


1. Versamine
2. Mecamine
3. Revertina
4. Mecamilamina
5. Mekamine
6. 60-40-2
7. 2-methylaminoisocamphane
8. Mevasine
9. 3-methylaminoisocamphane
10. N,2,3,3-tetramethylbicyclo[2.2.1]heptan-2-amine
11. N-methyl-2-isocamphanamine
12. 2-methylamino-2,3,3-trimethylnorbornane
13. N,2,3,3-tetramethyl-2-norbornamine
14. N,2,3,3-tetramethyl-2-norcamphanamine
15. 2-norbornanamine, N,2,3,3-tetramethyl-
16. Bicyclo(2.2.1)heptan-2-amine, N,2,3,3-tetramethyl-
17. Bicyclo[2.2.1]heptan-2-amine, N,2,3,3-tetramethyl-
18. Chebi:6706
19. Chembl267936
20. Mecamylaminum
21. Ncgc00015703-03
22. Mecamilamina [dcit]
23. Mecamilamina [italian]
24. Dsstox_cid_3240
25. Dsstox_rid_76937
26. Dsstox_gsid_23240
27. Mecamylamine [inn:ban]
28. Mecamylaminum [inn-latin]
29. 3-methylaminoisokamfan
30. 3-methylaminoisokamfan [czech]
31. Cas-60-40-2
32. Einecs 200-476-1
33. 3-beta-methylamino-2,2,3-trimethylbicyclo(2.2.1)heptane
34. Inversine (salt/mix)
35. Spectrum_000304
36. Specplus_000736
37. Prestwick0_001111
38. Prestwick1_001111
39. Prestwick2_001111
40. Prestwick3_001111
41. Spectrum3_000736
42. Spectrum4_001257
43. Spectrum5_001616
44. Lopac0_000841
45. Schembl34252
46. Bspbio_001242
47. Bspbio_002292
48. Kbiogr_001814
49. Kbioss_000784
50. Divk1c_006832
51. Spbio_003111
52. Bpbio1_001367
53. Gtpl3990
54. Dtxsid0023240
55. Hy-b1395a
56. Kbio1_001776
57. Kbio2_000784
58. Kbio2_003352
59. Kbio2_005920
60. Kbio3_001512
61. Hms3604d12
62. 107538-05-6
63. Albb-023410
64. Tox21_110198
65. Bdbm50061565
66. Akos003662515
67. Tox21_110198_1
68. Ccg-204924
69. Db00657
70. Sdccgsbi-0050817.p005
71. Ncgc00015703-02
72. Ncgc00015703-04
73. Ncgc00015703-06
74. Ncgc00015703-12
75. Ncgc00162272-01
76. Ncgc00162272-02
77. Sbi-0050817.p004
78. Ab00053764
79. Cs-0013657
80. C07511
81. Ab00053764_03
82. Q3332124
83. Brd-a20119038-001-01-5
84. Brd-a20119038-003-01-1
85. N,2,3,3-tetramethylbicyclo[2.2.1]heptan-2-amine #
86. Methyl-(2,3,3-trimethyl-bicyclo[2.2.1]hept-2-yl)-amine
87. (+)methyl-(2,3,3-trimethyl-bicyclo[2.2.1]hept-2-yl)-amine
88. (-)methyl-(2,3,3-trimethyl-bicyclo[2.2.1]hept-2-yl)-amine
89. 3-.beta.-methylamino-2,2,3-trimethylbicyclo(2.2.1)heptane
90. (+/-)methyl-(2,3,3-trimethyl-bicyclo[2.2.1]hept-2-yl)-amine
91. Methyl(2,3,3-trimethylbicyclo[2.2.1]hept-2-yl)amine Hydrochloride
92. Methyl-(2,3,3-trimethyl-bicyclo[2.2.1]hept-2-yl)-amine(mecamylamine)
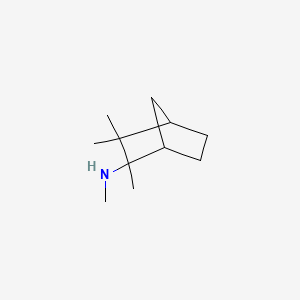
| Molecular Weight | 167.29 g/mol |
|---|---|
| Molecular Formula | C11H21N |
| XLogP3 | 2.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Exact Mass | 167.167399674 g/mol |
| Monoisotopic Mass | 167.167399674 g/mol |
| Topological Polar Surface Area | 12 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 197 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 3 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For the treatment of moderately severe to severe essential hypertension and in uncomplicated cases of malignant hypertension
Mecamylamine is a potent, oral antihypertensive agent and ganglion blocker, and is a secondary amine. Mecamylamine is indicated for the management of moderately severe to severe essential hypertension and in uncomplicated cases of malignant hypertension. Mecamylamine reduces blood pressure in both normotensive and hypertensive individuals. A small oral dosage often produces a smooth and predictable reduction of blood pressure. Although this antihypertensive effect is predominantly orthostatic, the supine blood pressure is also significantly reduced. Mecamylamine crosses the blood-brain and placental barriers.
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
Ganglionic Blockers
Agents having as their major action the interruption of neural transmission at nicotinic receptors on postganglionic autonomic neurons. Because their actions are so broad, including blocking of sympathetic and parasympathetic systems, their therapeutic use has been largely supplanted by more specific drugs. They may still be used in the control of blood pressure in patients with acute dissecting aortic aneurysm and for the induction of hypotension in surgery. (See all compounds classified as Ganglionic Blockers.)
Nicotinic Antagonists
Drugs that bind to nicotinic cholinergic receptors (RECEPTORS, NICOTINIC) and block the actions of acetylcholine or cholinergic agonists. Nicotinic antagonists block synaptic transmission at autonomic ganglia, the skeletal neuromuscular junction, and at central nervous system nicotinic synapses. (See all compounds classified as Nicotinic Antagonists.)
C - Cardiovascular system
C02 - Antihypertensives
C02B - Antiadrenergic agents, ganglion-blocking
C02BB - Secondary and tertiary amines
C02BB01 - Mecamylamine
Absorption
Mecamylamine is almost completely absorbed from the gastrointestinal tract
Route of Elimination
Mecamylamine is excreted slowly in the urine in the unchanged form. The rate of its renal elimination is influenced markedly by urinary pH. Alkalinization of the urine reduces, and acidification promotes, renal excretion of mecamylamine. Mecamylamine crosses the blood-brain and placental barriers.
Mecamylamine is a ganglionic blocker which prevents stimulation of postsynaptic receptors by acetylcholine released from presynaptic nerve endings. The hypotensive effect of Mecamylamine is attributed to reduction in sympathetic tone, vasodilation, and reduced cardiac output, and is primarily postural.
