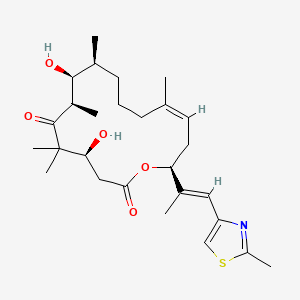

1. Desoxyepothilone B
2. 12,13-desoxyepothilone B
3. Depob Cpd
4. Epothilon D
5. Epothiolone D
6. Epothilone B, Desoxy
7. Kos 862
8. Kos-862
9. Kos862
1. 189453-10-9
2. Desoxyepothilone B
3. (-)-desoxyepothilone B
4. Kos-862
5. 12,13-desoxyepothilone B
6. (-)-epothilone D
7. Epothilon D
8. Nsc-703147
9. Kos 862
10. 12,13-deoxyepothilone B
11. Epothiolone D
12. Depob Cpd
13. Oxacyclohexadec-13-ene-2,6-dione, 4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]-, (4s,7r,8s,9s,13z,16s)-
14. Nsc 703147
15. T0358e0yuf
16. Epothilone B, Desoxy
17. 12,13-desoxyepothilone B, Cis
18. Kos862
19. R1492
20. Chebi:29579
21. Dtxsid70880053
22. Nsc703147
23. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-1-oxacyclohexadec-13-ene-2,6-dione
24. Oxacyclohexadec-13-ene-2,6-dione, 4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-((1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl)-, (4s,7r,8s,9s,13z,16s)-
25. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-((e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl)-1-oxacyclohexadec-13-ene-2,6-dione
26. Refchem:137339
27. Dtxcid901022595
28. (4s-(4r*,7s,8r*,9r*,13z,16r*(e)))-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-(1-methyl-2-(2-methyl-4-thiazolyl)ethenyl)oxacyclohexadec-13-ene-2,6-dione
29. R-1492
30. Mfcd27976357
31. (4s,7r,8s,9s,16s,z)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-((e)-1-(2-methylthiazol-4-yl)prop-1-en-2-yl)oxacyclohexadec-13-ene-2,6-dione
32. Epothilone D (synthetic)
33. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(e)-1-methyl-2-(2-methylthiazol-4-yl)vinyl]-1-oxacyclohexadec-13-ene-2,6-dione
34. Epd
35. Unii-t0358e0yuf
36. Utidelone
37. Epothilone-d
38. Depob
39. Utidelone [inn]
40. Epo D
41. Epothilone D [mi]
42. Schembl4415
43. 12,13-desoxy-epothilone B
44. Chembl96172
45. Z-12,13-desoxyepothilone B
46. Epothilone D [who-dd]
47. C27h41no5s
48. Orb1679023
49. Ex-a495
50. Glxc-06445
51. Biologically Synthesized Epothilone D
52. Lmpk04000001
53. Nsc721085
54. Akos025401598
55. Cs-0655
56. Db01873
57. Fe22752
58. Nsc-721085
59. Ac-22616
60. Hy-15278
61. Brd-k52762805-001-01-2
62. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-(1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl-oxacyclohexadec-13-ene-2,6-dione
63. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(1e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-1-oxacyclohexadec-13-ene-2,6-dione
64. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(1e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]oxacyclohexadec-13-ene-2,6-dione
65. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]oxacyclohexadec-13-ene- 2,6-dione
66. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]oxacyclohexadec-13-ene-2,6-dione; (-)-desoxyepothilone B; (-)-epothilone D; 12,13-deoxyepo
67. (4s,7r,8s,9s,13z,16s)-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-[(e)-1-methyl-2-(2-methyl-1,3-thiazol-4-yl)ethenyl]oxacyclohexadec-13-ene-2,6-dione
68. (4s-(4r*,8r*,9r*,13z,16r*(e)))-4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-(1-methyl-2-(2-methyl-4-thiazolyl)ethenyl)oxacyclohexadec-13-ene-2,6-dione
69. Oxacyclohexadec-13-ene-2,6-dione, 4,6-dihydroxy-5,5,7,9,13-pentamethyl-16-((1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl)-, (4s,7r,8s,9s,13z,16s)-
70. Oxacyclohexadec-13-ene-2,6-dione, 4,8-dihydroxy-5,5,7,9,13-pentamethyl-16-(1-methyl-2-(2-methyl-4-thiazolyl)ethenyl)-, (4s-(4r*,7s,8r*,9r*,13z,16r*(e)))-
| Molecular Weight | 491.7 g/mol |
|---|---|
| Molecular Formula | C27H41NO5S |
| XLogP3 | 5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 2 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 125 |
| Heavy Atom Count | 34 |
| Formal Charge | 0 |
| Complexity | 777 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 5 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 2 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Investigated for use/treatment in colorectal cancer, lung cancer, breast cancer, solid tumors, and prostate cancer.
Tubulin Modulators
Agents that interact with TUBULIN to inhibit or promote polymerization of MICROTUBULES.
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01D - Cytotoxic antibiotics and related substances
L01DC - Other cytotoxic antibiotics
L01DC05 - Utidelone
ATCvet Code
QL - Antineoplastic and immunomodulating agents
QL01 - Antineoplastic agents
QL01D - Cytotoxic antibiotics and related substances
QL01DC - Other cytotoxic antibiotics
QL01DC05 - Utidelone
The principal mechanism of the epothilone class is inhibition of microtubule function. Microtubules are essential to cell division, and epothilones therefore stop cells from properly dividing.
