

1. Brinsupri
2. Azd7986
3. Azd-7986
4. Ins1007
5. Ins-1007
1. 1802148-05-5
2. Azd7986
3. Ins1007
4. Azd 7986
5. Azd-7986
6. Ins-1007
7. 25cg88l0bb
8. Ins 1007
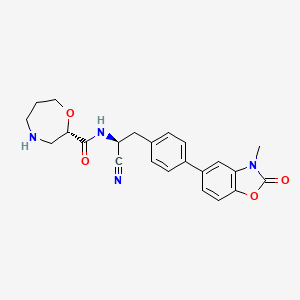
9. (s)-n-((s)-1-cyano-2-(4-(3-methyl-2-oxo-2,3-dihydrobenzo-(d)oxazol-5-yl)phenyl)ethyl)-1,4-oxazepane-2-carboxamide
10. 1,4-oxazepine-2-carboxamide, N-((1s)-1-cyano-2-(4-(2,3-dihydro-3-methyl-2-oxo-5-benzoxazolyl)phenyl)ethyl)hexahydro-, (2s)-
11. (2s)-n-((1s)-1-cyano-2-(4-(3-methyl-2-oxo-2,3-dihydro-1,3-benzoxazol-5-yl)phenyl)ethyl)-1,4-oxazepane-2-carboxamide
12. (2s)-n-{(1s)-1-cyano-2-[4-(3-methyl-2-oxo-2,3-dihydro-1,3-benzoxazol-5-yl)phenyl]ethyl}-1,4-oxazepane-2-carboxamide
13. Refchem:121325
14. (s)-n-((s)-1-cyano-2-(4-(3-methyl-2-oxo-2,3-dihydrobenzo[d]oxazol-5-yl)phenyl)ethyl)-1,4-oxazepane-2-carboxamide
15. Brensocatib [usan]
16. (2s)-n-[(1s)-1-cyano-2-[4-(3-methyl-2-oxo-1,3-benzoxazol-5-yl)phenyl]ethyl]-1,4-oxazepane-2-carboxamide
17. Unii-25cg88l0bb
18. Chembl3900409
19. Brinsupri
20. Who 11097
21. Aexfxnfmsaaelr-rxvvdrjesa-n
22. Brensocatib [inn]
23. Brensocatib (usan/inn)
24. Brensocatib [who-dd]
25. Gtpl9412
26. Orb1296252
27. Schembl16932317
28. Schembl29363739
29. C23h24n4o4
30. Dtxsid001376692
31. Glxc-21270
32. Ex-a1866
33. Azd7986;ins 1007
34. Bdbm50195235
35. Ac-36222
36. As-84225
37. Bb177302
38. Da-71203
39. Hy-101056
40. Cs-0020766
41. D12120
42. D84088
| Molecular Weight | 420.5 g/mol |
|---|---|
| Molecular Formula | C23H24N4O4 |
| XLogP3 | 2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 104 |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 699 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Brensocatib is indicated for the treatment of non-cystic fibrosis bronchiectasis in adult and pediatric patients 12 years of age and older.
Absorption
Brensocatib is rapidly absorbed following oral administration. The median time to maximum plasma concentration (Tmax) is 1.0-1.4 hours after a single dose of 10 mg or 25 mg. The absolute oral bioavailability of brensocatib has not been studied in humans, but based on mass balance studies in healthy subjects, oral absorption is greater than 80%. Following once-daily administration of brensocatib 10 mg or 25 mg, the estimated geometric mean Cmax is 85.4 ng/mL or 259 ng/mL, respectively. In healthy subjects, at steady state, Cmax increased by about 1.5-fold and AUCtau increased by about 2-fold compared to single-dose observations. The presence of food slightly delays the absorption of brensocatib, but does not result in clinically relevant differences in exposure.
Route of Elimination
Following administration of a single oral dose of radiolabeled brensocatib to healthy subjects, 54.2% of the dose was recovered in urine (22.8% as unchanged brensocatib) and 28.3% of the dose was recovered in feces (2.4% as unchanged brensocatib). Available data do not indicate a significant effect of renal impairment on brensocatib elimination and systemic exposure, suggesting that dose adjustment is not necessary in participants with renal impairment.
Volume of Distribution
Following once-daily administration of 10 mg or 25 mg brensocatib in patients with NCFB, the estimated volume of distribution at steady state ranged from 126 to 138 L.
Clearance
The apparent oral clearance of brensocatib ranged from 6.4 to 10.7 L/hour. In a study of healthy Japanese and White adults, mean steady-state CL/F was slightly higher in Japanese participants (10.1, 6.4, and 10.7 L/h for 10 mg, 25 mg, and 40 mg groups, respectively) compared with White participants (8.5, 7.0, and 6.5 L/h).
Brensocatib is primarily metabolized by CYP3A and to a lesser extent by CYP2C8 and CYP2D6. One major circulating metabolite, thiocyanate, was identified in plasma and accounted for 51% of the total radioactivity following administration of a radio-labeled brensocatib dose. In vitro studies indicate that brensocatib is also a weak inducer of CYP3A.
Following a single oral administration of brensocatib in healthy subjects, the elimination half-life ranged from 25 to 39 hours. In a study of healthy Japanese and White adults, the elimination half-life of brensocatib ranged from 22 to 28 hours.
Non-cystic fibrosis bronchiectasis (NCFB) is a chronic lung disease characterized by a cycle of infection, inflammation, and lung tissue damage. Pharmacologic treatment of NCFB is targeted against dipeptidyl peptidase 1 (DPP1), a cysteine protease which plays a crucial role in the activation of pro-inflammatory neutrophil serine proteases (NSPs) during neutrophil maturation in the bone marrow - including cathepsin G (CatG), neutrophil elastase (NE) and proteinase 3 (PR3) - which are implicated in the pathogenesis of neutrophil-mediated NCFB inflammation. Brensocatib is a competitive, reversible inhibitor of dipeptidyl peptidase 1 (DPP1). In cell-based assays, DPP1 inhibition by brensocatib reduces the activity of NSPs including neutrophil elastase, cathepsin G, and proteinase 3.
BUILDING BLOCK

CAS Number : 1802148-05-5
End Use API :
End Use API : Brensocatib
About the Company : Saiming Pharmaceutical is a technology-driven company with 12+ years of experience. Our business involves the R&D, production, and trading of pharmaceutical intermediates, APIs, and fin...
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Saiming Pharmaceutical – Delivering innovative, sustainable chemical solutions with global reach and 12+ years of trusted excellence.

