

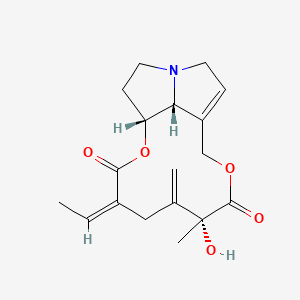

1. Seneciphylline, (12 Beta,15e)-isomer
1. 480-81-9
2. Seneciphyllin
3. Jacodine
4. Nsc30622
5. Chebi:9108
6. 13,19-didehydro-12-hydroxysenecionan-11,16-dione
7. Mls000737616
8. 0zyz9l5454
9. Nsc-30622
10. [1,6]dioxacyclododecino[2,3,4-gh]pyrrolizine-2,7-dione,3-ethylidene-3,4,5,6,9,11,13,14,14a,14b-decahydro-6-hydroxy-6-methyl-5-methylene-, (3z,6r,14ar,14br)-
11. Nsc 30622
12. (1r,4z,7r,17r)-4-ethylidene-7-hydroxy-7-methyl-6-methylidene-2,9-dioxa-14-azatricyclo[9.5.1.014,17]heptadec-11-ene-3,8-dione
13. Seniciphylline
14. Seneciphylline, Analytical Standard
15. Smr000393787
16. Nci-c61165
17. Senecionan-11,16-dione, 13,19-didehydro-12-hydroxy-
18. Seneciphynine
19. Unii-0zyz9l5454
20. Ccris 4336
21. Hsdb 5188
22. Prestwick_446
23. Alpha-longilobine
24. Prestwick2_000599
25. Prestwick3_000599
26. .alpha.-longilobine
27. Seneciphylline [mi]
28. Bspbio_000478
29. Seneciphylline [hsdb]
30. Seneciphylline [iarc]
31. Mls002222214
32. Schembl177867
33. Bpbio1_000526
34. Chembl523911
35. Dtxsid8026016
36. Chebi:92677
37. Hms1569h20
38. Hms2096h20
39. Hms2763j21
40. (1,6)dioxacyclododecino(2,3,4-gh)pyrrolizine-2,7-dione, 3-ethylidene-3,4,5,6,9,11,13,14,14a,14b-decahydro-6-hydroxy-6-methyl-5-methylene-, (3z,6r,14ar,14br)-
41. Hy-n1282
42. Bdbm50480273
43. Mfcd00221722
44. Zinc95627757
45. Akos024282719
46. Trans-15-ethylidene-12beta-hydroxy-12alpha-methyl-13-methylenesenec-1-enine
47. Ncgc00017384-04
48. Ncgc00142562-03
49. (1r,7r,17r)-4-ethylidene-7-hydroxy-7-methyl-6-methylene-2,9-dioxa-14-azatricyc Lo[9.5.1.0<14,17>]heptadec-11-ene-3,8-dione
50. Ac-34599
51. Cs-0016686
52. Senecionan-11, 13,19-didehydro-12-hydroxy-
53. (z)-ethylidene-hydroxy-methyl-methylene-[?]dione
54. Brd-k38449220-001-03-8
55. Q27108272
56. 2h-pyran-2-carboxylicacid,5-ethyltetrahydro-2,3-dimethyl-6-oxo-(9ci)
57. (3z,6r,14ar,14br)-3-ethylidene-3,4,5,6,9,11,13,14,14a,14b-decahydro-6-hydroxy-6-methyl-5-methylene(1,6)dioxacyclododecino(2,3,4-gh)pyrrolizine-2,7-dione
58. (3z,6r,14ar,14br)-3-ethylidene-6-hydroxy-6-methyl-5-methylene-3,4,5,6,9,11,13,14,14a,14b-decahydro[1,6]dioxacyclododecino[2,3,4-gh]pyrrolizine-2,7-dione
59. (6r,9a1r,14ar,z)-3-ethylidene-6-hydroxy-6-methyl-5-methylene-3,4,5,6,9,9a1,11,13,14,14a-decahydro-[1,6]dioxacyclododecino[2,3,4-gh]pyrrolizine-2,7-dione
60. Pyrrolizine-2, 3-ethylidene-3,4,5,6,9,11,13,14,14a,14b-decahydro-6-hydroxy-6-methyl-5-methylene(1,6)dioxacyclododecino[2,3,4-gh]-
| Molecular Weight | 333.4 g/mol |
|---|---|
| Molecular Formula | C18H23NO5 |
| XLogP3 | 0.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Exact Mass | 333.15762283 g/mol |
| Monoisotopic Mass | 333.15762283 g/mol |
| Topological Polar Surface Area | 76.1 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 650 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
In animal studies highest concentrations were found in the liver, lungs, kidneys and spleen. /pyrrolizidine alkaloids/
IPCS; Poisons Information Monograph (484) on Senecio Vulgaris L. (September 1989). Available from, as of September 15, 2003: https://www.inchem.org/documents/pims/plant/senecio.htm
After injection, radioactivity was rapidly excreted in the urine and feces (84% or greater) within 16 hr. The liver contained over 1.5% of the dose at 16 hr. A small amount, 0.04%, of the dose was transferred into the milk in 16 hr; the majority of radioactivity was found in the skim-milk fraction, suggesting that the /pyrrolizidine alkaloids/ were transferred to the milk as water-soluble metabolites. ...The binding to calf thymus DNA and microsomal macromolecules was measured in vitro. The binding was diminished in the absence of O2 or a NADPH-generating system or by boiling the microsomes. No inhibition of the binding by /potassium cyanide/ was observed. /Pyrrolizidine alkaloids/
PMID:6179729 Eastman DF et al; Drug Metab Dispos 10(3): 236-40 (1982)
...To investigate the transfer into cow's milk, a single dose of 1 mg of (3H)seneciphylline/kg of body weight was given orally to a dairy cow. The appearance of radioactivity derived from this compound was monitored in the blood and milk. Calculated as seneciphylline, over 100 ng/mL was found in the blood during the first 18 hr. After 54 hr, 11 ng/mL was still present. Alkaloid levels were similar in milk. After 64 hr, the concentration was still at 5 ng/mL. In total 0.16% of the dose was excreted in the milk. In the liver, 40 ng/g (0.06% of the dose) was found 3 weeks after treatment. In addition to unchanged seneciphylline and retronecine, N-oxides were detected in milkas metabolites (11.2% at 27 hr).
Candrian U et al; J Agric Food Chem 39 (5): 930-933 (1991)
In general, hepatotoxic pyrrolizidine alkaloids are metabolized in rat liver to give hydrolysis products, n-oxides and dehydropyrrolizidine (pyrrolic) deriv. ... Dehydroalkaloids are highly reactive alkylating agents... /pyrrolizidine alkaloids/
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V10 336 (1976)
Dehydroretronecine...water-soluble pyrrolic metabolite of...seneciphylline...has been shown to be carcinogenic. /pyrrolizidine alkaloids/
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V10 339 (1976)
In animals, the major metabolic routes of pyrrolizidine alkaloids are: (a) hydrolysis of the ester groups; (b) N-oxidation; and (c) dehydrogenation of the pyrrolizidine nucleus to pyrrolic derivatives. Routes (a) and (b) are believed to be detoxification mechanisms. Route (c) leads to toxic metabolites. Route (a) occurs in liver and blood; routes (b) and (c) are brought about in the liver by the microsomal mixed function oxidase system. /pyrrolizidine alkaloids/
IPCS; Poisons Information Monograph (484) on Senecio Vulgaris L. (September 1989). Available from, as of September 15, 2003: https://www.inchem.org/documents/pims/plant/senecio.htm
Within a few hours, only a relatively small proportion of the administered dose remains in the body. Much of this is in the form of metabolites bound to tissue contents. A pyrrolizidine N-oxide disappeared from the serum after IV administration in animals, with initial half-lives of 3 -20 minutes. /pyrrolizidine alkaloids/
IPCS; Poisons Information Monograph (484) on Senecio Vulgaris L. (September 1989). Available from, as of September 15, 2003: https://www.inchem.org/documents/pims/plant/senecio.htm
The activation of the alkaloids by mixed-function oxidases leads to pyrrolic dehydro-alkaloids which are reactive alkylating agents. The liver necrosis results from binding of the metabolites with the liver cell. Some metabolites are released into the circulation and are believed to pass beyond the liver to the lung causing vascular lesions. The pyrrolic metabolites are cytotoxic and act on the hepatocytes and on the endothelium of blood vessels of the liver and lung. /pyrrolizidine alkaloids/
IPCS; Poisons Information Monograph (484) on Senecio Vulgaris L. (September 1989). Available from, as of September 15, 2003: https://www.inchem.org/documents/pims/plant/senecio.htm
The effect of oral administration of the pyrrolizidine alkaloids, seneciphylline... from Senecio vulgaris (Compositae) on activities of hepatic epoxide hydrase, glutathione-S-transferase, aminopyrine-N-demethylase and arylhydrocarbon hydroxylase (AHH) was investigated in microsomes of young male albino rats. Seneciphylline significantly increased the activities of epoxide hydrase and glutathione-S-transferase but caused reduction of cytochrome P-450 and related monooxygenase activities. ...Seneciphylline... could not produce any prominent in vitro effect on the hepatic drug metabolizing enzymes under study, except slight stimulation of epoxide hydrase activity by both the alkaloids and slight reduction of aminopyrine demethylase activity by senecionine.
PMID:6533413 Kakrani HK, Kalyani GA ; J Ethnopharmacol 12 (3): 271-8 (1984)
