18 May 2026
// INDPHARMAPOST
17 Mar 2026
// PRESS RELEASE
09 Mar 2026
// FDA
Latest Content by PharmaCompass
 KEY PRODUCTS
KEY PRODUCTS
Synergice, a Strides Pharma initiative, is a new gen B2B platform for diverse portofolio needs.
About
Industry Trade Show
Booth #14D10
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Pharmex Middle EastPharmex Middle East
Industry Trade Show
Not Confirmed
26-28 May, 2026
CONTACT DETAILS




Events
Webinars & Exhibitions
Industry Trade Show
Booth #14D10
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Pharmex Middle EastPharmex Middle East
Industry Trade Show
Not Confirmed
26-28 May, 2026
CORPORATE CONTENT #SupplierSpotlight
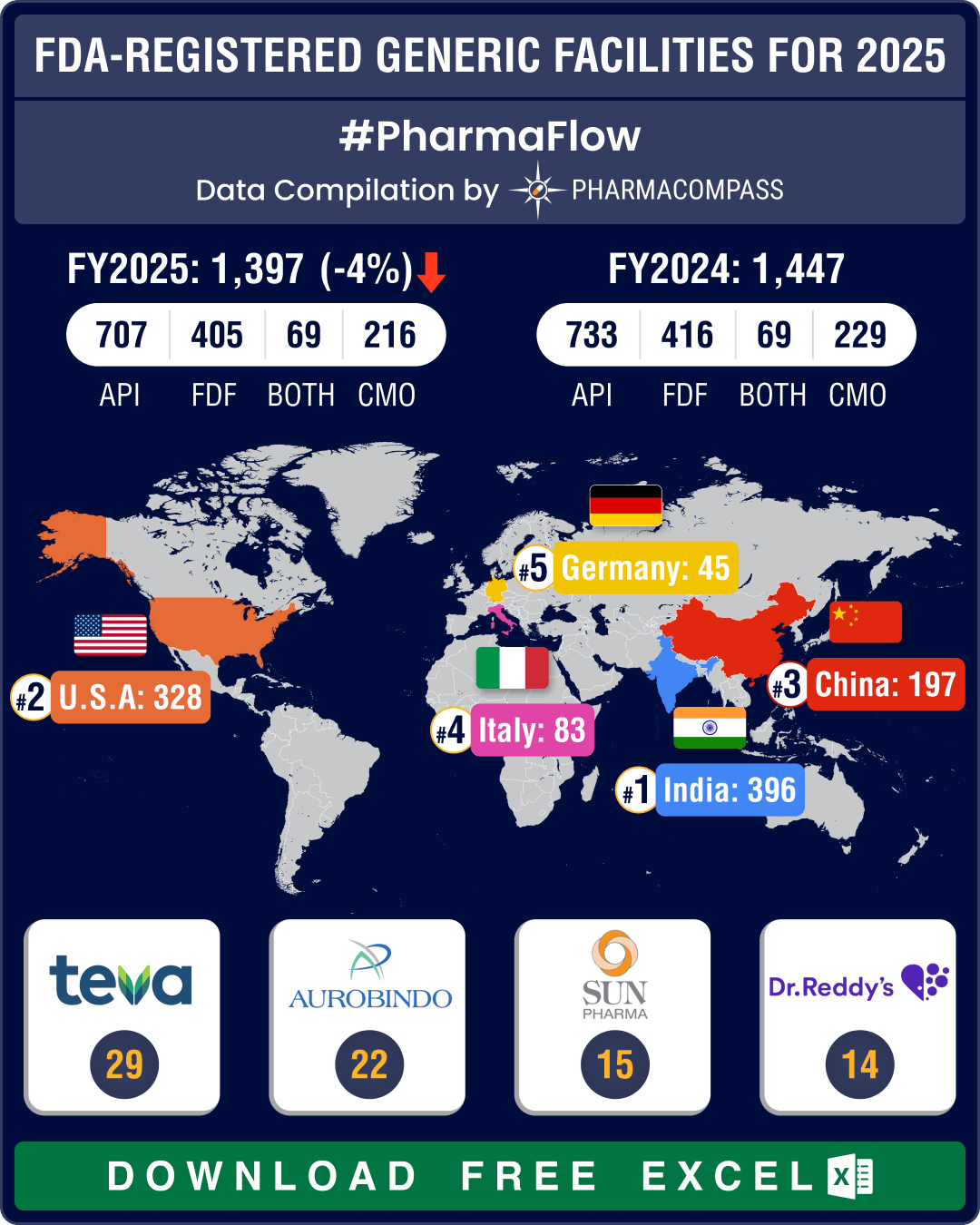
https://www.pharmacompass.com/radio-compass-blog/chinese-fda-registered-generic-facilities-gain-steam-india-maintains-lead-with-396-facilities

18 May 2026
// INDPHARMAPOST
https://www.indianpharmapost.com/biopharma/strides-pharma-reports-rs-24897-million-revenue-during-fy26-2-yoy-growth-20237

17 Mar 2026
// PRESS RELEASE
https://www.strides.com/Upload/PDF/pr-strides-africa-business-acquisition-multiple-generic-sandoz.pdf

09 Mar 2026
// FDA
https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=207399

30 Jan 2026
// PRESS RELEASE
https://www.strides.com/Upload/PDF/strides-q3fy26-investor-presentation.pdf

04 Jan 2026
// INDPHARMAPOST
https://www.indianpharmapost.com/people/strides-appoints-yajuvendra-singh-m-r-as-vp-digital-transformation-18763

03 Dec 2025
// PRESS RELEASE
https://www.strides.com/Upload/PDF/strides-movement-senior-management-personnel-03122025.pdf
Details:
Under the acquisition, an undisclosed miscellaneous product for infection diseases was acquired to advance research in this key focus area.
Lead Product(s): Undisclosed
Therapeutic Area: Infections and Infectious Diseases Brand Name: Undisclosed
Study Phase: Approved FDFProduct Type: Miscellaneous
Recipient: Sandoz B2B
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Acquisition March 17, 2026
Lead Product(s) : Undisclosed
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved FDF
Recipient : Sandoz B2B
Deal Size : Undisclosed
Deal Type : Acquisition
Strides Acquires Multiple Generic Brands from Sandoz to Strengthen Africa Business
Details : Under the acquisition, an undisclosed miscellaneous product for infection diseases was acquired to advance research in this key focus area.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Undisclosed
March 17, 2026
Details:
Medroxyprogesterone Acetate, a hormone targeting the progesterone receptor, shows promise in contraception.
Lead Product(s): Medroxyprogesterone Acetate
Therapeutic Area: Obstetrics/Gynecology (Women’s Health) Brand Name: Medogen SubQ
Study Phase: Approved FDFProduct Type: Hormone
Recipient: Incepta Pharmaceuticals
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable October 23, 2025
Lead Product(s) : Medroxyprogesterone Acetate
Therapeutic Area : Obstetrics/Gynecology (Women’s Health)
Highest Development Status : Approved FDF
Recipient : Incepta Pharmaceuticals
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides to Market WHO-Prequalified Generic DMPA-SC in Africa
Details : Medroxyprogesterone Acetate, a hormone targeting the progesterone receptor, shows promise in contraception.
Product Name : Medogen SubQ
Product Type : Hormone
Upfront Cash : Inapplicable
October 23, 2025
Details:
The portfolio consists of liquids, including Nitrofurantoin, and immediate release solid orals to treat infections associated with UTIs, Pain Management, Allergy Symptoms, ADHD and Narcolepsy.
Lead Product(s): Nitrofurantoin
Therapeutic Area: Infections and Infectious Diseases Brand Name: Nitrofurantoin-Generic
Study Phase: Approved FDFProduct Type: Antibiotic
Recipient: Nostrum Laboratories
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Acquisition May 03, 2025
Lead Product(s) : Nitrofurantoin
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved FDF
Recipient : Nostrum Laboratories
Deal Size : Undisclosed
Deal Type : Acquisition
Strides Acquires Identified ANDAs from Nostrum Laboratories, USA
Details : The portfolio consists of liquids, including Nitrofurantoin, and immediate release solid orals to treat infections associated with UTIs, Pain Management, Allergy Symptoms, ADHD and Narcolepsy.
Product Name : Nitrofurantoin-Generic
Product Type : Antibiotic
Upfront Cash : Undisclosed
May 03, 2025
Details:
Celebrex-Generic (celecoxib) is a COX-2 inhibitor, small molecule drug candidate. It is indicated for the treatment of osteoarthritis & rheumatoid arthritis in adults.
Lead Product(s): Celecoxib
Therapeutic Area: Musculoskeletal Brand Name: Celebrex-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 30, 2025
Lead Product(s) : Celecoxib
Therapeutic Area : Musculoskeletal
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides Pharma receives USFDA approval for Celecoxib Capsules in multiple strengths
Details : Celebrex-Generic (celecoxib) is a COX-2 inhibitor, small molecule drug candidate. It is indicated for the treatment of osteoarthritis & rheumatoid arthritis in adults.
Product Name : Celebrex-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 30, 2025
Details:
The company has received approval from the US Food and Drug Administration (USFDA) for generic version for Acetaminophen and Ibuprofen tablets (250 mg/125 mg) indicated for pain and inflammation.
Lead Product(s): Ibuprofen,Paracetamol
Therapeutic Area: Neurology Brand Name: Advil Dual Action
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable January 20, 2025
Lead Product(s) : Ibuprofen,Paracetamol
Therapeutic Area : Neurology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides Gains USFDA Approval for Acetaminophen and Ibuprofen Tablets, 125 mg /250 mg
Details : The company has received approval from the US Food and Drug Administration (USFDA) for generic version for Acetaminophen and Ibuprofen tablets (250 mg/125 mg) indicated for pain and inflammation.
Product Name : Advil Dual Action
Product Type : Miscellaneous
Upfront Cash : Inapplicable
January 20, 2025
Details:
Fluoxetine hydrochloride, is an antidepressant of the SSRIs class. It is used for the treatment of major depressive disorder, obsessive–compulsive disorder, bulimia nervosa, etc.
Lead Product(s): Fluoxetine Hydrochloride
Therapeutic Area: Psychiatry/Psychology Brand Name: Prozac-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 16, 2024
Lead Product(s) : Fluoxetine Hydrochloride
Therapeutic Area : Psychiatry/Psychology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides Pharma Subsidiary Receives USFDA Approval for 60 mg Fluoxetine Tablets
Details : Fluoxetine hydrochloride, is an antidepressant of the SSRIs class. It is used for the treatment of major depressive disorder, obsessive–compulsive disorder, bulimia nervosa, etc.
Product Name : Prozac-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 16, 2024
Details:
Theophylline extended-release tablets formulation approved for the use in the treatment of asthma and chronic obstructive pulmonary disease.
Lead Product(s): Theophylline
Therapeutic Area: Pulmonary/Respiratory Diseases Brand Name: Theo-Dur-Generic
Study Phase: Approved FDFProduct Type: Plant Extract/Herbal
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 06, 2024
Lead Product(s) : Theophylline
Therapeutic Area : Pulmonary/Respiratory Diseases
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides Gets USFDA Nod for Theophylline Extended-Release Tablets
Details : Theophylline extended-release tablets formulation approved for the use in the treatment of asthma and chronic obstructive pulmonary disease.
Product Name : Theo-Dur-Generic
Product Type : Plant Extract/Herbal
Upfront Cash : Inapplicable
September 06, 2024
Details:
Carafate-generic (sucralfate) is a USFDA approved pepsin A inhibitor small molecule drug candidate, which is indicated for the treatment of active duodenal ulcer.
Lead Product(s): SucrAlphate
Therapeutic Area: Gastroenterology Brand Name: Carafate-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 22, 2024
Lead Product(s) : SucrAlphate
Therapeutic Area : Gastroenterology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides receives USFDA approval for Sucralfate Oral Suspension, 1gm/10 mL
Details : Carafate-generic (sucralfate) is a USFDA approved pepsin A inhibitor small molecule drug candidate, which is indicated for the treatment of active duodenal ulcer.
Product Name : Carafate-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
May 22, 2024
Details:
USFDA approved generic version of Sevelamer Carbonate which is used to manage elevated levels of phosphate in the blood of patients with chronic kidney disease.
Lead Product(s): Sevelamer Hydrochloride
Therapeutic Area: Nephrology Brand Name: Renvela-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 11, 2024
Lead Product(s) : Sevelamer Hydrochloride
Therapeutic Area : Nephrology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides Receives USFDA Approval for Sevelamer Carbonate Tablets
Details : USFDA approved generic version of Sevelamer Carbonate which is used to manage elevated levels of phosphate in the blood of patients with chronic kidney disease.
Product Name : Renvela-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
May 11, 2024
Details:
Fluoxetine hydrochloride (Prozac) is an SSRI antidepressant used for major depressive disorder, OCD, bulimia, and panic disorder.
Lead Product(s): Fluoxetine Hydrochloride
Therapeutic Area: Psychiatry/Psychology Brand Name: Prozac-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 09, 2024
Lead Product(s) : Fluoxetine Hydrochloride
Therapeutic Area : Psychiatry/Psychology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Strides Pharma Gets USFDA Nod for Antidepressant Medication
Details : Fluoxetine hydrochloride (Prozac) is an SSRI antidepressant used for major depressive disorder, OCD, bulimia, and panic disorder.
Product Name : Prozac-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 09, 2024
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Regulatory Info :
Registration Country : India
Dosage Form : Tablet
Brand Name : Abacavir
Dosage Strength : 300MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
Regulatory Info : RX
Registration Country : USA
Dosage Form : TABLET;ORAL
Brand Name : ACARBOSE
Dosage Strength : 25MG
Packaging :
Approval Date : 2011-07-27
Application Number : 90912
Regulatory Info : RX
Registration Country : USA
Regulatory Info : RX
Registration Country : USA
Dosage Form : TABLET;ORAL
Brand Name : ACARBOSE
Dosage Strength : 50MG
Packaging :
Approval Date : 2011-07-27
Application Number : 90912
Regulatory Info : RX
Registration Country : USA
Regulatory Info : RX
Registration Country : USA
Dosage Form : TABLET;ORAL
Brand Name : ACARBOSE
Dosage Strength : 100MG
Packaging :
Approval Date : 2011-07-27
Application Number : 90912
Regulatory Info : RX
Registration Country : USA
Regulatory Info :
Registration Country : India
Dosage Form : Tablet
Brand Name : Acarbose
Dosage Strength : 25MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
Regulatory Info :
Registration Country : India
Dosage Form : Tablet
Brand Name : Acarbose
Dosage Strength : 50MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
Regulatory Info :
Registration Country : India
Dosage Form : Tablet
Brand Name : Acarbose
Dosage Strength : 100MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
Regulatory Info : RX
Registration Country : USA
ACETAMINOPHEN; BUTALBITAL; CAFFEINE
Dosage Form : TABLET;ORAL
Brand Name : BUTALBITAL, ACETAMINOPHEN...
Dosage Strength : 325MG;50MG;40MG
Packaging :
Approval Date : 2003-08-27
Application Number : 40511
Regulatory Info : RX
Registration Country : USA
Regulatory Info : DISCN
Registration Country : USA
ACETAMINOPHEN; BUTALBITAL; CAFFEINE
Dosage Form : TABLET;ORAL
Brand Name : BUTALBITAL, ACETAMINOPHEN...
Dosage Strength : 325MG;50MG;40MG
Packaging :
Approval Date : 2020-09-21
Application Number : 203647
Regulatory Info : DISCN
Registration Country : USA
Regulatory Info :
Registration Country : India
Acetaminophen; Butalbital; Caffeine
Dosage Form : Tablet
Brand Name : Acetaminophen; Butalbital...
Dosage Strength : 50MG; 325MG; 40MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Inspections and registrations
ABOUT THIS PAGE


 Strides Pharma Science
Strides Pharma Science