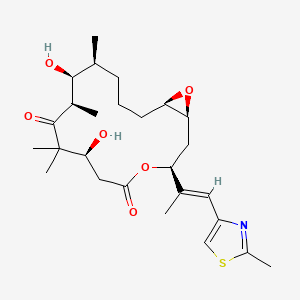

1. Epothilon A
1. 152044-53-6
2. (-)-epothilone A
3. Epo A
4. Epothilon A
5. Epothilone A [mi]
6. Epoa
7. Epothilone-a
8. Chebi:31549
9. 51e07ybx96
10. (1r,5s,6s,7r,10s,14s,16s)-6,10-dihydroxy-5,7,9,9-tetramethyl-14-[(e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-13,17-dioxabicyclo[14.1.0]heptadecane-8,12-dione
11. 4,17-dioxabicyclo(14.1.0)heptadecane-5,9-dione, 7,11-dihydroxy-8,8,10,12-tetramethyl-3-((1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl)-, (1s,3s,7s,10r,11s,12s,16r)-
12. Epothile
13. Epothilones
14. Epothilonea
15. Unii-51e07ybx96
16. Schembl4775
17. Epothilone A [who-dd]
18. Chembl329993
19. Chebi:94722
20. Dtxsid10332288
21. Ex-a2056
22. Zinc3951738
23. Lmpk04000040
24. Nsc684362
25. S1297
26. Akos025401597
27. Bcp9000654
28. Ccg-269643
29. Nsc-684362
30. Ncgc00346505-02
31. (1s,3s,7s,10r,11s,12s,16r)-7,11-dihydroxy-8,8,10,12-tetramethyl-3-((e)-1-(2-methylthiazol-4-yl)prop-1-en-2-yl)-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione
32. Ac-22615
33. Hy-13503
34. Cs-0007126
35. E78311
36. J-008878
37. Brd-k71823332-001-01-1
38. Brd-k71823332-001-03-7
39. Q27104197
40. (-)-epothilone A, From Sorangium Cellulosum, >95% (hplc), Solid
41. (1r,5s,6s,7r,10s,14s,16s)-6,10-dihydroxy-5,7,9, 9-tetramethyl-14-[(e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-13, 17-dioxabicyclo[14.1.0]heptadecane-8,12-dione
42. (1s,3s,7s,10r,11s,12s,16r)-7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(1e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione
43. (1s,3s,7s,10r,11s,12s,16r)-7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(1e)-1-methyl-2-(2-methyl-4-thiazolyl)ethenyl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione
44. (1s,3s,7s,10r,11s,12s,16r)-7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(e)-1-(2-methyl-1,3-thiazol-4-yl)prop-1-en-2-yl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione
45. (1s,3s,7s,10r,11s,12s,16r)-7,11-dihydroxy-8,8,10,12-tetramethyl-3-[(e)-1-methyl-2-(2-methyl-1,3-thiazol-4-yl)ethenyl]-4,17-dioxabicyclo[14.1.0]heptadecane-5,9-dione
46. (1s,7s,10r,11s,12s,16r)-7-hydroxy-11-(s)-hydroxy-8,8,10,12-tetramethyl-3-[(e)-1-methyl-2-(2-methyl-thiazol-4-yl)-vinyl]-4,17-dioxa-bicyclo[14.1.0]heptadecane-5,9-dione
| Molecular Weight | 493.7 g/mol |
|---|---|
| Molecular Formula | C26H39NO6S |
| XLogP3 | 4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 2 |
| Exact Mass | 493.24980914 g/mol |
| Monoisotopic Mass | 493.24980914 g/mol |
| Topological Polar Surface Area | 138 Ų |
| Heavy Atom Count | 34 |
| Formal Charge | 0 |
| Complexity | 770 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 7 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Tubulin Modulators
Agents that interact with TUBULIN to inhibit or promote polymerization of MICROTUBULES. (See all compounds classified as Tubulin Modulators.)
