10 Oct 2023
// PRESS RELEASE
05 Jun 2026
// PR NEWSWIRE
05 Jun 2026
// ECONOMICTIMES
Latest Content by PharmaCompass

About
Industry Trade Show
Booth #3C1
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Industry Trade Show
Not Confirmed
03-05 June, 2026
CONTACT DETAILS






Events
Webinars & Exhibitions
Industry Trade Show
Booth #3C1
06-08 October, 2026
PEGS Boston SummitPEGS Boston Summit
Industry Trade Show
Not Confirmed
11 May-15 November, 2026
Industry Trade Show
Not Confirmed
03-05 June, 2026
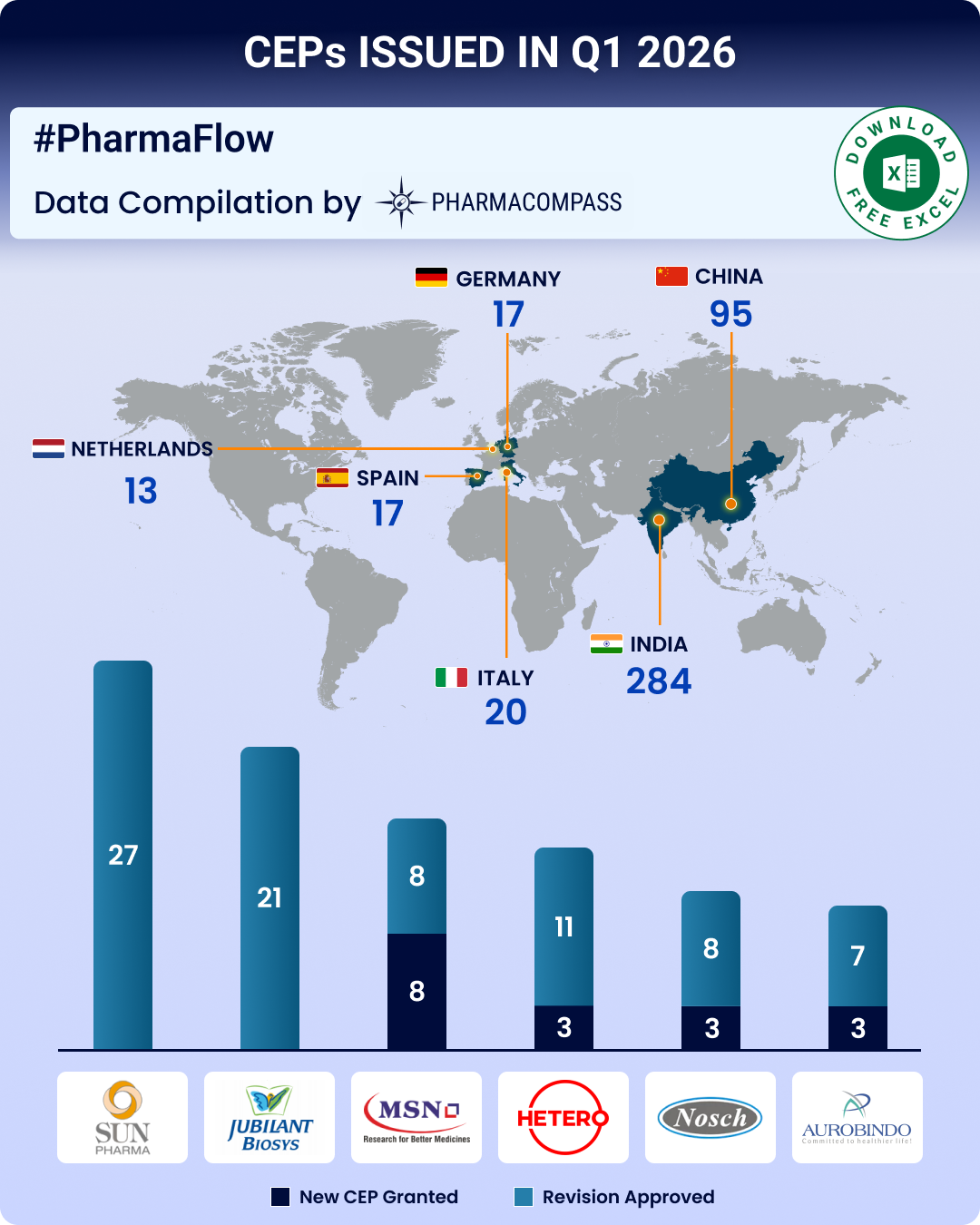
https://www.pharmacompass.com/radio-compass-blog/cep-q1-2026-update-cep-2-0-edqm-s-new-guidelines-strengthen-ecosystem-indian-firms-top-list-of-ceps-issued
https://www.pharmacompass.com/radio-compass-blog/dmf-filings-rise-4-5-in-q3-2025-china-holds-lead-india-records-20-growth-in-submissions
https://www.pharmacompass.com/radio-compass-blog/cdmo-activity-tracker-veranova-carbogen-lead-adc-investments-axplora-polfa-tarchomin-famar-expand-european-footprint

https://www.axplora.com/farmabios-an-axplora-company-granted-cgmp-approval-from-aifa-for-extended-production-capacity-in-hpapis-and-steroids/
Inspections and registrations
Country : Italy
City/Region : Gropello Cairoli
Audit Date : 2026-02-18
Audit Type : On-Site
Country : Italy
City/Region : Gropello Cairoli
Audit Date : 2025-05-15
Audit Type : On-Site
Country : Italy
City/Region : Gropello Cairoli
Audit Date : 2024-06-12
Audit Type : On-Site
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE
Farmabios SPA is a supplier offers 4 products (APIs, Excipients or Intermediates).
Find Prednisolone bulk with DMF offered by Farmabios SPA
Find Testosterone Cypionate bulk with DMF offered by Farmabios SPA
Find TRIAMCINOLONE HEXACETONIDE MICRONIZED STERILE bulk with DMF offered by Farmabios SPA
Find DEXAMETHASONE MICRONIZED STERILE bulk with DMF offered by Farmabios SPA


 Farmabios SPA
Farmabios SPA