


1. Arq 197
2. Arq-197
3. Arq197
1. 905854-02-6
2. Arq-197
3. Arq 197
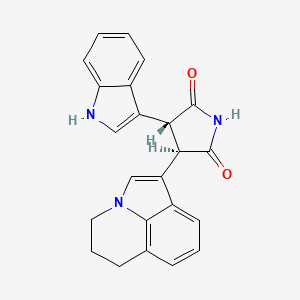
4. (3r,4r)-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione
5. Arq197
6. Pj4h73il17
7. Dtxsid60920316
8. (3r,4r)-3-(5,6-dihydro-4h-pyrrolo(3,2,1-ij)quinolin-1-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione
9. Tivantinibum
10. Refchem:898517
11. C-met Inhibitor Arq 197
12. Dtxcid301349230
13. (3r,4r)-3-(1-azatricyclo(6.3.1.0^(4,12))dodeca-2,4,6,8(12)-tetraen-3-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione
14. Tivantinib (arq 197)
15. 905853-99-8
16. Mfcd11977597
17. (rel)-tivantinib
18. Rel-(3r,4r)-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl)-2,5-pyrrolidinedione
19. 1000873-98-2
20. (3r,4r)-3-(1-azatricyclo[6.3.1.04,12]dodeca-2,4,6,8(12)-tetraen-3-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione
21. Tivantinib [inn]
22. Tivantinib [usan:inn]
23. Unii-pj4h73il17
24. Tivantinib; Arq197
25. Tivantinib [mi]
26. Tivantinib [jan]
27. Tivantinib [usan]
28. Arq 197 (tivantinib)
29. Tivantinib [who-dd]
30. Schembl44944
31. Orb322231
32. Tivantinib (jan/usan/inn)
33. Gtpl7948
34. Orb1301058
35. Orb1941247
36. Chembl2103882
37. Schembl29355700
38. Chebi:91398
39. Uceqxrcjxivodc-pmacekpbsa-n
40. Arq-197,tivantinib, Arq197
41. Arq197; Arq-197;tivantinib
42. Cs-m1640
43. Ex-a2169
44. Cas:905854-02-6;tivantinib
45. Bdbm50146168
46. Nsc758242
47. Nsc800951
48. S2753
49. Akos022182739
50. Ccg-268285
51. Db12200
52. Ebc-218037
53. Ft44530
54. Nsc-758242
55. Nsc-800951
56. Sdccgsbi-0654487.p001
57. (-)-trans-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione
58. Ac-25009
59. Ac-35963
60. As-16991
61. Da-67145
62. Da-68209
63. Hy-50686
64. Hy-77493
65. Db-362059
66. Ns00073191
67. C77073
68. D10173
69. E82820
70. Brd-k33379087-001-01-8
71. Brd-k33379087-001-07-5
72. Q17123902
73. Z2242166213
74. (+/-)-trans-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4(1h-indol-3-yl)pyrrolidine-2,5-dione
75. (+/-)-trans-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl) Pyrrolidine-2,5-dione
76. (+/-)-trans-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione
77. (_)-trans-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4(1h-indol-3-yl)pyrrolidine-2,5-dione
78. (3r,4r)-3-(2,3-dihydro-1h-pyrrolo[3,2,1-ij]quinolin-6-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione
79. (3r,4r)-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl)-pyrrolidine-2,5-dione
80. (3r,4r)-3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl)pyrrolidine-2,5-dione; Arq 197;
81. 2,5-pyrrolidinedione, 3-(5,6-dihydro-4h-pyrrolo(3,2,1-ij)quinolin-1-yl)-4-(1h-indol-3-yl)-, (3r,4r)-
82. Tiv
83. Trans 3-(5,6-dihydro-4h-pyrrolo[3,2,1-ij]quinolin-1-yl)-4-(1h-indol-3-yl)-pyrrolidine-2,5-dione
| Molecular Weight | 369.4 g/mol |
|---|---|
| Molecular Formula | C23H19N3O2 |
| XLogP3 | 2.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 2 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 66.9 |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 666 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Treatment of hepatoblastoma
Tivantinib mediates its effects by inhibiting the activity of c-Met, a receptor tyrosine kinase that plays multiple key roles in human cancer, including cancer cell growth, survival, angiogenesis, invasion and metastasis. C-Met is abnormally activated in most cancers and is believed to control multiple signal transduction pathways involved in tumor growth and metastasis.
