

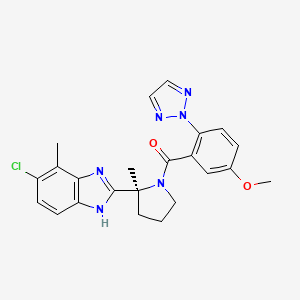

1. ((s)-(2-(5-chloro-4-methyl-1h-benzo(d)imidazol-2-yl)-2-methylpyrrolidin-1-yl) (5 Methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone)
2. Methanone, ((2s)-2-(6-chloro-7-methyl-1h-benzimidazol-2-yl)-2-methyl-1-pyrrolidinyl)(5-methoxy-2-(2h-1,2,3-traizol-2-yl)phenyl)-
3. Nemorexant
4. Act-541468
5. Quviviq
1. Nemorexant
2. 1505484-82-1
3. Act-541468
4. Quviviq
5. Lmq24g57e9
6. Dea No. 2410
7. Dtxsid701336356
8. [(2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl]-[5-methoxy-2-(triazol-2-yl)phenyl]methanone
9. Methanone, ((2s)-2-(6-chloro-7-methyl-1h-benzimidazol-2-yl)-2-methyl-1-pyrrolidinyl)(5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)-
10. Daridorexantum
11. ((2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl)-(5-methoxy-2-(triazol-2-yl)phenyl)methanone
12. Refchem:56310
13. Dtxcid901766549
14. ((s)-(2-(5-chloro-4-methyl-1h-benzo(d)imidazol-2-yl)-2-methylpyrrolidin-1-yl) (5 Methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone)
15. 5-chloro-2-((2s)-1-(5-methoxy-2-(2h-1,2,3-triazol-2-yl)benzoyl)-2-methylpyrrolidin-2-yl)-4-methyl-1h-1,3-benzodiazole
16. Methanone, ((2s)-2-(6-chloro-7-methyl-1h-benzimidazol-2-yl)-2-methyl-1-pyrrolidinyl)(5-methoxy-2-(2h-1,2,3-traizol-2-yl)phenyl)-
17. Daridorexant [usan]
18. Act541468
19. Mfcd31630772
20. Act 541468;act-541468
21. (s)-[2-(6-chloro-7-methyl-2-benzimidazolyl)-2-methyl-1-pyrrolidinyl][5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl]methanone
22. [(2~{s})-2-(6-chloranyl-7-methyl-1~{h}-benzimidazol-2-yl)-2-methyl-pyrrolidin-1-yl]-[5-methoxy-2-(1,2,3-triazol-2-yl)phenyl]methanone
23. Nemorexant?
24. Daridorexant Free Base
25. Nemorexant (former Name)
26. Daridorexant [inn]
27. Unii-lmq24g57e9
28. Nemorexant (discontinued)
29. Daridorexant [who-dd]
30. Orb1473166
31. Chembl4297590
32. Schembl16766318
33. Schembl16778804
34. Schembl30028832
35. Ex-a7813a
36. Gtpl11648
37. Bdbm334973
38. Glxc-15297
39. Fkc48482
40. Who 10679
41. Us9732075, Example 5.36
42. Akos040742277
43. Db15031
44. [(2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl][5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl]methanone
45. Da-66016
46. Sy251586
47. Hy-109095
48. Cs-0039396
49. (2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl)(5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
50. (s)-(2-(5-chloro-4-methyl-1h-benzo[d]imidazol-2-yl)-2-methylpyrrolidin-1-yl)(5 Methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
51. (s)-(2-(6-chloro-7-methyl-1h-benzo[d]imidazol-2-yl)-2-methylpyrrolidin-1-yl)(5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
52. (s)-2-(5-chloro-4-methyl-1h Benzo[d]imidazol-2-yl)-2-methylpyrrolidin-1-yl](5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
53. [(2s)-2-(5-chloro-4-methyl-1h-benzimidazol-2-yl)-2-methyl-pyrrolidin-1-yl]-[5-methoxy-2-(triazol-2-yl)phenyl]methanone
54. [(s)-2-(5-chloro-4-methyl-1h-benzoimidazol-2-yl)-2-methyl-pyrrolidin-1-yl]-(5-methoxy-2-[1,2,3]triazol-2-yl-phenyl)-methanone
55. Daridorexant; (s)-(2-(6-chloro-7-methyl-1h-benzo[d]imidazol-2-yl)-2-methylpyrrolidin-1-yl)(5-methoxy-2-(2h-1,2,3-triazol-2-yl)phenyl)methanone
| Molecular Weight | 450.9 g/mol |
|---|---|
| Molecular Formula | C23H23ClN6O2 |
| XLogP3 | 4.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 88.9 |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 693 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
In the US and Europe, daridorexant is indicated for the treatment of adult patients with insomnia characterized by difficulties with sleep onset and/or sleep maintenance. The European prescribing information states that insomnia should be characterized by symptoms that are present for at least three months and have a considerable impact on daytime functioning.
N - Nervous system
N05 - Psycholeptics
N05C - Hypnotics and sedatives
N05CJ - Orexin receptor antagonists
N05CJ03 - Daridorexant
ATCvet Code
QN - Nervous system
QN05 - Psycholeptics
QN05C - Hypnotics and sedatives
QN05CJ - Orexin receptor antagonists
QN05CJ03 - Daridorexant
Absorption
Daridorexant reaches peak plasma concentrations within one to two hours. Daridorexant has an absolute bioavailability of 62%. While a high-fat and high-calorie meal delayed the Tmax by 1.3 hours and decreased the Cmax by 16% in healthy subjects, the total exposure (AUC) was not affected.
Route of Elimination
The primary route of excretion is via feces, accounting for approximately 57% of drug excretion. About 28% of the drug is excreted via urine primarily in the form of metabolites. Trace amounts of the parent drug were found in feces and urine.
Volume of Distribution
Daridorexant has a volume of distribution of 31 L. The blood to plasma ratio is 0.64. It effectively passes the blood-brain barrier.
Clearance
There is limited information on clearance.
Daridorexant undergoes extensive metabolism primarily mediated by CYP3A4 (89%), mostly via oxidative transformations. Other CYP enzymes individually contribute to less than 3% of metabolic clearance of daridorexant.
The terminal half-life is approximately 8 hours.
The sleep and wake cycle is regulated by complex interactions between sleep-promoting systems, including inhibitory GABA activity, and wake-promoting systems, including orexins, acetylcholine and monoaminergic systems. Orexin, also called hypocretin, is a wake-promoting neuropeptide produced by a small group of neurons in the lateral hypothalamus. Orexin stabilizes wakefulness by activating orexin neurons with the highest activity during active wakefulness and minimal activity during sleep. Orexin neurons project to other wake-promoting neurons that also express orexin receptors: these include the histaminergic neurons of the tuberomammillary nucleus, noradrenergic neurons of the locus coeruleus, serotoninergic neurons of the dorsal raphe, dopaminergic neurons of the ventral tegmental area, and cholinergic neurons of the basal forebrain and the pedunculopontine and laterodorsal tegmental nuclei. These wake-promoting neurons are part of the ascending reticular activating system that operates under a feedback loop in the sleep and wake cycle. There are two identified types of orexin (OXA and OXB) that bind to orexin type 1 and 2 receptors (OX1R and OX2R), which are G-protein coupled receptors. OXA binds more preferentially to OX1R, while OX2R shows a dual affinity for OXA and OXB. The defined role of each orexin receptor is still unclear; however, there is some evidence suggesting that OX2R regulates sleep and wake, while OX1R have some role in sleep maintenance. Daridorexant blocks the binding of wake-promoting neuropeptides OXA and OXB to OX1R and OX2R, thereby suppressing wake drive. Daridorexant selectively targets orexin neurons and inhibits downstream neuronal pathways that promote wakefulness; however, it does not affect neuronal pathways that cause side effects commonly seen in positive allosteric GABA-A receptor modulators.
