05 Jun 2026
// PR NEWSWIRE
05 Jun 2026
// PR NEWSWIRE
05 Jun 2026
// PR NEWSWIRE
Latest Content by PharmaCompass
 KEY PRODUCTS
KEY PRODUCTS
Apex Healthcare Limited: ISO 9001:2008, WHO-GMP, US FDA-audited manufacturer & exporter of APIs, bulk drugs, and formulations.
About
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Industry Trade Show
Not Confirmed
03-05 June, 2026
Peptide Therapeutics F...Peptide Therapeutics Forum 2026
Industry Trade Show
Not Confirmed
04-05 June, 2026
CONTACT DETAILS




Events
Webinars & Exhibitions
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Industry Trade Show
Not Confirmed
03-05 June, 2026
Peptide Therapeutics F...Peptide Therapeutics Forum 2026
Industry Trade Show
Not Confirmed
04-05 June, 2026
CORPORATE CONTENT #SupplierSpotlight
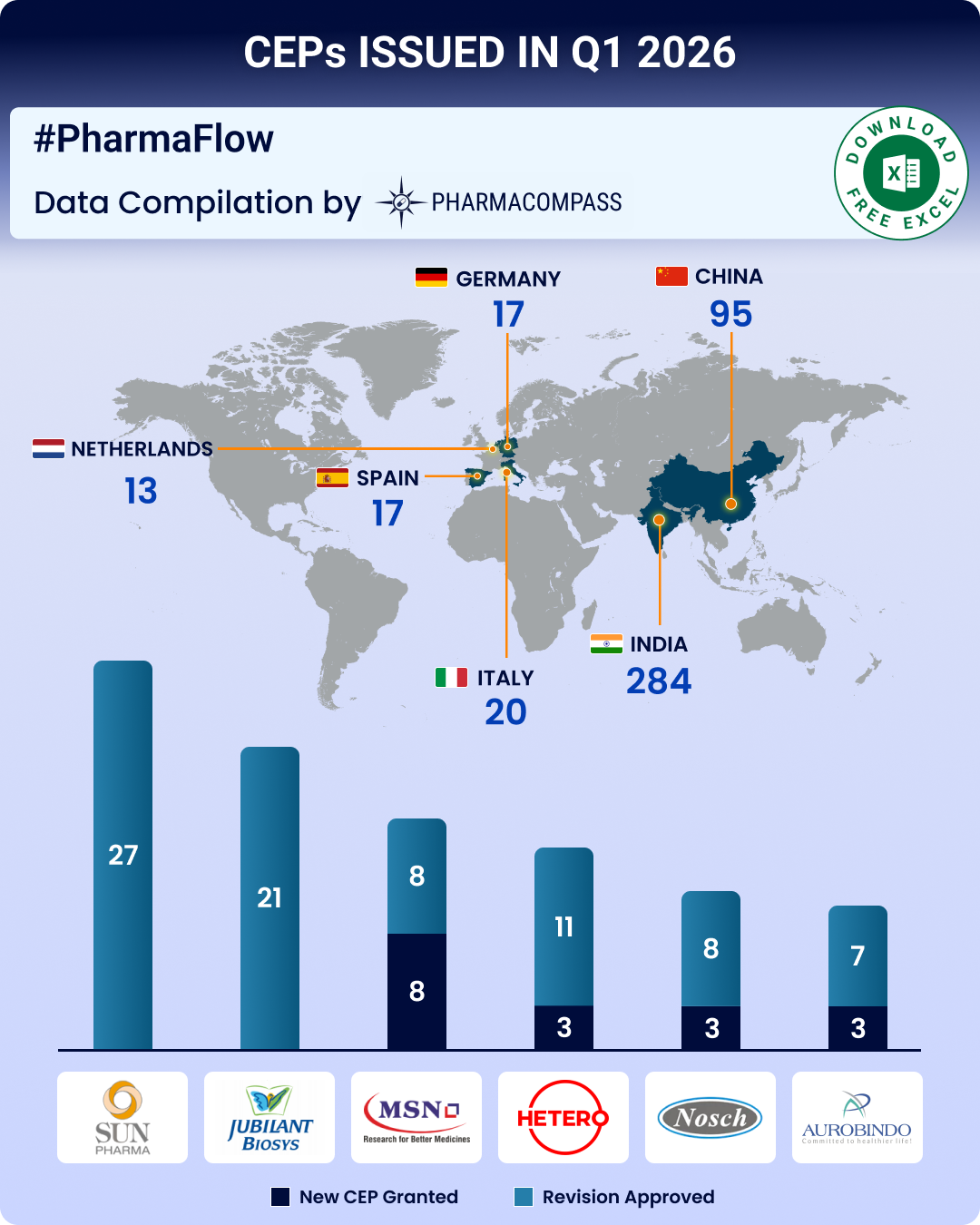
https://www.pharmacompass.com/radio-compass-blog/cep-q1-2026-update-cep-2-0-edqm-s-new-guidelines-strengthen-ecosystem-indian-firms-top-list-of-ceps-issued
https://www.pharmacompass.com/radio-compass-blog/dmf-filings-rise-4-5-in-q3-2025-china-holds-lead-india-records-20-growth-in-submissions
ABOUT THIS PAGE
Apex Healthcare Limited is a supplier offers 23 products (APIs, Excipients or Intermediates).
Find Benzocaine bulk with DMF, CEP, WC offered by Apex Healthcare Limited
Find Lidocaine bulk with DMF, CEP, WC offered by Apex Healthcare Limited
Find Lidocaine Hydrochloride bulk with DMF, CEP, WC offered by Apex Healthcare Limited
Find Meloxicam bulk with DMF, CEP, WC offered by Apex Healthcare Limited
Find Piroxicam bulk with DMF, CEP, WC offered by Apex Healthcare Limited
Find Acyclovir bulk with DMF, WC offered by Apex Healthcare Limited
Find Bisacodyl bulk with DMF, WC offered by Apex Healthcare Limited
Find Prilocaine bulk with DMF, WC offered by Apex Healthcare Limited
Find Alendronate Sodium bulk with WC offered by Apex Healthcare Limited
Find Articaine Hydrochloride bulk with WC offered by Apex Healthcare Limited
Find Bupivacaine Hydrochloride bulk with DMF offered by Apex Healthcare Limited
Find Lornoxicam bulk with WC offered by Apex Healthcare Limited
Find Phenyramidol Hydrochloride bulk with WC offered by Apex Healthcare Limited
Find Ropivacaine Hydrochloride bulk with DMF offered by Apex Healthcare Limited
Find Rosuvastatin Calcium bulk with WC offered by Apex Healthcare Limited
Find Sodium Picosulfate bulk with WC offered by Apex Healthcare Limited
Find Bupivacaine bulk offered by Apex Healthcare Limited
Find Dextromethorphan Hydrobromide bulk offered by Apex Healthcare Limited
Find Dimethyl Fumarate bulk offered by Apex Healthcare Limited
Find Prilocaine Hydrochloride bulk offered by Apex Healthcare Limited
Find Tenoxicam bulk offered by Apex Healthcare Limited
Find Tetracaine bulk offered by Apex Healthcare Limited
Find Urea Hydrogen Peroxide bulk offered by Apex Healthcare Limited


 Apex Healthcare Limited
Apex Healthcare Limited