
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
Canada
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
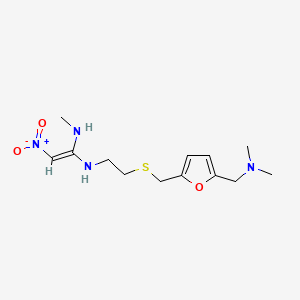

1. Ah 19065
2. Ah-19065
3. Ah19065
4. Biotidin
5. Hydrochloride, Ranitidine
6. N (2-(((5-((dimethylamino)methyl)-2-furanyl)methyl)thio)ethyl)-n'-methyl-2-nitro-1,1-ethenediamine
7. Ranisen
8. Ranitidin
9. Ranitidine Hydrochloride
10. Sostril
11. Zantac
12. Zantic
1. 66357-35-5
2. Ranitidine Base
3. Raticina
4. Ratic
5. Ranitidina
6. Ranitidinum
7. Coralen
8. Gastrial
9. Quantor
10. Rantidine
11. Zantac
12. Gastrosedol
13. Microtid
14. Ptinolin
15. Ranidine
16. Raniogas
17. Ranisen
18. Ranitiget
19. Rantacid
20. Taural
21. Duractin
22. Ulceranin
23. Weichilin
24. Xanidine
25. Zantadin
26. Achedos
27. Acidex
28. Atural
29. Axoban
30. Ezopta
31. Istomar
32. Logast
33. Mauran
34. Quicran
35. Radinat
36. Randin
37. Raniter
38. Sampep
39. Urantac
40. Verlost
41. Vesyca
42. Vizerul
43. Weidos
44. Zantab
45. Ranin
46. Ul-pep
47. Ranitidine Free Base
48. Chebi:8776
49. Ranitidine Impurity J
50. 1,1-ethenediamine, N-(2-(((5-((dimethylamino)methyl)-2-furanyl)methyl)thio)ethyl)-n'-methyl-2-nitro-
51. Ranitidinum [inn-latin]
52. Ranitidina [inn-spanish]
53. Zantac (tn)
54. Chembl512
55. 1,1-ethenediamine, N-[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-n'-methyl-2-nitro-
56. 66357-35-5 (free Base)
57. Ncgc00015876-07
58. Dsstox_cid_25191
59. Dsstox_rid_80738
60. Dsstox_gsid_45191
61. Ranitidine [usan:ban:inn]
62. Rnd
63. Ranitidine (tn)
64. {2-[({5-[(dimethylamino)methyl]furan-2-yl}methyl)sulfanyl]ethyl}[(e)-1-(methylamino)-2-nitroethenyl]amine
65. Dimethyl[(5-{[(2-{[(e)-1-(methylamino)-2-nitroethenyl]amino}ethyl)sulfanyl]methyl}furan-2-yl)methyl]amine
66. Cas-66357-35-5
67. (e)-n-(2-(((5-((dimethylamino)methyl)furan-2-yl)methyl)thio)ethyl)-n-methyl-2-nitroethene-1,1-diamine
68. 71130-06-8
69. Ranitidine (usan/inn)
70. Gavilast
71. Sr-01000075288
72. (e)-ranitidine
73. Tocris-1967
74. Lopac-r-101
75. Prestwick2_000201
76. Spectrum5_001189
77. Epitope Id:127515
78. Ranitidine Hcl 1/2 Type
79. Lopac0_001073
80. N'-[2-[[5-(dimethylaminomethyl)-2-furyl]methylsulfanyl]ethyl]-n-methyl-2-nitro-ethene-1,1-diamine Hydrochloride
81. Bidd:gt0179
82. Spectrum1501151
83. Gtpl1234
84. Bdbm22893
85. Chebi:92246
86. Hms501f22
87. N-[2-[[5-[(dimethylamino)methyl]furfuryl]thio]ethyl]-n'-methyl-2-nitro-1,1-ethenediamine
88. Bdbm237183
89. Dtxsid101112063
90. Hms1921l07
91. Hms2092h15
92. Hms3886a16
93. Pharmakon1600-01501151
94. Ranitidine (form I And Form Ii)
95. Bcp21325
96. Hy-b0693
97. Zinc1530728
98. Tox21_110250
99. Tox21_302372
100. Ccg-39025
101. Mfcd00081180
102. Nsc757851
103. S5662
104. Stk619092
105. Akos005552967
106. Tox21_110250_1
107. Ks-5230
108. Sdccgsbi-0051043.p004
109. Idi1_000440
110. Ncgc00015876-01
111. Ncgc00015876-02
112. Ncgc00015876-03
113. Ncgc00015876-04
114. Ncgc00015876-05
115. Ncgc00015876-06
116. Ncgc00015876-08
117. Ncgc00015876-09
118. Ncgc00015876-11
119. Ncgc00015876-16
120. Ncgc00018108-01
121. Ncgc00018108-02
122. Ncgc00018108-03
123. Ncgc00024387-02
124. Ncgc00094913-01
125. Ncgc00094913-02
126. Ncgc00094913-03
127. Ncgc00094913-04
128. Ncgc00094913-05
129. Ncgc00256269-01
130. ({5-[(2-{[(1e)-1-(methylamino)-2-nitrovinyl]amino}ethylthio)methyl](2-furyl)}m Ethyl)dimethylamine
131. (e)-n-{2-[({5-[(dimethylamino)methyl]-2-furyl}methyl)sulfanyl]ethyl}-n'-methyl-2-nitroethene-1,1-diamine
132. (e)-n-{2-[({5-[(dimethylamino)methyl]furan-2-yl}methyl)sulfanyl]ethyl}-n'-methyl-2-nitroethene-1,1-diamine
133. 82530-72-1
134. Ac-12712
135. Br166204
136. Sbi-0051043.p003
137. Cas-66357-59-3
138. Cs-0009591
139. D00422
140. D97790
141. Ab00052223-10
142. Ab00052223_11
143. Ab00052223_12
144. 357r355
145. A835434
146. A899862
147. L000504
148. Sr-01000075288-3
149. Brd-k70505054-001-02-9
150. Q21971328
151. N-[2-[[5-[(dimethylamino)methyl]furfuryl]thio]ethyl]-n'-methyl-2-nitro-1,1-ethylenediamine
152. (1e)-n-[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-n'-methyl-2-nitro-1,1-ethenediamine
153. (e)-1-n'-[2-[[5-[(dimethylamino)methyl]furan-2-yl]methylsulfanyl]ethyl]-1-n-methyl-2-nitroethene-1,1-diamine
154. (e)-n-(2-((5-((dimethylamino)methyl)furan-2-yl)methylthio)ethyl)-n-methyl-2-nitroethene-1,1-diamine
155. (e)-n1'-[2-[[5-(dimethylaminomethyl)-2-furyl]methylsulfanyl]ethyl]-n1-methyl-2-nitro-ethene-1,1-diamine
156. (e)-n1'-[2-[[5-[(dimethylamino)methyl]-2-furyl]methylsulfanyl]ethyl]-n1-methyl-2-nitro-ethene-1,1-diamine;ranitidine Base
157. 1,1-ethenediamine, N-[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-n'-methyl-2-nitro-, (e)-
158. N-[(e)-1-(methylamino)-2-nitroethenyl]-2-[[[2-[(dimethylamino)methyl]-5-furanyl]methyl]thio]ethanamine
159. N-[2-[[[5-[(dimethylamino)methyl]-2 -furanyl]methyl]thio]ethyl]-n'-methyl-2-nitro-1,1-ethanediamine
160. N-[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-n'-methyl-2-nitro-1,1-ethenediamine
161. N-[2-[[5-(dimethylaminomethyl)furan-2-yl]methylsulfanyl]ethyl]-n'-methyl-2-nitroethene-1,1-diamine
162. N-[2-[[5-[(dimethylamino)methyl]furfuryl]thio]ethyl]-n'-methyl-2-nitrovinylidenediamine Monohydrochloride;ranitidine Hcl
163. N1'-[2-[[5-[(dimethylamino)methyl]-2-furanyl]methylthio]ethyl]-n1-methyl-2-nitroethene-1,1-diamine
| Molecular Weight | 314.41 g/mol |
|---|---|
| Molecular Formula | C13H22N4O3S |
| XLogP3 | 0.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 9 |
| Exact Mass | 314.14126175 g/mol |
| Monoisotopic Mass | 314.14126175 g/mol |
| Topological Polar Surface Area | 112 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 347 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Ranitidine |
| PubMed Health | Ranitidine |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in ZANTAC 150 Tablets, ZANTAC 300 Tablets, and ZANTAC Syrup is ranitidine hydrochloride (HCl), USP, a histamine H2-receptor antagonist. Chemically it is N[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N-methyl-2... |
| Active Ingredient | Ranitidine |
| Dosage Form | Solution |
| Route | oral |
| Strength | 15mg/ml |
| Market Status | Tentative Approval |
| Company | Actavis Elizabeth |
| 2 of 4 | |
|---|---|
| Drug Name | Zantac |
| PubMed Health | Ranitidine (Injection) |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in ZANTAC 150 Tablets, ZANTAC 300 Tablets, and ZANTAC Syrup is ranitidine hydrochloride (HCl), USP, a histamine H2-receptor antagonist. Chemically it is N[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N-methyl-2... |
| Active Ingredient | Ranitidine hydrochloride |
| Dosage Form | Syrup; Injectable |
| Route | Injection; Oral |
| Strength | eq 15mg base/ml; eq 25mg base/ml |
| Market Status | Prescription |
| Company | Covis Injectables; Glaxo Grp |
| 3 of 4 | |
|---|---|
| Drug Name | Ranitidine |
| PubMed Health | Ranitidine |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in ZANTAC 150 Tablets, ZANTAC 300 Tablets, and ZANTAC Syrup is ranitidine hydrochloride (HCl), USP, a histamine H2-receptor antagonist. Chemically it is N[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N-methyl-2... |
| Active Ingredient | Ranitidine |
| Dosage Form | Solution |
| Route | oral |
| Strength | 15mg/ml |
| Market Status | Tentative Approval |
| Company | Actavis Elizabeth |
| 4 of 4 | |
|---|---|
| Drug Name | Zantac |
| PubMed Health | Ranitidine (Injection) |
| Drug Classes | Antiulcer, Gastric Acid Secretion Inhibitor |
| Drug Label | The active ingredient in ZANTAC 150 Tablets, ZANTAC 300 Tablets, and ZANTAC Syrup is ranitidine hydrochloride (HCl), USP, a histamine H2-receptor antagonist. Chemically it is N[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N-methyl-2... |
| Active Ingredient | Ranitidine hydrochloride |
| Dosage Form | Syrup; Injectable |
| Route | Injection; Oral |
| Strength | eq 15mg base/ml; eq 25mg base/ml |
| Market Status | Prescription |
| Company | Covis Injectables; Glaxo Grp |
Anti-Ulcer Agents; Histamine H2 Antagonists
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Ranitidine is effective for the treatment of duodenal or gastric ulcer and relieves symptoms of reflux esophagitis. It heals some NSAID-induced ulcers but does not appear to prevent their initial occurrence. ... Investigationally, this drug prevented aspiration pneumonitis during surgery, and it appears to be useful for the prophylaxis of bleeding due to stress ulcers.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 901
Studies show that ranitidine can adequately inhibit acid secretion in patients with gastric hypersecretory disorders, is safe at high doses, does not cause the antiandrogen side effects frequently seen with high doses of cimetidine, & is threefold more potent than cimetidine. Patients relatively resistant to cimetidine will have proportional resistance to ranitidine.
PMID:6318628 Collen MJ et al; Ann Intern Med 100 (1): 52-8 (1984)
Ranitidine and a placebo were evaluated in the 28 day treatment of duodenal ulcer through an open randomized study performed in 120 patients. At the end of the treatment, ranitidine demonstrated a significantly higher efficacy on ulcer healing as well as on symptom relief in comparison with placebo (P less than 0.05).
Giacosa A et al; Scand J Gastroenterol (Suppl) 17 (72): 215-9 (1982)
For more Therapeutic Uses (Complete) data for RANITIDINE (30 total), please visit the HSDB record page.
Minor adverse effects occur infrequently (incidence less than 3%) and include headache and rashes that usually subside with continued therapy, malaise, nausea, constipation, dizziness, and abdominal pain. ... Usual doses of ranitidine only rarely produce confusion, gynecomastia, hyperprolactinemia, sexual dysfunction, bradycardia, blood dyscrasias, or hepatitis.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 901
Reversible blurred vision suggestive of a change in accommodation has occurred rarely. Exacerbation of ocular pain and blurred vision assoc with increased intraocular pressure and chronic glaucoma have been reported in at least one patient during ranitidine therapy. Loss of color vision, which recurred following rechallenge, has also occurred in at least one patient.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2182
Side effects of /ranitidine and cimetidine/ that are hard to explain are headache, dizziness, malaise, myalgia, nausea, vomiting, diarrhea, and constipation.
Haddad, L.M., Clinical Management of Poisoning and Drug Overdose. 2nd ed. Philadelphia, PA: W.B. Saunders Co., 1990., p. 824
Sexual impotence has occurred in at least one male during ranitidine therapy, but disappeared following discontinuance of the drug; impotence recurred upon rechallenge. Painful gynecomastia also has occurred during oral admin of ranitidine, but disappeared gradually following discontinuance of the drug; gynecomastia reappeared upon rechallenge.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2182
For more Drug Warnings (Complete) data for RANITIDINE (19 total), please visit the HSDB record page.
Anti-Ulcer Agents
Various agents with different action mechanisms used to treat or ameliorate PEPTIC ULCER or irritation of the gastrointestinal tract. This has included ANTIBIOTICS to treat HELICOBACTER INFECTIONS; HISTAMINE H2 ANTAGONISTS to reduce GASTRIC ACID secretion; and ANTACIDS for symptomatic relief. (See all compounds classified as Anti-Ulcer Agents.)
Histamine H2 Antagonists
Drugs that selectively bind to but do not activate histamine H2 receptors, thereby blocking the actions of histamine. Their clinically most important action is the inhibition of acid secretion in the treatment of gastrointestinal ulcers. Smooth muscle may also be affected. Some drugs in this class have strong effects in the central nervous system, but these actions are not well understood. (See all compounds classified as Histamine H2 Antagonists.)
A02BA02
S66 | EAWAGTPS | Parent-Transformation Product Pairs from Eawag | DOI:10.5281/zenodo.3754448
A02BA02
S66 | EAWAGTPS | Parent-Transformation Product Pairs from Eawag | DOI:10.5281/zenodo.3754448
A02BA02
S66 | EAWAGTPS | Parent-Transformation Product Pairs from Eawag | DOI:10.5281/zenodo.3754448
A02BA02
S66 | EAWAGTPS | Parent-Transformation Product Pairs from Eawag | DOI:10.5281/zenodo.3754448
A - Alimentary tract and metabolism
A02 - Drugs for acid related disorders
A02B - Drugs for peptic ulcer and gastro-oesophageal reflux disease (gord)
A02BA - H2-receptor antagonists
A02BA02 - Ranitidine
Twelve normal male subjects received 20, 40, or 80 mg of ranitidine orally 90 min before starting a 3-hr continuous infusion of pentagastrin, 2 ug/kg/hr. Ranitidine reduced hydrogen ion output by 29%, 50% and 70% & secretion volume by 21%, 37%, and 47%. Pepsin activity was reduced by 8%, 50% & 49% by the same doses. Peak serum concn was correlated positively with percent reduction in hydrogen ion output (r= 0.81, P= less than 0.001) & volume (r= 0.71, P less than 0.01) over a 2-hr period. A 50% inhibition of hydrogen ion output was associated with a peak ranitidine serum concn of 165 ug/l and subjects reached peak serum concn 60 to 120 min after oral dosing.
PMID:6269788 Lebert PA et al; Clin Pharmacol Ther 30 (4): 539-44 (1981)
Ranitidine is rapidly absorbed from the GI tract following oral admin and from parenteral sites following IM injection; however, following oral admin, the drug undergoes extensive first-pass metabolism. ... The absolute bioavailability of orally admin ranitidine has been reported to be about 50%; similar oral bioavailability of the drug has been reported in children 3.5-16 yr of age. Following oral admin, area under the plasma conc-time curve may be substantially increased in geriatric individuals compared with younger adults. Following IM admin, the absolute bioavailability of ranitidine is 90-100%.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2180
Following oral admin of 150 mg doses, mean peak serum ranitidine conc of 372-545 ng/ml occur within 2-3 hr and may be positively correlated with age in adults. Following oral admin of single doses of the drug in one study, peak serum conc were biphasic in some individuals with an initial peak occurring @ 0.5-1.5 hr after admin and a second peak occurring about 3 hr after admin. Following IM admin of a single 50-mg dose of the drug, mean peak serum ranitidine conc of 576 ng/ml occur within 15 min.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2180
Ranitidine is widely distributed throughout the body and is 10-19% protein bound. The apparent volume of distribution of ranitidine is reported to be 1.2-1.9 l/kg. The apparent volume of distribution in children 3.5-16 yr of age is reported to be 2.3-2.5 l/kg (range: 1.1-3.7 l/kg).
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2180
For more Absorption, Distribution and Excretion (Complete) data for RANITIDINE (32 total), please visit the HSDB record page.
Ranitidine undergoes significant first-pass metabolism after oral admin. It is metabolized in the liver to the pharmacologically inactive desmethylranitidine, ranitidine-N-oxide, and ranitidine-S-oxide.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 902
Less than 10% of an iv or oral dose is excreted as metabolites; 68% to 79% of an iv dose and 30% of an oral dose appear in the urine as unchanged drug.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 902
The chromatographic behavior of putative N-oxide metabolites relative to the parent amino compounds with the aim of predicting retention data for N-oxides is described. Model compounds were evaluated by reversed phase HPLC and standard TLC systems and the data generated to predict retention values for ranitidine N-oxide and tamoxifen N-oxide based upon those of the parent compounds. The deviation between actual and predicted values was larger than expected.
PMID:2100636 Ballard P, Law B; J Pharm Biomed Anal 8 (8-12): 877-80 (1990)
Ranitidine has known human metabolites that include Desmethylranitidine.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The elimination half-life of ranitidine in adults averages 1.7-3.2 hr and may be positively correlated with age in adults. The elimination half-life is prolonged in patients with renal impairment. In children 3.5-16 yr of age, the elimination half-life averages 1.8-2 hr (range: 1.4-2.9 hr).
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2180
In one study following oral admin of a single 150-mg dose of ranitidine in patients with creatinine clearances averaging 27.2 ml/min, the terminal elimination half-life of ranitidine was 8.7 hr; a correlation between the degree of impairment and the elimination half-life of the drug was not apparent. However, in another study in patients with GFRs ... ranging from 3-69 ml/min per 1.73 sq m, ranitidine clearance was shown to correlate with GFR and elimination half-life of ranitidine was correlated with degree of renal impairment. In a study in patients with creatinine clearances of 0.5-34 ml/min, the elimination half-life ranged from 3-10 hr following IV admin of a single 50-mg dose.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2180
The half-life of ranitidine reportedly is prolonged to about 6 hr in geriatric individuals following oral admin of a 100 mg dose of the drug.
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 2180
H2 antagonists inhibit gastric acid secretion elicited by histamine and other H2 agonists in a dose dependent, competitive manner; the degree of inhibition parallels the concentration of the drug in plasma over a wide range. The H2 antagonists also inhibit acid secretion elicited by gastrin and, to a lesser extent, by muscarinic agonists. Importantly, these drugs inhibit basal (fasting) and nocturnal acid secretion and that stimulated by food, sham feeding, fundic distention, and various pharmacological agents; this property reflects the vital role of histamine in mediating the effects of diverse stimuli. /H2 Receptor Antagonists/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 899
... /H2 Antagonists/ measurably inhibit effects on the cardiovascular and other systems that are elicited through H2 receptors by exogenous or endogenous histamine. /H2 Receptor Antagonists/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 899
...IS A COMPETITIVE ANTAGONIST OF HISTAMINE-INDUCED GASTRIC ACID SECRETION... INHIBITS BOTH THE VOLUME AND CONCENTRATION OF GASTRIC ACID INDUCED NOCTURNALLY AND BY FOOD BUT DOES NOT AFFECT GASTRIC MUCUS OR ITS PRODUCTION. ...DOES NOT AFFECT LOWER ESOPHAGEAL SPHINCTER PRESSURE...
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 901
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
51
PharmaCompass offers a list of Ranitidine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ranitidine manufacturer or Ranitidine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ranitidine manufacturer or Ranitidine supplier.
A Vizerul manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Vizerul, including repackagers and relabelers. The FDA regulates Vizerul manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Vizerul API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Vizerul manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Vizerul supplier is an individual or a company that provides Vizerul active pharmaceutical ingredient (API) or Vizerul finished formulations upon request. The Vizerul suppliers may include Vizerul API manufacturers, exporters, distributors and traders.
click here to find a list of Vizerul suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Vizerul DMF (Drug Master File) is a document detailing the whole manufacturing process of Vizerul active pharmaceutical ingredient (API) in detail. Different forms of Vizerul DMFs exist exist since differing nations have different regulations, such as Vizerul USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Vizerul DMF submitted to regulatory agencies in the US is known as a USDMF. Vizerul USDMF includes data on Vizerul's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Vizerul USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Vizerul suppliers with USDMF on PharmaCompass.
A Vizerul written confirmation (Vizerul WC) is an official document issued by a regulatory agency to a Vizerul manufacturer, verifying that the manufacturing facility of a Vizerul active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Vizerul APIs or Vizerul finished pharmaceutical products to another nation, regulatory agencies frequently require a Vizerul WC (written confirmation) as part of the regulatory process.
click here to find a list of Vizerul suppliers with Written Confirmation (WC) on PharmaCompass.
Vizerul Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Vizerul GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Vizerul GMP manufacturer or Vizerul GMP API supplier for your needs.
A Vizerul CoA (Certificate of Analysis) is a formal document that attests to Vizerul's compliance with Vizerul specifications and serves as a tool for batch-level quality control.
Vizerul CoA mostly includes findings from lab analyses of a specific batch. For each Vizerul CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Vizerul may be tested according to a variety of international standards, such as European Pharmacopoeia (Vizerul EP), Vizerul JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Vizerul USP).

