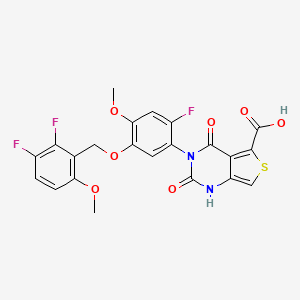

1. 3-(5-((2,3-difluoro-6-methoxyphenyl)methoxy)-2-fluoro-4-methoxyphenyl)-2,4-dioxo-1h-thieno(3,4-d)pyrimidine-5-carboxylic Acid
2. Obe-2109
3. Obe2109
4. Klh2109
5. Klh-2109
1. 935283-04-8
2. Obe-2109
3. Obe2109
4. 7cdw97huex
5. Klh-2109
6. Klh2109
7. Dtxsid801337395
8. 3-[5-[(2,3-difluoro-6-methoxyphenyl)methoxy]-2-fluoro-4-methoxyphenyl]-2,4-dioxo-1h-thieno[3,4-d]pyrimidine-5-carboxylic Acid
9. 3-{5-[(2,3-difluoro-6-methoxyphenyl)methoxy]-2-fluoro-4-methoxyphenyl}-2,4-dioxo-1,2,3,4-tetrahydrothieno[3,4-d]pyrimidine-5-carboxylic Acid
10. Thieno(3,4-d)pyrimidine-5-carboxylic Acid, 3-(5-((2,3-difluoro-6-methoxyphenyl)methoxy)-2-fluoro-4-methoxyphenyl)-1,2,3,4-tetrahydro-2,4-dioxo-
11. Linzagolixum
12. 3-(5-((2,3-difluoro-6-methoxyphenyl)methoxy)-2-fluoro-4-methoxyphenyl)-2,4-dioxo-1,2,3,4-tetrahydrothieno(3,4-d)pyrimidine-5-carboxylic Acid
13. 3-(5-((2,3-difluoro-6-methoxyphenyl)methoxy)-2-fluoro-4-methoxyphenyl)-2,4-dioxo-1h-thieno(3,4-d)pyrimidine-5-carboxylic Acid
14. Refchem:58174
15. Dtxcid101767779
16. Klh 2109
17. Linzagolix [inn]
18. Linzagolix [usan]
19. 3-(5-((2,3-difluoro-6-methoxybenzyl)oxy)-2-fluoro-4-methoxyphenyl)-2,4-dioxo-1,2,3,4-tetrahydrothieno[3,4-d]pyrimidine-5-carboxylic Acid
20. Yselty
21. Klh-2109; Obe-2109
22. Linzagolix (usan/inn)
23. Linzagolix [usan:inn]
24. Unii-7cdw97huex
25. Linzagolix [who-dd]
26. Orb1305022
27. Schembl2333748
28. Chembl3668014
29. Schembl29374644
30. Schembl29720096
31. Schembl30002660
32. Gtpl12283
33. Bdbm160329
34. Glxc-24095
35. Who 10711
36. Akos040748752
37. Ac-38209
38. Da-64989
39. Ms-29443
40. Hy-109093
41. D11608
42. G80955
43. Us9040693, 233
| Molecular Weight | 508.4 g/mol |
|---|---|
| Molecular Formula | C22H15F3N2O7S |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 7 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 143 |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 826 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Linzagolix is indicated for the treatment of moderate to severe symptoms of uterine fibroids in adult women of reproductive age.
H - Systemic hormonal preparations, excl. sex hormones and insulins
H01 - Pituitary and hypothalamic hormones and analogues
H01C - Hypothalamic hormones
H01CC - Anti-gonadotropin-releasing hormones
H01CC04 - Linzagolix
ATCvet Code
QH - Systemic hormonal preparations, excl. sex hormones and insulin
QH01 - Pituitary and hypothalamic hormones and analogues
QH01C - Hypothalamic hormones
QH01CC - Anti-gonadrotropin-releasing hormones
QH01CC04 - Linzagolix
Absorption
Linzagolix is quickly absorbed following oral administration, with Cmax occurring approximately 2 hours following administration.
Route of Elimination
Linzagolix is primarily excreted in the urine, with approximately one-third eliminated via the feces.
Volume of Distribution
After seven days of oral administration of linzagolix 100mg or 200mg, the volume of distribution was 11.067 L and 11.178 L, respectively.
Clearance
The geometric mean apparent clearance following multiple oral doses of linzagolix 100mg or 200mg was 0.522 L/h and 0.499 L/h, respectively.
Up to seven metabolites of linzagolix have been quantified in patient plasma, urine, and feces, although plasma metabolites represent less than 10% of the total linzagolix-related exposure. Two primary demethylated metabolites - KP017 and KP046 - have been identified, with CYP2C9 primarily responsible for the formation of KP017 and CYP2C8, CYP2C9, and CYP3A4 are primarily responsible for the formation of KP046. Unchanged parent drug is the predominant circulating component in human plasma and in the urine, and one of the major components in the feces.
The half-life of linzagolix following multiple doses is approximately 15 hours.
Linzagolix is a selective antagonist of the gonadotropin-releasing hormone (GnRH) receptor. It binds competitively to GnRH receptors in the pituitary gland, thereby inhibiting endogenous signaling and, in turn, the hypothalamic-pituitary-gonadal axis. More specifically, this inhibition of GnRH signaling results in the suppression of both luteinizing hormone and follicle-stimulating hormone signaling, the latter of which is responsible for stimulating the production of estrogen in the ovaries. Linzagolix, therefore, indirectly suppresses estrogen production and signaling, making it useful in the management of estrogen-dependent conditions like uterine fibroids.
BUILDING BLOCK


