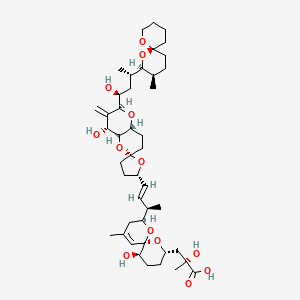

1. Ocadaic Acid
2. Okadaic Acid
1. Okadaic Acid
| Molecular Weight | 805.0 g/mol |
|---|---|
| Molecular Formula | C44H68O13 |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 10 |
| Exact Mass | 804.46599222 g/mol |
| Monoisotopic Mass | 804.46599222 g/mol |
| Topological Polar Surface Area | 183 Ų |
| Heavy Atom Count | 57 |
| Formal Charge | 0 |
| Complexity | 1520 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 17 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Carcinogens
Substances that increase the risk of NEOPLASMS in humans or animals. Both genotoxic chemicals, which affect DNA directly, and nongenotoxic chemicals, which induce neoplasms by other mechanism, are included. (See all compounds classified as Carcinogens.)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Ionophores
Chemical agents that increase the permeability of biological or artificial lipid membranes to specific ions. Most ionophores are relatively small organic molecules that act as mobile carriers within membranes or coalesce to form ion permeable channels across membranes. Many are antibiotics, and many act as uncoupling agents by short-circuiting the proton gradient across mitochondrial membranes. (See all compounds classified as Ionophores.)
The results in this study show... that this marine toxin is able to cross the transplacental barrier. Fetal tissue contains more okadaic acid than the liver or kidney: 5.60% compared to 1.90 and 2.55% respectively as measured by HPLC and fluorescent detection after derivatization with 9-Anthryldiazomethane (ADAM).
PMID:8839210 Matias W et al; Human & Experimental Toxicology 15 (3): 226-230 (1996)
This study concerns the distribution of 3H-okadaic acid (OA) in organs and biological fluids of Swiss mice having received a single dose per os of OA (50 ug/kg). The determination of the intestinal tissues and contents 24 hr after administration demonstrates a slow elimination of OA. When the dose of OA was increased from 50-90 ug/kg, the concentrations of the toxin in the intestinal content and feces increased proportionally. A good correlation was found between an increase of OA in the intestinal tissue and the diarrhea in animals given 90 ug/kg orally. Moreover OA was present in liver and bile and in all organs including skin and also fluids. Altogether these results confirmed an enterohepatic circulation of OA as previously shown.
PMID:10372758 Matias W et al; Human & Experimental Toxicology 18 (5): 345-350 (1999)
The influence of nutritional regime and water temperature on depuration rates of OA-group toxins in the wedge shell Donax trunculus was examined by exposing naturally contaminated specimens to three nutritional regimes (microalgae, commercial paste of microalgae, and starvation) for 14 days at 16 C and 20 C. Total OA was quantified in the whole soft tissues of the individuals collected in days 2, 4, 6, 8, 10, 12 and 14. Mortality, dry weight, condition index, gross biochemical composition and gametogenic stages were surveyed. Low variation of glycogen and carbohydrates during the experiments suggest that wedge shells were under non-dramatic stress conditions. Wedge shells fed with non-toxic diets showed similar depuration rates being 15 and 38% higher than in starvation, at 16 and 20 C, respectively. Depuration rates under non-toxic diets at 20 C were 71% higher than at 16 C. These results highlight the influence of water temperature on the depuration rate of total OA accumulated by D. trunculus, even when the increase is of only 4 C, as commonly observed in week time scales in the southern Portuguese coastal waters. These results open the possibility of a faster release of OA in harvested wedge shells translocated to depuration systems when under a slight increase of water temperature.
PMID:28987405 Botelho MJ et al; Chemosphere 190: 166-173 (2018)
... acidic toxins, include okadaic acid (OA) and its derivatives named dinophysistoxins (DTXs). OA and its derivatives (DTX1, DTX2 and DTX3) are lipophilic and accumulate in the fatty tissue of shellfish. These compounds are potent phosphatase inhibitors and this property is linked to inflammation of the intestinal tract and diarrhea in humans.
FAO; Marine Biotoxins (2004); Available from, as of October 9, 2018: https://www.fao.org/docrep/007/y5486e/y5486e00.htm
Okadaic acid is known as a diarrheal shellfish poison. It is thought that there is no specific target organ for okadaic acid after it has been absorbed into the body. However, the details of its pharmacokinetics are still unknown. In this study, we demonstrated that okadaic acid was more toxic to the hepatocyte-specific uptake transporter OATP1B1- or OATP1B3-expressing cells than control vector-transfected cells. In addition, PP2A activity, which is a target molecule of okadaic acid, was more potently inhibited by okadaic acid in OATP1B1- or OATP1B3-expressing cells compared with control vector-transfected cells. The cytotoxicity of okadaic acid in OATP1B1- or OATP1B3-expressing cells was attenuated by known substrates of OATP1B1- and OATP1B3, but not in control vector-transfected cells. Furthermore, after uptake inhibition study using OATP1B3-expressing cells, Dixon plot showed that okadaic acid inhibited the uptake of hepatotoxin microcystin-LR, which is a substrate for OATP1B1 and OATP1B3, in a competitive manner. These results strongly suggested that okadaic acid is a substrate for OATP1B3 and probably for OATP1B1, and could be involved in unknown caused liver failure and liver cancer. Since okadaic acid possesses cytotoxicity and cell proliferative activity by virtue of its known phosphatase inhibition activity.
PMID:26134461 Ikema S et al; Food Chem Toxicol 83: 229-36 (2015)
The ingestion of seafood contaminated with the marine biotoxin okadaic acid (OA) can lead to diarrhetic shellfish poisoning with symptoms like nausea, vomiting and abdominal cramps. Both rat and the human hepatic cytochrome P450 monooxygenases (CYP) metabolize OA. However, liver cell toxicity of metabolized OA is mainly unclear. The aim of our study was to detect the cellular effects in HepG2 cells exposed to OA in the presence of recombinant CYP enzymes of both rat and human for the investigation of species differences. The results should be set in correlation with a CYP-specific metabolite pattern. Comparative metabolite profiles of OA after incubation in rat and human recombinant CYP enzymes were established by using LC-MS/MS technique. Results demonstrated that metabolism of OA to oxygenated metabolites correlates with detoxification which was mainly catalyzed by human CYP3A4 and CYP3A5. Detoxification by rat Cyp3a1 was lower compared to human CYP3A enzymes and activation of OA by Cyp3a2 was observed, coincident with minor overall conversion capacity of OA. By contrast human and rat CYP1A2 seem to activate OA into cytotoxic intermediates. In conclusion, different mechanisms of OA metabolism may occur in the liver. At low OA doses, the human liver is likely well protected against cytotoxic OA, but for high shellfish consumers a potential risk cannot be excluded.
PMID:26374342 Kolrep F et al; Arch Toxicol 90 (8): 2025-36 (2016)
Four metabolites of okadaic acid were generated by incubation with human recombinant cytochrome P450 3A4. The structures of two of the four metabolites have been determined by MS/MS experiments and 1D and 2D NMR methods using 94 and 133 ug of each metabolite. The structure of a third metabolite was determined by oxidation to a metabolite of known structure. Like okadaic acid, the metabolites are inhibitors of protein phosphatase PP2A. Although one of the metabolites does have an alpha,beta unsaturated carbonyl with the potential to form adducts with an active site cysteine, all of the metabolites are reversible inhibitors of PP2A.
PMID:22608922 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3601775 Liu L et al; Bioorg Med Chem 20 (12): 3742-5 (2012)
Microcystins, potent heptapeptide hepatotoxins produced by certain bloom-forming cyanobacteria, are strong protein phosphatase inhibitors. They covalently bind the serine/threonine protein phosphatases 1 and 2A (PP1 and PP2A), thereby influencing regulation of cellular protein phosphorylation. The paralytic shellfish poison, okadaic acid, is also a potent inhibitor of these PPs. Inhibition of PP1 and PP2A has a dualistic effect on cells exposed to okadaic acid or microcystin-LR, with both apoptosis and increased cellular proliferation being reported.
Gehringer M et al; FEBS Lett 557 (1-3): 1-8 2004
