
Synopsis
Synopsis
0
VMF
0
Australia
Annual Reports
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
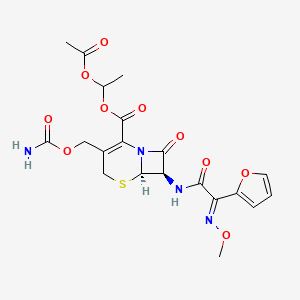

1. Acetoxyethylcefuroxime
2. Cci 15641
3. Cci-15641
4. Ceftin
5. Cefurax
6. Cefuroxime 1-acetoxyethyl Ester
7. Cepazine
8. Cetoxil
9. Curocef
10. Elobact
11. Novador
12. Novocef
13. Oraxim
14. Zinat
15. Zinnat
1. 97232-96-7
2. Cefuroxime Axetil, (e)-
3. 64544-07-6
4. (+/-)-cefuroxime Axetil, (e)-
5. Xsl6i3sb26
6. Ceftin
7. Cefuroxime Axetil E-isomers
8. 1-acetyloxyethyl (6r,7r)-3-(carbamoyloxymethyl)-7-[[(2e)-2-(furan-2-yl)-2-methoxyiminoacetyl]amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate
9. (1rs)-1-(acetyloxy)ethyl (6r,7r)-3-((carbamoyloxy)methyl)-7-(((e)-2-(furan-2-yl)-2-(methoxyimino)acetyl)amino)-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2carboxylate
10. 5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic Acid, 3-(((aminocarbonyl)oxy)methyl)-7-(((2e)-2-(2-furanyl)-2-(methoxyimino)acetyl)amino)-8-oxo-, 1-(acetyloxy)ethyl Ester, (6r,7r)-
11. Unii-xsl6i3sb26
12. Cefuroxime Axetil Impurity B [ep]
13. Cefuroximeaxetil
14. Cefuroxime Axetil Specified Impurity B [ep]
15. Schembl41099
16. Schembl379336
17. Dtxsid20242788
18. Mfcd00864991
19. Akos037643328
20. Ncgc00185751-06
21. 5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic Acid, 3-(((aminocarbonyl)oxy)methyl)-7-((2-furanyl(methoxyimino)acetyl)amino)-8-oxo-, 1-(acetyloxy)ethyl Ester, (6r-(6alpha,7beta(e)))-
22. Cefuroxime Axetil E-isomers [usp-rs]
23. Cefuroxime Axetil Impurity B [ep Impurity]
24. Q3604308
25. 1-(acetyloxy)ethyl (6r,7r)-3-{[(aminocarbonyl)oxy]methyl}-7-({(2e)-2-furan-2-yl-2-[(methyloxy)imino]acetyl}amino)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate
26. 1-acetoxyethyl (6r,7r)-3-carbamoyloxymethyl-7-[(z)-2-(fur-2-yl)-2-methoxyiminoacetamido]ceph-3-em-4-carboxylate
27. 5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic Acid, 3-(((aminocarbonyl)oxy)methyl)-7-((2-furanyl(methoxyimino)acetyl)amino)-8-oxo-, 1-(acetyloxy)ethyl Ester, (6r-(6.alpha.,7.beta.(e)))-
28. 5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid, 3-[[(aminocarbonyl)oxy]methyl]-7-[[(2z)-2-(2-furanyl)-2-(methoxyimino)acetyl]amino]-8-oxo-, 1- (acetyloxy)ethyl Ester, (6r,7r)-
| Molecular Weight | 510.5 g/mol |
|---|---|
| Molecular Formula | C20H22N4O10S |
| XLogP3 | 0.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 12 |
| Exact Mass | 510.10566408 g/mol |
| Monoisotopic Mass | 510.10566408 g/mol |
| Topological Polar Surface Area | 214 Ų |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 968 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Cefuroxime axetil |
| Drug Label | Cefuroxime axetil tablets USP contain cefuroxime as cefuroxime axetil. Cefuroxime axetil is a semisynthetic, broad-spectrum cephalosporin antibiotic for oral administration.Chemically, cefuroxime axetil, the 1-(acetyloxy) ethyl ester of cefuroxime, i... |
| Active Ingredient | Cefuroxime axetil |
| Dosage Form | Tablet; For suspension |
| Route | Oral |
| Strength | eq 250mg base/5ml; eq 500mg base; eq 125mg base; eq 250mg base; eq 125mg base/5ml |
| Market Status | Prescription |
| Company | Wockhardt; Ranbaxy; Teva; Apotex; Alkem Labs; Aurobindo Pharma; Lupin; Orchid Hlthcare |
| 2 of 2 | |
|---|---|
| Drug Name | Cefuroxime axetil |
| Drug Label | Cefuroxime axetil tablets USP contain cefuroxime as cefuroxime axetil. Cefuroxime axetil is a semisynthetic, broad-spectrum cephalosporin antibiotic for oral administration.Chemically, cefuroxime axetil, the 1-(acetyloxy) ethyl ester of cefuroxime, i... |
| Active Ingredient | Cefuroxime axetil |
| Dosage Form | Tablet; For suspension |
| Route | Oral |
| Strength | eq 250mg base/5ml; eq 500mg base; eq 125mg base; eq 250mg base; eq 125mg base/5ml |
| Market Status | Prescription |
| Company | Wockhardt; Ranbaxy; Teva; Apotex; Alkem Labs; Aurobindo Pharma; Lupin; Orchid Hlthcare |
Anti-Bacterial Agent
National Library of Medicine's Medical Subject Headings. Cefuroxime axetil. Online file (MeSH, 2014). Available from, as of November 19, 2013: https://www.nlm.nih.gov/mesh/2014/mesh_browser/MBrowser.html
Ceftin tablets and oral suspension /are indicated for/ acute bacterial otitis media caused by Streptococcus pneumoniae, Haemophilusinfluenzae(including beta-lactamase-producing strains), Moraxellacatarrhalis (including beta-lactamase-producing strains), or Streptococcus pyogenes. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for ICEFTIN (cefuroxime axetil) powder, for suspension CEFTIN (cefuroxime axetil) tablet, film coated (July 2011). Available from, as of November 13, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2f439b86-59d6-4350-aeb4-c904b3781db4
Ceftin tablets and oral suspension /are indicated for/ pharyngitis/tonsillitis caused by Streptococcus pyogenes. NOTE: The usual drug of choice in the treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever, is penicillin given by the intramuscular route. Ceftin Tablets are generally effective in the eradication of streptococci from the nasopharynx; however, substantial data establishing the efficacy of cefuroxime in the subsequent prevention of rheumatic fever are not available. Please also note that in all clinical trials, all isolates had to be sensitive to both penicillin and cefuroxime. There are no data from adequate and well-controlled trials to demonstrate the effectiveness of cefuroxime in the treatment of penicillin-resistant strains of Streptococcus pyogenes. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for ICEFTIN (cefuroxime axetil) powder, for suspension CEFTIN (cefuroxime axetil) tablet, film coated (July 2011). Available from, as of November 13, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2f439b86-59d6-4350-aeb4-c904b3781db4
Ceftin tablets /are indicated for/ acute bacterial maxillary sinusitis caused by Streptococcus pneumoniaeorHaemophilusinfluenzae (non-beta-lactamase-producing strains only). NOTE: In view of the insufficient numbers of isolates of beta-lactamase-producing strains of Haemophilusinfluenzae and Moraxellacatarrhalis that were obtained from clinical trials with Ceftin Tablets for patients with acute bacterial maxillary sinusitis, it was not possible to adequately evaluate the effectiveness of Ceftin Tablets for sinus infections known, suspected, or considered potentially to be caused by beta-lactamase-producing Haemophilusinfluenzae or Moraxellacatarrhalis. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for ICEFTIN (cefuroxime axetil) powder, for suspension CEFTIN (cefuroxime axetil) tablet, film coated (July 2011). Available from, as of November 13, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2f439b86-59d6-4350-aeb4-c904b3781db4
For more Therapeutic Uses (Complete) data for Cefuroxime axetil (15 total), please visit the HSDB record page.
Patients should be advised that antibacterials (including cefuroxime) should only be used to treat bacterial infections and not used to treat viral infections (e.g., the common cold). Patients also should be advised about the importance of completing the full course of therapy, even if feeling better after a few days, and that skipping doses or not completing therapy may decrease effectiveness and increase the likelihood that bacteria will develop resistance and will not be treatable with cefuroxime or other antibacterials in the future.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 71
To reduce development of drug-resistant bacteria and maintain effectiveness of cefuroxime and other antibacterials, the drug should be used only for the treatment or prevention of infections proven or strongly suspected to be caused by susceptible bacteria. When selecting or modifying anti-infective therapy, use results of culture and in vitro susceptibility testing. In the absence of such data, consider local epidemiology and susceptibility patterns when selecting anti-infectives for empiric therapy.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 71
Prior to initiation of cefuroxime therapy, careful inquiry should be made concerning previous hypersensitivity reactions to cephalosporins, penicillins, or other drugs. Cefuroxime axetil /is/ contraindicated in patients who are hypersensitive to cefuroxime or other cephalosporins and should be used with caution in patients with a history of hypersensitivity to penicillins. Use of cephalosporins should be avoided in patients who have had an immediate-type (anaphylactic) hypersensitivity reaction to penicillins.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 71
Prescribing Ceftin in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
US Natl Inst Health; DailyMed. Current Medication Information for ICEFTIN (cefuroxime axetil) powder, for suspension CEFTIN (cefuroxime axetil) tablet, film coated (July 2011). Available from, as of November 13, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2f439b86-59d6-4350-aeb4-c904b3781db4
For more Drug Warnings (Complete) data for Cefuroxime axetil (18 total), please visit the HSDB record page.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
Following oral administration of cefuroxime axetil in pediatric patients with acute otitis media with effusion or with chronic or recurrent otitis media with effusion, cefuroxime is distributed into middle ear effusions. In a study in pediatric patients 1-4 years of age with acute otitis media with effusion who received a single 15 mg/kg dose of cefuroxime as cefuroxime axetil oral suspension, cefuroxime concentrations in middle ear effusions 2-5 hours after a dose ranged from 0.2-3.6 ug/mL; concurrent serum concentrations were 2.8-7.3 ug/mL.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 74
In pharmacokinetic studies of cefuroxime axetil oral suspension in children, the drug was administered postprandially or with food; no data are available regarding absorption of the suspension in fasting children. Following oral administration to children 3 months to 12 years of age (mean age: 23 months) of a single 10-, 15-, or 20-mg/kg dose of commercially available cefuroxime axetil oral suspension concomitantly with milk or milk products, peak serum cefuroxime concentrations are attained approximately 3.6, 2.7, or 3.1 hours after the dose, respectively, and average 3.3, 5.1, or 7 mcg/mL, respectively.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 73
Results of a study in healthy adults indicate that cefuroxime axetil oral suspensions containing 125 mg/5 mL or 250 mg/5 mL are bioequivalent. In healthy adults who received a 250-mg dose of cefuroxime axetil given as a suspension containing 125 mg/5 mL or 250 mg/5 mL with food, peak plasma concentrations of cefuroxime were 2.4 or 2.2 aug/mL, respectively, and were attained 3 hours after the dose.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 73
Following oral administration in adults of a single 125-mg, 250-mg, 500-mg, or 1-g dose of commercially available cefuroxime axetil tablets immediately following a meal, peak serum cefuroxime concentrations are attained approximately 2-3 hours after the dose and average 2.1, 4.1, 7, or 13.6 ug/mL, respectively; serum concentrations 6 hours after the dose average 0.3, 0.7, 2.2, or 3.4 ug/mL, respectively. AUC of the drug in these individuals averaged 6.7, 12.9, 27.4, or 50 mcg-h/mL, respectively.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 73
For more Absorption, Distribution and Excretion (Complete) data for Cefuroxime axetil (13 total), please visit the HSDB record page.
Following oral administration, cefuroxime axetil is rapidly hydrolyzed to cefuroxime by nonspecific esterases in the intestinal mucosa and blood; the axetil moiety is metabolized to acetaldehyde and acetic acid. Cefuroxime is not metabolized and is excreted unchanged principally in urine by both glomerular filtration and tubular secretion.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 74
In neonates and children, the serum half-life of cefuroxime is inversely proportional to age.6 25 30 Following oral administration of cefuroxime axetil oral suspension in children 3 months to 12 years of age, the serum half-life of cefuroxime averages 1.4-1.9 hours.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 74
In adults, the serum or plasma half-life of cefuroxime following oral administration of commercially available cefuroxime axetil tablets or oral suspension ranges from 1.2-1.6 hours.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 74
Because cefuroxime is renally excreted, the serum half-life is prolonged in patients with reduced renal function. In a study of 20 elderly patients (mean age = 83.9 years) having a mean creatinine clearance of 34.9 mL/min, the mean serum elimination half-life was 3.5 hours.
US Natl Inst Health; DailyMed. Current Medication Information for ICEFTIN (cefuroxime axetil) powder, for suspension CEFTIN (cefuroxime axetil) tablet, film coated (July 2011). Available from, as of November 13, 2013: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=2f439b86-59d6-4350-aeb4-c904b3781db4
Cefuroxime is usually bactericidal in action.Like other cephalosporins, the antibacterial activity of the drug results from inhibition of mucopeptide synthesis in the bacterial cell wall.
American Society of Health-System Pharmacists 2013; Drug Information 2013. Bethesda, MD. 2013, p. 72
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21508
Submission : 2008-03-29
Status : Active
Type : II
Date of Issue : 2022-05-17
Valid Till : 2025-05-16
Written Confirmation Number : WC-00008
Address of the Firm :


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21615
Submission : 2008-05-10
Status : Active
Type : II
Date of Issue : 2022-05-17
Valid Till : 2025-05-16
Written Confirmation Number : WC-0008
Address of the Firm :


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21671
Submission : 2008-05-27
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23088
Submission : 2009-08-28
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 36198
Submission : 2021-08-17
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15980
Submission : 2002-05-13
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 14653
Submission : 2000-01-03
Status : Inactive
Type : II

Certificate Number : R1-CEP 2000-398 - Rev 00
Issue Date : 2007-02-23
Type : Chemical
Substance Number : 1300
Status : Valid

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
28
PharmaCompass offers a list of Cefuroxime Axetil API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cefuroxime Axetil manufacturer or Cefuroxime Axetil supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cefuroxime Axetil manufacturer or Cefuroxime Axetil supplier.
A Zinnat manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Zinnat, including repackagers and relabelers. The FDA regulates Zinnat manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Zinnat API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Zinnat manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Zinnat supplier is an individual or a company that provides Zinnat active pharmaceutical ingredient (API) or Zinnat finished formulations upon request. The Zinnat suppliers may include Zinnat API manufacturers, exporters, distributors and traders.
click here to find a list of Zinnat suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Zinnat DMF (Drug Master File) is a document detailing the whole manufacturing process of Zinnat active pharmaceutical ingredient (API) in detail. Different forms of Zinnat DMFs exist exist since differing nations have different regulations, such as Zinnat USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Zinnat DMF submitted to regulatory agencies in the US is known as a USDMF. Zinnat USDMF includes data on Zinnat's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Zinnat USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Zinnat suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Zinnat Drug Master File in Japan (Zinnat JDMF) empowers Zinnat API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Zinnat JDMF during the approval evaluation for pharmaceutical products. At the time of Zinnat JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Zinnat suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Zinnat Drug Master File in Korea (Zinnat KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Zinnat. The MFDS reviews the Zinnat KDMF as part of the drug registration process and uses the information provided in the Zinnat KDMF to evaluate the safety and efficacy of the drug.
After submitting a Zinnat KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Zinnat API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Zinnat suppliers with KDMF on PharmaCompass.
A Zinnat CEP of the European Pharmacopoeia monograph is often referred to as a Zinnat Certificate of Suitability (COS). The purpose of a Zinnat CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Zinnat EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Zinnat to their clients by showing that a Zinnat CEP has been issued for it. The manufacturer submits a Zinnat CEP (COS) as part of the market authorization procedure, and it takes on the role of a Zinnat CEP holder for the record. Additionally, the data presented in the Zinnat CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Zinnat DMF.
A Zinnat CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Zinnat CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Zinnat suppliers with CEP (COS) on PharmaCompass.
A Zinnat written confirmation (Zinnat WC) is an official document issued by a regulatory agency to a Zinnat manufacturer, verifying that the manufacturing facility of a Zinnat active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Zinnat APIs or Zinnat finished pharmaceutical products to another nation, regulatory agencies frequently require a Zinnat WC (written confirmation) as part of the regulatory process.
click here to find a list of Zinnat suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Zinnat as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Zinnat API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Zinnat as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Zinnat and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Zinnat NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Zinnat suppliers with NDC on PharmaCompass.
Zinnat Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Zinnat GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Zinnat GMP manufacturer or Zinnat GMP API supplier for your needs.
A Zinnat CoA (Certificate of Analysis) is a formal document that attests to Zinnat's compliance with Zinnat specifications and serves as a tool for batch-level quality control.
Zinnat CoA mostly includes findings from lab analyses of a specific batch. For each Zinnat CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Zinnat may be tested according to a variety of international standards, such as European Pharmacopoeia (Zinnat EP), Zinnat JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Zinnat USP).

