
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
Canada
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
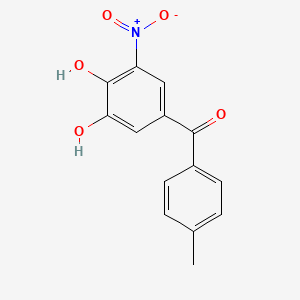

1. 3,4 Dihydroxy 5' Methyl 5 Nitrobenzophenone
2. 3,4-dihydroxy-5'-methyl-5-nitrobenzophenone
3. Ro 40 7592
4. Ro 40-7592
5. Ro 407592
6. Ro-40-7592
7. Ro407592
8. Som0226
9. Tasmar
1. 134308-13-7
2. Tasmar
3. Ro 40-7592
4. 3,4-dihydroxy-4'-methyl-5-nitrobenzophenone
5. (3,4-dihydroxy-5-nitrophenyl)(4-methylphenyl)methanone
6. Ro-40-7592
7. (3,4-dihydroxy-5-nitrophenyl)(p-tolyl)methanone
8. (3,4-dihydroxy-5-nitrophenyl)-(4-methylphenyl)methanone
9. Methanone, (3,4-dihydroxy-5-nitrophenyl)(4-methylphenyl)-
10. Mfcd00866569
11. Ro-407592
12. Chembl1324
13. Cif6334oly
14. Chebi:63630
15. Ncgc00181767-01
16. 3,4-dihydroxy-5-nitro-4'-methylbenzophenone
17. 4'-methyl-3,4-dihydroxy-5-nitrobenzophenone
18. Dsstox_cid_3685
19. Dsstox_rid_77146
20. Dsstox_gsid_23685
21. Talcapone
22. Unii-cif6334oly
23. Tasmar (tn)
24. Ccris 7904
25. Cas-134308-13-7
26. Tolcapone [usan:usp:inn:ban]
27. Tolcapone- Bio-x
28. Tcw
29. Tolcapone [inn]
30. Tolcapone [jan]
31. Tolcapone [mi]
32. Tolcapone [usan]
33. Tolcapone [vandf]
34. Tolcapone [mart.]
35. Tolcapone [usp-rs]
36. Tolcapone [who-dd]
37. Tolcapone (jan/usp/inn)
38. Schembl33869
39. Tolcapone [ema Epar]
40. Mls006012044
41. Bidd:gt0032
42. Gtpl6646
43. Tolcapone [orange Book]
44. Dtxsid3023685
45. Tolcapone, >=98% (hplc)
46. Methanone,(3,4-dihydroxy-5-nitrophenyl)(4-methylphenyl)-
47. Tolcapone [usp Monograph]
48. 3s68
49. Hms2089k14
50. Hms3652m17
51. Hms3715d16
52. Hms3872f03
53. Hms3885g18
54. Bcp09156
55. Tox21_112963
56. Tox21_302414
57. 3,4-dihydroxy-4 Inverted Exclamation Mark -methyl-5-nitrobenzophenone
58. Ac-791
59. Bdbm50108877
60. S4021
61. Zinc35342789
62. Akos015902328
63. Tox21_112963_1
64. As-7085
65. Ccg-221217
66. Cs-1173
67. Db00323
68. Ks-1314
69. Ncgc00181767-02
70. Ncgc00181767-03
71. Ncgc00255188-01
72. Bt164481
73. Hy-17406
74. Smr001614567
75. Sy102278
76. Db-013561
77. Ft-0631149
78. Sw219511-1
79. T3856
80. C07949
81. D00786
82. O10068
83. Ab01275441-01
84. Ab01275441_02
85. (3,4-dihydroxy-5-nitro-phenyl)-p-tolyl-methanone
86. 308t137
87. Q413840
88. Sr-05000001444
89. Q-201840
90. Sr-05000001444-1
91. Brd-k10852020-001-01-1
92. 5-[(4-methylphenyl)carbonyl]-3-nitrobenzene-1,2-diol
93. Tolcapone, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 273.24 g/mol |
|---|---|
| Molecular Formula | C14H11NO5 |
| XLogP3 | 3.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 273.06372245 g/mol |
| Monoisotopic Mass | 273.06372245 g/mol |
| Topological Polar Surface Area | 103 Ų |
| Heavy Atom Count | 20 |
| Formal Charge | 0 |
| Complexity | 372 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Tasmar |
| PubMed Health | Tolcapone (By mouth) |
| Drug Classes | Antiparkinsonian |
| Drug Label | TASMAR is available as tablets containing 100 mg tolcapone.Tolcapone, an inhibitor of catechol-O-methyltransferase (COMT), is used in the treatment of Parkinson's disease as an adjunct to levodopa/carbidopa therapy. It is a yellow, odorless, non-hy.. |
| Active Ingredient | Tolcapone |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 100mg |
| Market Status | Prescription |
| Company | Valeant Pharms |
| 2 of 2 | |
|---|---|
| Drug Name | Tasmar |
| PubMed Health | Tolcapone (By mouth) |
| Drug Classes | Antiparkinsonian |
| Drug Label | TASMAR is available as tablets containing 100 mg tolcapone.Tolcapone, an inhibitor of catechol-O-methyltransferase (COMT), is used in the treatment of Parkinson's disease as an adjunct to levodopa/carbidopa therapy. It is a yellow, odorless, non-hy.. |
| Active Ingredient | Tolcapone |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 100mg |
| Market Status | Prescription |
| Company | Valeant Pharms |
Used as an adjunct to levodopa/carbidopa therapy for the symptomatic treatment of Parkinson's Disease. This drug is generally reserved for patients with parkinsonian syndrome receiving levodopa/carbidopa who are experiencing symptom fluctuations and are not responding adequately to or are not candidates for other adjunctive therapies.
FDA Label
Tasmar is indicated in combination with levodopa / benserazide or levodopa / carbidopa for use in patients with levodopa-responsive idiopathic Parkinsons disease and motor fluctuations, who failed to respond to or are intolerant of other catechol-O-methyltransferase (COMT) inhibitors.
Because of the risk of potentially fatal, acute liver injury, Tasmar should not be considered as a first-line adjunct therapy to levodopa / benserazide or levodopa / carbidopa.
Since Tasmar should be used only in combination with levodopa / benserazide and levodopa / carbidopa, the prescribing information for these levodopa preparations is also applicable to their concomitant use with Tasmar.
Tolcapone is a potent, selective, and reversible inhibitor of catechol-O-methyltransferase (COMT). In humans, COMT is distributed throughout various organs. COMT catalyzes the transfer of the methyl group of S-adenosyl-L-methionine to the phenolic group of substrates that contain a catechol structure. Physiological substrates of COMT include dopa, catecholamines (dopamine, norepinephrine, epinephrine) and their hydroxylated metabolites. The function of COMT is the elimination of biologically active catechols and some other hydroxylated metabolites. COMT is responsible for the elimination of biologically active catechols and some other hydroxylated metabolites. In the presence of a decarboxylase inhibitor, COMT becomes the major metabolizing enzyme for levodopa catalyzing it to 3-methoxy-4-hydroxy-L-phenylalanine (3-OMD) in the brain and periphery. When tolcapone is given in conjunction with levodopa and an aromatic amino acid decarboxylase inhibitor, such as carbidopa, plasma levels of levodopa are more sustained than after administration of levodopa and an aromatic amino acid decarboxylase inhibitor alone. It is believed that these sustained plasma levels of levodopa result in more constant dopaminergic stimulation in the brain, leading to greater effects on the signs and symptoms of Parkinson's disease in patients as well as increased levodopa adverse effects, sometimes requiring a decrease in the dose of levodopa.
Antiparkinson Agents
Agents used in the treatment of Parkinson's disease. The most commonly used drugs act on the dopaminergic system in the striatum and basal ganglia or are centrally acting muscarinic antagonists. (See all compounds classified as Antiparkinson Agents.)
Catechol O-Methyltransferase Inhibitors
Compounds and drugs that inhibit or block the activity of CATECHOL O-METHYLTRANSFERASE enzymes. Drugs in this class are used in management of central nervous system disorders such as PARKINSON DISEASE. (See all compounds classified as Catechol O-Methyltransferase Inhibitors.)
N04BX01
N04BX01
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
N - Nervous system
N04 - Anti-parkinson drugs
N04B - Dopaminergic agents
N04BX - Other dopaminergic agents
N04BX01 - Tolcapone
Absorption
Rapidly absorbed (absolute bioavailability is about 65%)
Route of Elimination
Tolcapone is almost completely metabolized prior to excretion, with only a very small amount (0.5% of dose) found unchanged in urine. The glucuronide conjugate of tolcapone is mainly excreted in the urine but is also excreted in the bile.
Volume of Distribution
9 L
Clearance
7 L/h
The main metabolic pathway of tolcapone is glucuronidation
Tolcapone has known human metabolites that include (2S,3S,4S,5R)-3,4,5-trihydroxy-6-[2-hydroxy-4-(4-methylbenzoyl)-6-nitrophenoxy]oxane-2-carboxylic acid.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
2-3.5 hours
The precise mechanism of action of tolcapone is unknown, but it is believed to be related to its ability to inhibit COMT and alter the plasma pharmacokinetics of levodopa, resulting in an increase in plasma levodopa concentrations. The inhibition of COMT also causes a reduction in circulating 3-OMD as a result of decreased peripheral metabolism of levodopa. This may lead to an increase distribution of levodopa into the CNS through the reduction of its competitive substrate, 3-OMD, for transport mechanisms. Sustained levodopa concentrations presumably result in more consistent dopaminergic stimulation, resulting in greater reduction in the manifestations of parkinsonian syndrome.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
29
PharmaCompass offers a list of Tolcapone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tolcapone manufacturer or Tolcapone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Tolcapone manufacturer or Tolcapone supplier.
A Tolcapone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Tolcapone, including repackagers and relabelers. The FDA regulates Tolcapone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Tolcapone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Tolcapone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Tolcapone supplier is an individual or a company that provides Tolcapone active pharmaceutical ingredient (API) or Tolcapone finished formulations upon request. The Tolcapone suppliers may include Tolcapone API manufacturers, exporters, distributors and traders.
click here to find a list of Tolcapone suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Tolcapone DMF (Drug Master File) is a document detailing the whole manufacturing process of Tolcapone active pharmaceutical ingredient (API) in detail. Different forms of Tolcapone DMFs exist exist since differing nations have different regulations, such as Tolcapone USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Tolcapone DMF submitted to regulatory agencies in the US is known as a USDMF. Tolcapone USDMF includes data on Tolcapone's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Tolcapone USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Tolcapone suppliers with USDMF on PharmaCompass.
A Tolcapone written confirmation (Tolcapone WC) is an official document issued by a regulatory agency to a Tolcapone manufacturer, verifying that the manufacturing facility of a Tolcapone active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Tolcapone APIs or Tolcapone finished pharmaceutical products to another nation, regulatory agencies frequently require a Tolcapone WC (written confirmation) as part of the regulatory process.
click here to find a list of Tolcapone suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Tolcapone as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Tolcapone API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Tolcapone as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Tolcapone and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Tolcapone NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Tolcapone suppliers with NDC on PharmaCompass.
Tolcapone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Tolcapone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tolcapone GMP manufacturer or Tolcapone GMP API supplier for your needs.
A Tolcapone CoA (Certificate of Analysis) is a formal document that attests to Tolcapone's compliance with Tolcapone specifications and serves as a tool for batch-level quality control.
Tolcapone CoA mostly includes findings from lab analyses of a specific batch. For each Tolcapone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Tolcapone may be tested according to a variety of international standards, such as European Pharmacopoeia (Tolcapone EP), Tolcapone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Tolcapone USP).

