. Dsstox_gsid_32329
63. Schembl55205
64. Chembl1895252
65. Dtxsid9032329
66. Tox21_300733
67. Bensulide 100 Microg/ml In Methanol
68. Akos015902619
69. O,o-bis(1-methylethyl) S-(2-((phenylsulfonyl)amino)ethyl)phosphorodithioate
70. Bensulide 1000 Microg/ml In Methanol
71. Bensulide 10 Microg/ml In Acetonitrile
72. Ncgc00168292-01
73. Ncgc00168292-02
74. Ncgc00168292-03
75. Ncgc00168292-04
76. Ncgc00254639-01
77. Cas-741-58-2
78. Bensulide, Pestanal(r), Analytical Standard
79. C18703
80. Q414584
81. O,o'-diisopropyl S-[2-(benzenesulfonamido)ethyl]dithiophosphate
82. O,o'-diisopropyl S-[2-(benzenesulfonamido)ethyl]phosphorodithioate
83. O,o-diisopropyl S-(2-benzenesulfonylaminoethyl)phosphorodithioate
84. S-.beta.-(benzenesulfonamido)ethyl O,o-diisopropyl Dithiophosphate
85. O,o-diisopropyl S-(2-[(phenylsulfonyl)amino]ethyl) Dithiophosphate #
86. (n-(2-ethylthio)benzene Sulfonamido-s,o,o- Diisopropylphosphorodithioate
87. Phosphorodithioic Acid O,o-bis(1-methylethyl) S-[2-[(phenylsulfonyl)amino]ethyl] Ester
88. Phosphorodithioic Acid-o,o-bis(1-methylethyl)-s-(2-((phenylsulfonyl)amino)ethyl)ester
| Molecular Weight | 397.5 g/mol |
|---|---|
| Molecular Formula | C14H24NO4PS3 |
| XLogP3 | 4.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 10 |
| Exact Mass | 397.06050877 g/mol |
| Monoisotopic Mass | 397.06050877 g/mol |
| Topological Polar Surface Area | 130 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 471 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Selective herbicide, adsorbed on the root surfaces, and a small amount is absorbed by the roots. Translocation of bensulide to the leaves does not occur, but metabolites are translocated.
Tomlin, C.D.S. (ed.). The Pesticide Manual - World Compendium. 10th ed. Surrey, UK: The British Crop Protection Council, 1994., p. 87
In a metabolism study, bensulide technical, labelled with C in the 14 phenyl ring (> 96.4% radiopurity; 925 MBq/mMole) was dissolved in corn oil (vehicle) and administered to Sprague-Dawley rats (5/sex/group; 7-8 weeks of age; 185-235 g body weight) following 3 treatment regimes. Animals in Group I received a single oral dose of radioactive bensulide at 1 mg/kg bw. Animals in Group II received 14 consecutive doses (1 mg/kg/day) of non-radioactive bensulide technical (99% a.i.) in corn oil, followed by a 1 mg/kg dose of radiolabelled bensulide technical in corn oil on day 15. Group III animals received a single oral dose of radiolabelled bensulide technical at 100 mg/kg bw. An additional group of animals (Group IV; 3/sex/group) were given a single oral dose of radiolabelled bensulide 18 technical at 1 mg/kg bw and were subseqently used for autoradiological radiolabelled carbon dioxide release determinations. ... In Group I, total urinary excretion of 7 days after admin of radioactive bensulide technical accounted for 70 and 75% of the administered dose in males and females, respectively. ... In Group III, total urinary excretion accounted for 75 and 87% of the administered dose in males and females, respectively. ... For Group II (prior 14-day admin of non-radioactive bensulide technical before radioactive bensulide admin, both at 1 mg/kg), total urinary excretion of radioactivity over 7 days past dosing with radioactive bensulide accounted for 79 and 88% of the administered dose in males and females, respectively. For Group IV, urinary excretion of 14-C radioactivity derived from bensulide technical over a 48-hr period accounted for 67% for one male and 86% in one male. For Group I, total fecal excretion of radioactivity derived from 14C-bensulide technical over 7 days post-dosing accounted for 22 and 20% of the administered dose in males and females, respectively. ... For Group III, total fecal elim over 7 days post-dosing of bensulide-derived radioactivity accounted for 22 and 11% of the administered dose for males and females, respectively. ... In Group II animals, total fecal excretion of radioactivity over 7 days post-dosing accounted for 14 and 8% of the administered dose for males and females, respectively. ... In Group IV, fecal excretion of radioactivity over 48 hrs post-dosing accounted for 12% of the administered dose in one male and 7% in one female. ... The radioactivity found in the carcasses and in other tissues accounted for 0.3% to 2.5% and <0.1% of the administered dose, respectively.
USEPA; Bensulide: Health Effects Division's Chapter for the Reregistration and Eligibility Decision Document. Washington, DC: USEPA, Off Pest Prog. p.17-18 (March 3, 1998)
Unchanged ring-labelled (14)-C bensulide could be detected in roots of lettuce plants after root application ... . A large part of the radioactive material in the leaves was evolved as radioactive carbon dioxide and some was present as labelled amino acids.
Weed Science Society of America. Herbicide Handbook. 5th ed. Champaign, Illinois: Weed Science Society of America, 1983., p. 49
ANALYTICAL

ABOUT THIS PAGE
79
PharmaCompass offers a list of Bensulide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Bensulide manufacturer or Bensulide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Bensulide manufacturer or Bensulide supplier.
PharmaCompass also assists you with knowing the Bensulide API Price utilized in the formulation of products. Bensulide API Price is not always fixed or binding as the Bensulide Price is obtained through a variety of data sources. The Bensulide Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Bensulide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Bensulide, including repackagers and relabelers. The FDA regulates Bensulide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Bensulide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Bensulide supplier is an individual or a company that provides Bensulide active pharmaceutical ingredient (API) or Bensulide finished formulations upon request. The Bensulide suppliers may include Bensulide API manufacturers, exporters, distributors and traders.
Bensulide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Bensulide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Bensulide GMP manufacturer or Bensulide GMP API supplier for your needs.
A Bensulide CoA (Certificate of Analysis) is a formal document that attests to Bensulide's compliance with Bensulide specifications and serves as a tool for batch-level quality control.
Bensulide CoA mostly includes findings from lab analyses of a specific batch. For each Bensulide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Bensulide may be tested according to a variety of international standards, such as European Pharmacopoeia (Bensulide EP), Bensulide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Bensulide USP).

