

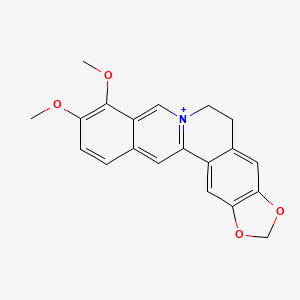

1. Umbellatine
1. 2086-83-1
2. Umbellatine
3. Berberin
4. Berbericine
5. Majarine
6. Thalsine
7. Umbellatin
8. Berberone
9. Benzo[g]-1,3-benzodioxolo[5,6-a]quinolizinium, 5,6-dihydro-9,10-dimethoxy-
10. 0i8y3p32uf
11. Dtxsid9043857
12. Benzo(g)-1,3-benzodioxolo(5,6-a)quinolizinium, 5,6-dihydro-9,10-dimethoxy-
13. 16,17-dimethoxy-5,7-dioxa-13-azoniapentacyclo[11.8.0.02,10.04,8.015,20]henicosa-1(13),2,4(8),9,14,16,18,20-octaene
14. Chebi:16118
15. 9,10-dimethoxy-5,6-dihydro[1,3]dioxolo[4,5-g]isoquino[3,2-a]isoquinolin-7-ium
16. 5,6-dihydro-9,10-dimethoxy-1,3-benzodioxolo(5,6-a)benzo(g)quinolizinium
17. 9,10-dimethoxy-5,6-dihydro(1,3)dioxolo(4,5-g)isoquino(3,2-a)isoquinolin-7-ium
18. 16,17-dimethoxy-5,7-dioxa-13$l^(5)-azapentacyclo(11.8.0.0^(2,10).0^(4,8).0^(15,20))henicosa-1(13),2,4(8),9,14,16,18,20-octaen-13-ylium
19. 16,17-dimethoxy-5,7-dioxa-13$l^{5}-azapentacyclo[11.8.0.0^{2,10}.0^{4,8}.0^{15,20}]henicosa-1(13),2,4(8),9,14,16,18,20-octaen-13-ylium
20. 16,17-dimethoxy-5,7-dioxa-13-azoniapentacyclo(11.8.0.02,10.04,8.015,20)henicosa-1(13),2,4(8),9,14,16,18,20-octaene
21. Refchem:5599
22. Dtxcid7023857
23. 218-229-1
24. 9,10-dimethoxy-5,6-dihydro-[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinolin-7-ium
25. Mfcd01175817
26. Berberal
27. St055798
28. 9,10-dimethoxy-2,3-(methylenedioxy)-7,8,13,13a-tetrahydroberbinium
29. 5,6-dihydro-9,10-dimethoxybenzo[g]-1,3-benzodioxolo[5,6-a]quinolizinium
30. Chembl12089
31. Gnf-pf-4545
32. Ber
33. Nsc646666
34. Ncgc00016526-02
35. Ncgc00016526-07
36. Cas-633-65-8
37. Berbinium
38. Berberine Dimer
39. Coptis Rhizome
40. Berberinechloride
41. C20h18no4
42. 5,6-dihydro-9,10-dimethoxybenzo(g)-1,3-benzodioxolo(5,6-a)quinolizinium
43. Einecs 218-229-1
44. Umbellatine (6ci)
45. Brn 3570374
46. Spectrum_001110
47. Berbinium, 7,8,13,13a-tetradehydro-9,10-dimethoxy-2,3-(methylenedioxy)-, Chloride
48. Berberine [mi]
49. Prestwick0_000586
50. Prestwick1_000586
51. Prestwick2_000586
52. Prestwick3_000586
53. Spectrum2_000894
54. Spectrum3_000618
55. Spectrum4_000785
56. Spectrum5_001458
57. Berberine [vandf]
58. 7,8,13,13a-tetradehydro-9,10-dimethoxy-2,3-(methylenedioxy)berbinium
59. Berberine [mart.]
60. Upcmld-dp032
61. Ncimech_000354
62. Berberine [who-dd]
63. Unii-0i8y3p32uf
64. Schembl25632
65. Bspbio_000432
66. Bspbio_002156
67. Kbiogr_001230
68. Kbioss_001590
69. Cid_12456
70. Divk1c_000265
71. Inverted Exclamation Marky97%
72. Spbio_000708
73. Spbio_002651
74. Bpbio1_000476
75. Chembl295124
76. Megxp0_001923
77. Orb1302341
78. Schembl29354144
79. Schembl30153843
80. Upcmld-dp032:001
81. Acon1_001957
82. Bcbcmap01_000112
83. Gtpl11353
84. Kbio1_000265
85. Kbio2_001590
86. Kbio2_004158
87. Kbio2_006726
88. Kbio3_001656
89. Ninds_000265
90. 34md1011dm
91. Hms3561d13
92. Hms5086k15
93. Hy-n0716
94. Ac-117
95. Bbl029198
96. Bdbm50203126
97. Ccg-35898
98. S9046
99. Stk870320
100. 5,6-dihydro-9,10-dimethoxy-benzo[g]-1,3-benzodioxolo[5,6-a]quinolizinium
101. Akos002141363
102. Db04115
103. Sdccgmls-0066718.p001
104. 9,10-dimethoxy-5,6-dihydro-[1,3]dioxolo-[4,5-g]isoquinolino[3,2-a]isoquinolin-7-ium
105. Berbinium, 7,8,13,13a-tetrahydro-9,10-dimethoxy-2,3-(methylenedioxy)-
106. Idi1_000265
107. Smp1_000298
108. Ncgc00016526-01
109. Ncgc00016526-03
110. Ncgc00016526-04
111. Ncgc00016526-05
112. Ncgc00016526-06
113. Ncgc00016526-08
114. Ncgc00016526-11
115. Ncgc00016526-13
116. Ncgc00016526-19
117. Ncgc00091896-03
118. Nci60_001050
119. Nci60_001224
120. Nci60_004319
121. Sy232626
122. Sbi-0051613.p002
123. Db-050153
124. Cs-0009734
125. Ns00009669
126. C00757
127. 086b831
128. 2,3-methylenedioxy-9,10-dimethoxy-protoberberine
129. Q176525
130. Berberine (constituent Of Goldenseal) [dsc]
131. Sr-01000711827-5
132. Brd-k14796088-003-06-0
133. Brd-k14796088-003-17-7
134. Brd-k14796088-003-25-0
135. Brd-k14796088-003-26-8
136. 7,8,13,13a-tetradehydro-9,10-dimethoxy-2,3-[methylenebis(oxy)]berbinium
137. 9,10-dimethoxy-2,3-(methylenedioxy)-7,8,13,13a-tetradehydroberbinium
138. Berbinium, 7,8,13,13a-tetradehydro-9,10-dimethoxy-2,3-(methylenedioxy)-
139. 3,4-dimethoxy-6,7-dihydro-[1,3]dioxolo[4,5-g]pyrido[2,1-a]isoquinolin-5-ylium
140. 9,10-dimethoxy-5,6-dihydro-[1,3]dioxolo[4,5-g]isoquino[3,2-a]isoquinolin-7-ylium
141. 9,10-dimethoxy-5,6-dihydro-2h-1,3-dioxoleno[4,5-g]isoquinolino[3,2-a]isoquinol Ine
142. 9,10-dimethoxy-5,6-dihydro-7lambda~5~-[1,3]dioxolo[4,5-g]isoquino[3,2-a]isoquinoline
143. Benzo[g]-1,3-benzodioxolo[5,6-a]quinolizinium, 5,6-dihydro-9,10-dimethoxy- (9ci)
144. 9,10-dimethoxy-5,6-dihydro-[1,3]dioxolo[4,5-g]isoquino[3,2-a]isoquinolin-7-ylium Chloride
145. 9,10-dimethoxy-5,6-dihydro-[1,3]dioxolo[4,5-g]isoquino[3,2-a]isoquinolin-7-ylium; Chloride
146. Inchi=1/c20h18no4/c1-22-17-4-3-12-7-16-14-9-19-18(24-11-25-19)8-13(14)5-6-21(16)10-15(12)20(17)23-2/h3-4,7-10h,5-6,11h2,1-2h3/q+
| Molecular Weight | 336.4 g/mol |
|---|---|
| Molecular Formula | C20H18NO4+ |
| XLogP3 | 3.6 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 40.8 |
| Heavy Atom Count | 25 |
| Formal Charge | 1 |
| Complexity | 488 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
This publication noted that berberine is an alkaloid from Hydrastis canadensis L., Chinese herb Huanglian, and many other plants. It is widely used in traditional Chinese medicine as an antimicrobial in the treatment of dysentery and infectious diarrhea. The authors describe the cardiovascular effects of berberine and its derivatives, tetrahydroberberine and 8-oxoberberine, e.g., berberine has positive inotropic, negative chronotropic, antiarrhythmic, and vasodilator properties. Some cardiovascular effects of berberine and its derivatives are attributed to the blockade of K+ channels (delayed rectifier and K(ATP)) and stimulation of Na+ -Ca(2+) exchanger. Also, berberine prolongs the duration of ventricular action potential. Its vasodilator activity has been attributed to multiple cellular mechanisms. The authors note that the cardiovascular effects of berberine suggest its possible clinical usefulness in the treatment of arrhythmias and/or heart failure.
doi:10.1111/j.1527-3466.2001.tb00068.x
Berberine, an isoquinoline alkaloid isolated from the Chinese herb Coptis chinensis and other Berberis plants, has a wide range of pharmacological properties. This publication notes the mechanisms of berberine's functions have been extensively explored, and that berberine: 1) can be used to treat many diseases, such as cancer and digestive, metabolic, cardiovascular, and neurological diseases, 2) has protective capacities in digestive diseases, 3) can inhibit toxins and bacteria, including Helicobacter pylori, protect the intestinal epithelial barrier from injury, and ameliorate liver injury, 4) inhibits the proliferation of various types of cancer cells and impedes invasion and metastasis, 5) improves the efficacy and safety of chemoradiotherapies, 6) regulates glycometabolism and lipid metabolism, improves energy expenditure, reduces body weight, and alleviates nonalcoholic fatty liver disease, 7) improves cardiovascular hemodynamics, suppresses ischemic arrhythmias, attenuates the development of atherosclerosis, and reduces hypertension, 8) shows potent neuroprotective effects, including antioxidative, antiapoptotic, and anti-ischemic, and 9) exerts protective effects against other diseases.
doi:10.1007/s11684-019-0724-6
