
1. Echinomycin
2. Nsc 526417
3. Nsc-526417
4. Nsc526417
1. Echinomycin
2. Tg824j6rqt
3. 512-64-1
4. S-426-s (lepetit)
5. Nsc-526417
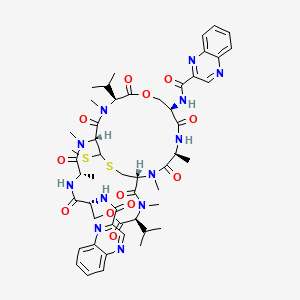
6. N,n'-((1r,4s,7r,11s,14r,17s,20r,24s)-11,24-diisopropyl-2,4,12,15,17,25-hexamethyl-27-(methylthio)-3,6,10,13,16,19,23,26-octaoxo-9,22-dioxa-28-thia-2,5,12,15,18,25-hexaazabicyclo[12.12.3]nonacosane-7,20-diyl)bis(quinoxaline-2-carboxamide)
7. Unii-tg824j6rqt
8. Nsc-13502
9. Sk 302b
10. Nsc 526417
11. Brn 0078671
12. Echinomycin [mi]
13. Antibiotic A-654i
14. 4-27-00-09726 (beilstein Handbook Reference)
15. Chembl503868
16. Schembl13464701
17. Sk-302b
18. Db15582
19. Stereoisomer Of N,n'-(2,4,12,15,17,25-hexamethyl-11,24-bis(1-methylethyl)-27-(methylthio)-3,6,10,13,16,19,23,26-octaoxo-9,22-dioxa-28-thia-2,5,12,15,18,25-hexaazabicyclo(12.12.3)nonacosane-7,20-diyl)bis(2-quinoxalinecarboxamide)
20. Hy-106101
21. Cs-0024860
22. L-valine, N-(2-quinoxalinylcarbonyl)-o-(n-(2-quinoxalinylcarbonyl)-d-seryl-l-alanyl-3-mercapto-n,s-dimethylcysteinyl-n-methyl-l-valyl)-d-seryl-l-alanyl-n-methylcysteinyl-n-methyl-, (8->1)-lactone, Cyclic (3->7)-thioether
23. N-(2-quinoxalinylcarbonyl)-o-(n-(2-quinoxalinylcarbonyl)-d-seryl-l-alanyl-3-mercapto-n,s-dimethylcysteinyl-n-methyl-l-valyl)-d-seryl-l-alanyl-n-methylcysteinyl-n-methyl L-valine (8->1)-lactone Cyclic (3->7)-thioester
| Molecular Weight | 1101.3 g/mol |
|---|---|
| Molecular Formula | C51H64N12O12S2 |
| XLogP3 | 2.7 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 7 |
| Exact Mass | 1100.42080787 g/mol |
| Monoisotopic Mass | 1100.42080787 g/mol |
| Topological Polar Surface Area | 352 Ų |
| Heavy Atom Count | 77 |
| Formal Charge | 0 |
| Complexity | 2200 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 8 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
Antibiotics, Antineoplastic
Chemical substances, produced by microorganisms, inhibiting or preventing the proliferation of neoplasms. (See all compounds classified as Antibiotics, Antineoplastic.)
Nucleic Acid Synthesis Inhibitors
Compounds that inhibit cell production of DNA or RNA. (See all compounds classified as Nucleic Acid Synthesis Inhibitors.)
