18 May 2026
// INDPHARMAPOST
17 Mar 2026
// PRESS RELEASE
09 Mar 2026
// FDA
Latest Content by PharmaCompass
 KEY PRODUCTS
KEY PRODUCTS
Synergice, a Strides Pharma initiative, is a new gen B2B platform for diverse portofolio needs.
About
Industry Trade Show
Booth #14D10
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Pharmex Middle EastPharmex Middle East
Industry Trade Show
Not Confirmed
26-28 May, 2026
CONTACT DETAILS




Events
Webinars & Exhibitions
Industry Trade Show
Booth #14D10
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Pharmex Middle EastPharmex Middle East
Industry Trade Show
Not Confirmed
26-28 May, 2026
CORPORATE CONTENT #SupplierSpotlight
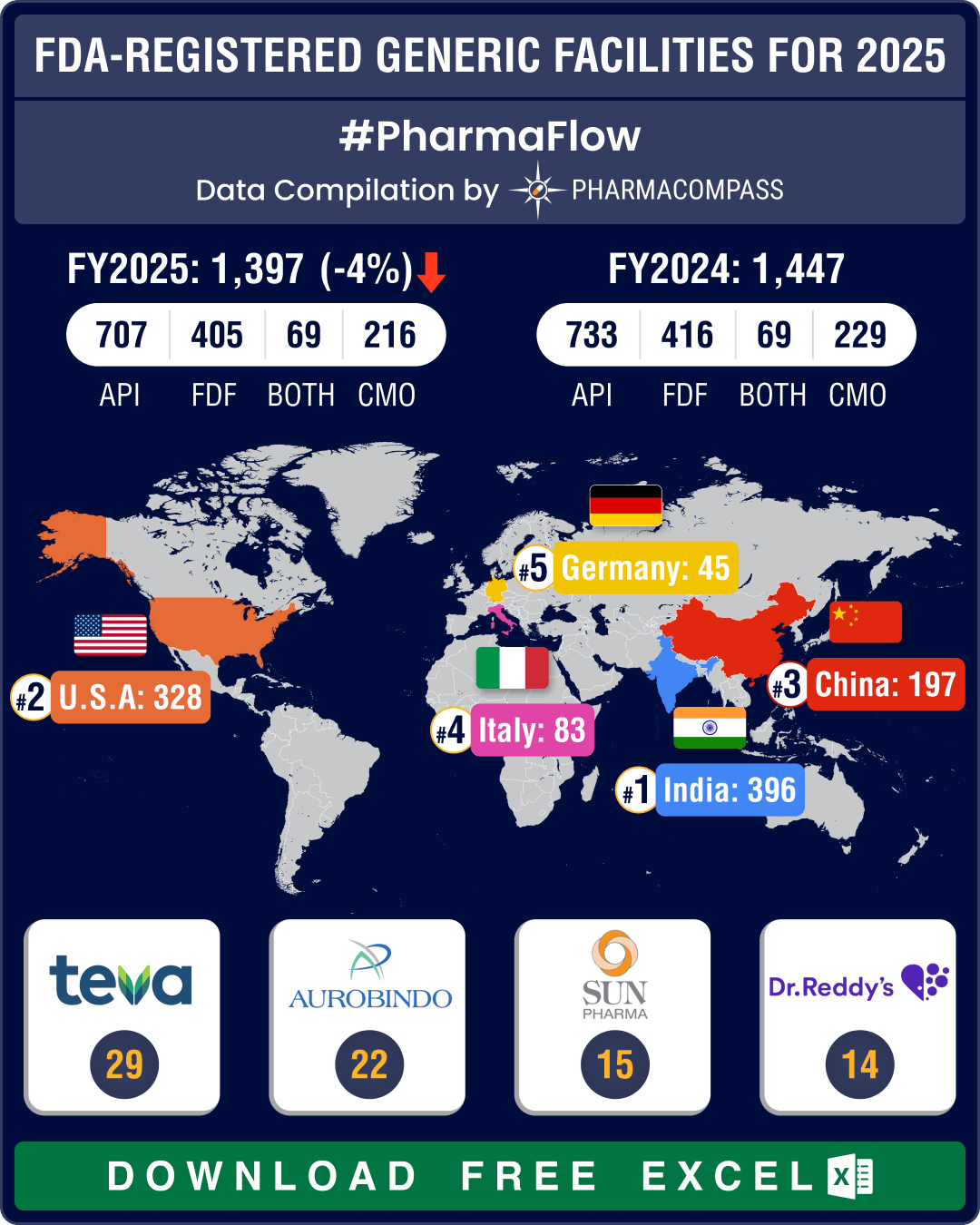
https://www.pharmacompass.com/radio-compass-blog/chinese-fda-registered-generic-facilities-gain-steam-india-maintains-lead-with-396-facilities

18 May 2026
// INDPHARMAPOST
https://www.indianpharmapost.com/biopharma/strides-pharma-reports-rs-24897-million-revenue-during-fy26-2-yoy-growth-20237

17 Mar 2026
// PRESS RELEASE
https://www.strides.com/Upload/PDF/pr-strides-africa-business-acquisition-multiple-generic-sandoz.pdf

09 Mar 2026
// FDA
https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=207399

30 Jan 2026
// PRESS RELEASE
https://www.strides.com/Upload/PDF/strides-q3fy26-investor-presentation.pdf

04 Jan 2026
// INDPHARMAPOST
https://www.indianpharmapost.com/people/strides-appoints-yajuvendra-singh-m-r-as-vp-digital-transformation-18763

03 Dec 2025
// PRESS RELEASE
https://www.strides.com/Upload/PDF/strides-movement-senior-management-personnel-03122025.pdf
Inspections and registrations
ABOUT THIS PAGE


 Strides Pharma Science
Strides Pharma Science