
Synopsis
Synopsis
0
CEP/COS
0
KDMF
0
VMF
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Annual Reports
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
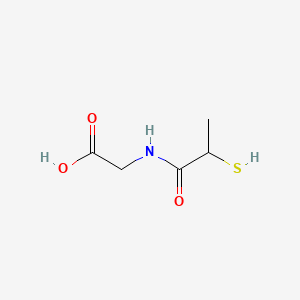

1. 2 Mercaptopropionylglycine
2. 2 Thiol Propionamido Acetic Acid
3. 2 Thiolpropionamidoacetic Acid
4. 2-mercaptopropionylglycine
5. 2-thiol-propionamido-acetic Acid
6. 2-thiolpropionamidoacetic Acid
7. Acadione
8. Acid, 2-thiol-propionamido-acetic
9. Acid, 2-thiolpropionamidoacetic
10. Alpha Mercaptopropionylglycine
11. Alpha-mercaptopropionylglycine
12. Captimer
13. Meprin
14. Mercaptopropionylglycine
15. Thiola
16. Thiopronine
17. Tiopronine
1. 1953-02-2
2. N-(2-mercaptopropionyl)glycine
3. Acadione
4. Captimer
5. Thiopronine
6. Mucolysin
7. Capen
8. Thiola
9. Tiopronine
10. Epatiol
11. 2-(2-sulfanylpropanoylamino)acetic Acid
12. Thiosol
13. Glycine, N-(2-mercapto-1-oxopropyl)-
14. Tiopronin (thiola)
15. A-mercaptopropionyl Glycine
16. 2-(2-mercaptopropanamido)acetic Acid
17. Nsc-760416
18. Chembl1314
19. C5w04go61s
20. N-(2-mercaptopropanoyl)glycine
21. Thiopronin
22. Chebi:32229
23. 2-(2-sulfanylpropanamido)acetic Acid
24. Ncgc00159422-02
25. Ncgc00159422-04
26. Sutilan
27. Dsstox_cid_3678
28. Glycine, N-(2-mercapto-1-oxopropyl)- (9ci)
29. Dsstox_rid_77142
30. Dsstox_gsid_23678
31. Thiolpropionamidoacetic Acid
32. Tioglis
33. Vincol
34. Cas-1953-02-2
35. Meprin (detoxicant)
36. N-(2-mercapto-1-oxopropyl)glycine
37. Tiopronine [inn-french]
38. Tioproninum [inn-latin]
39. Tiopronino [inn-spanish]
40. (2-mercaptopropionyl)glycine
41. Tiopronin [inn:dcf:jan]
42. Tiopronino
43. Tioproninum
44. Ccris 1935
45. (s)-2-(2-mercaptopropanamido)acetic Acid
46. Einecs 217-778-4
47. Thiola (tn)
48. Brn 1859822
49. Thiola Ec
50. Tiopronin (jan/inn)
51. Tiopronin [inn]
52. Tiopronin [jan]
53. Tiopronin [mi]
54. Glycine, N-(2-mercaptopropionyl)-
55. Tiopronin [vandf]
56. 2-mercapto-propionylglycine
57. Tiopronin [mart.]
58. Tiopronin [who-dd]
59. (2-mercaptopropanoyl)glycine
60. Unii-c5w04go61s
61. Schembl19989
62. Mls006010632
63. Alpha-mercaptopropionyl Glycine
64. Mercaptopropionylglycine-
65. N-(2-sulfanylpropanoyl)glycine
66. Tiopronin [orange Book]
67. Dtxsid4023678
68. N-(2-mercaptopropionyl) Glycine
69. N-(2-mercaptopropanoyl) Glycine
70. Acadione; Capen; Epatiol; Vincol
71. Hms3264b11
72. Hms3655l15
73. Pharmakon1600-01506190
74. Amy39003
75. Bcp13354
76. Hy-b0373
77. 2-(2-mercaptopropanamido)aceticacid
78. Tox21_111654
79. Bdbm50020805
80. Mfcd00004861
81. Mfcd30157366
82. Nsc760416
83. S2062
84. (2-mercaptopropionylamino)acetic Acid
85. Akos015895408
86. N-(2-mercaptopropionyl)glycine, 99%
87. Tox21_111654_1
88. Ac-2087
89. Ccg-214007
90. Db06823
91. Nsc 760416
92. (2-mercapto-propionylamino)-acetic Acid
93. Ncgc00159422-03
94. As-12522
95. Smr001550282
96. Sy262996
97. Ft-0603530
98. Ft-0653686
99. Sw219206-1
100. T2614
101. Tiopronin, Vetranal(tm), Analytical Standard
102. C73708
103. D01430
104. Ab00376096_02
105. 953t022
106. A813794
107. Q414456
108. Sr-01000942263
109. J-012651
110. Sr-01000942263-1
111. Tiopronin, Dextiopronin, Tiopronin (n-2-mercaptopropionyl Glycine)
| Molecular Weight | 163.20 g/mol |
|---|---|
| Molecular Formula | C5H9NO3S |
| XLogP3 | -0.1 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 163.03031432 g/mol |
| Monoisotopic Mass | 163.03031432 g/mol |
| Topological Polar Surface Area | 67.4 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 148 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Tiopronin is indicated for the prevention of kidney stone formation in patients with severe homozygous cystinuria consisting of a urinary cystine concentration greater than 500 mg/day, and who have failed treatment with non-pharmacological measures of increased fluid intake, decreased sodium and protein intake, and urine alkalinization.
G - Genito urinary system and sex hormones
G04 - Urologicals
G04B - Urologicals
G04BX - Other urologicals
G04BX16 - Tiopronin
Absorption
Tiopronin undergoes slow absorption, reaching peak plasma concentration 3-6 hours after ingestion. In a study of healthy subjects, the bioavailability of total and unbound tiopronin was found to be 63% and 40%, respectively.
Route of Elimination
Tiopronin is 100% excreted in urine.
Volume of Distribution
The volume of distribution of tiopronin is high at 455 L, indicating that a large portion of the drug is bound to tissues outside plasma.
Clearance
Total renal clearance for the total and unbound fractions of tiopronin were found to be 3.3 and 13.3 L/h respectively.
The principle metabolite of tiopronin is 2-mercaptopropionic acid (2-MPA). Between 10-15% of the drug is metabolized to 2-MPA via hydrolysis.
Tiopronin has a long terminal half life of 53 hours in healthy subjects. However, the unbound drug fraction of tiopronin is eliminated much more rapidly from plasma with a calculated half life of 1.8 hours.
Kidney stones form when the solubility limit is exceeded and urine becomes supersaturated with endogenous cystine. Tiopronin is an active reducing agent which undergoes a thiol-disulfide exchange with cystine to form a water-soluble mixed disulfide complex. Thus, the amount of sparingly soluble cystine is reduced. By reducing urinary cystine concentrations below the solubility limit, tiopronin helps reduce cystine stone formation.
Date of Issue : 2023-11-15
Valid Till : 2026-11-14
Written Confirmation Number : WC-0404
Address of the Firm : Plot No. 40/A, Sy Nos. 13,15,16,51,52 & 182, J.N. Pharma city, Thanam Village, P...

Date of Issue : 2025-09-24
Valid Till : 2028-08-08
Written Confirmation Number : WC-0383
Address of the Firm : Unit-ll, Sy No * Parts of 454,455,457,458 & 459 Chandampet-Village, Shankarampet...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Venxxiva is indicated, in combination with high fluid intake, alkali, and diet modification, for the prevention of cystine stone formation with severe homozygous cystinuria.
Lead Product(s): Tiopronin,Inapplicable
Therapeutic Area: Nephrology Brand Name: Venxxiva
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable March 18, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Tiopronin,Inapplicable
Therapeutic Area : Nephrology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
US Launch of VENXXIVA for Cystinuria Treatment
Details : Venxxiva is indicated, in combination with high fluid intake, alkali, and diet modification, for the prevention of cystine stone formation with severe homozygous cystinuria.
Product Name : Venxxiva
Product Type : Miscellaneous
Upfront Cash : Inapplicable
March 18, 2025

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Thiola EC-Generic (tiopronin) is a reducing & cystine-binding thiol drug, which is indicated to help prevent the formation of one type (cystine) of kidney stones in certain adult & pediatric patients.
Lead Product(s): Tiopronin,Inapplicable
Therapeutic Area: Nephrology Brand Name: Thiola EC-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable July 15, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Tiopronin,Inapplicable
Therapeutic Area : Nephrology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Endo Launches Tiopronin Delayed-Release Tablets, Generic Version of THIOLA EC®
Details : Thiola EC-Generic (tiopronin) is a reducing & cystine-binding thiol drug, which is indicated to help prevent the formation of one type (cystine) of kidney stones in certain adult & pediatric patients.
Product Name : Thiola EC-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
July 15, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tiopronin is an active reducing agent which undergoes thiol-disulfide exchange with cystine to form a mixed disulfide of tiopronin-cysteine. It is indicated for cystinuria.
Lead Product(s): Tiopronin,Inapplicable
Therapeutic Area: Nephrology Brand Name: Tiopronin-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Torrent Pharmaceuticals Limited
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 13, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Tiopronin,Inapplicable
Therapeutic Area : Nephrology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Torrent Pharmaceuticals Limited
Deal Size : Inapplicable
Deal Type : Inapplicable
New Tiopronin Delayed-Release Tablets Launch For Cystinuria Treatment
Details : Tiopronin is an active reducing agent which undergoes thiol-disulfide exchange with cystine to form a mixed disulfide of tiopronin-cysteine. It is indicated for cystinuria.
Product Name : Tiopronin-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
May 13, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Teva will provide patients with first-to-market access to a generic version of THIOLA® (Tiopronin) tablets for the prevention of kidney stones, and who are unresponsive to treatment with high fluid intake, alkali, and diet modification.
Lead Product(s): Tiopronin,Inapplicable
Therapeutic Area: Nephrology Brand Name: Tiopronin-Generic
Study Phase: Approved FDFProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable May 17, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Tiopronin,Inapplicable
Therapeutic Area : Nephrology
Highest Development Status : Approved FDF
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Teva Announces the U.S. Launch of the First Generic Version of THIOLA® (tiopronin) Tablets
Details : Teva will provide patients with first-to-market access to a generic version of THIOLA® (Tiopronin) tablets for the prevention of kidney stones, and who are unresponsive to treatment with high fluid intake, alkali, and diet modification.
Product Name : Tiopronin-Generic
Product Type : Miscellaneous
Upfront Cash : Inapplicable
May 17, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tiopronin is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Cystinuria.
Lead Product(s): Tiopronin,Inapplicable
Therapeutic Area: Nephrology Brand Name: Undisclosed
Study Phase: Phase IIProduct Type: Miscellaneous
Sponsor: Undisclosed
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable September 10, 2018

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Tiopronin,Inapplicable
Therapeutic Area : Nephrology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : Undisclosed
Deal Size : Inapplicable
Deal Type : Inapplicable
Effect of Increasing Doses of Tiopronin on Cystine Capacity in Patients With Cystinuria
Details : Tiopronin is a Other Small Molecule drug candidate, which is currently being evaluated in phase II clinical studies for the treatment of Cystinuria.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
September 10, 2018

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Tiopronin is a Other Small Molecule drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Cystinuria.
Lead Product(s): Tiopronin,Inapplicable
Therapeutic Area: Nephrology Brand Name: Undisclosed
Study Phase: Phase IVProduct Type: Miscellaneous
Sponsor: National Institute of Diabetes and Digestive and Kidney Diseases
Deal Size: Inapplicable Upfront Cash: Inapplicable
Deal Type: Inapplicable April 29, 2014

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Tiopronin,Inapplicable
Therapeutic Area : Nephrology
Highest Development Status : Phase IV
Partner/Sponsor/Collaborator : National Institute of Diabetes and Digestive and Kidney Diseases
Deal Size : Inapplicable
Deal Type : Inapplicable
Details : Tiopronin is a Other Small Molecule drug candidate, which is currently being evaluated in phase IV clinical studies for the treatment of Cystinuria.
Product Name : Undisclosed
Product Type : Miscellaneous
Upfront Cash : Inapplicable
April 29, 2014

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Reply
03 Jul 2020
Reply
17 Dec 2019
Reply
28 Nov 2019
Reply
05 Feb 2019
Reply
01 Mar 2016
Reply
08 Feb 2016

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
68
PharmaCompass offers a list of Tiopronin API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tiopronin manufacturer or Tiopronin supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Tiopronin manufacturer or Tiopronin supplier.
A Vincol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Vincol, including repackagers and relabelers. The FDA regulates Vincol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Vincol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Vincol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Vincol supplier is an individual or a company that provides Vincol active pharmaceutical ingredient (API) or Vincol finished formulations upon request. The Vincol suppliers may include Vincol API manufacturers, exporters, distributors and traders.
click here to find a list of Vincol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Vincol DMF (Drug Master File) is a document detailing the whole manufacturing process of Vincol active pharmaceutical ingredient (API) in detail. Different forms of Vincol DMFs exist exist since differing nations have different regulations, such as Vincol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Vincol DMF submitted to regulatory agencies in the US is known as a USDMF. Vincol USDMF includes data on Vincol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Vincol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Vincol suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Vincol Drug Master File in Japan (Vincol JDMF) empowers Vincol API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Vincol JDMF during the approval evaluation for pharmaceutical products. At the time of Vincol JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Vincol suppliers with JDMF on PharmaCompass.
A Vincol written confirmation (Vincol WC) is an official document issued by a regulatory agency to a Vincol manufacturer, verifying that the manufacturing facility of a Vincol active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Vincol APIs or Vincol finished pharmaceutical products to another nation, regulatory agencies frequently require a Vincol WC (written confirmation) as part of the regulatory process.
click here to find a list of Vincol suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Vincol as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Vincol API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Vincol as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Vincol and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Vincol NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Vincol suppliers with NDC on PharmaCompass.
Vincol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Vincol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Vincol GMP manufacturer or Vincol GMP API supplier for your needs.
A Vincol CoA (Certificate of Analysis) is a formal document that attests to Vincol's compliance with Vincol specifications and serves as a tool for batch-level quality control.
Vincol CoA mostly includes findings from lab analyses of a specific batch. For each Vincol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Vincol may be tested according to a variety of international standards, such as European Pharmacopoeia (Vincol EP), Vincol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Vincol USP).

