
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
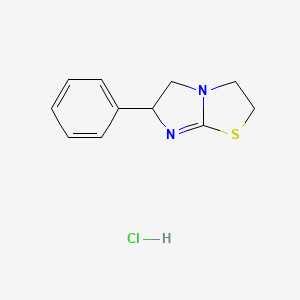

1. Nilverm
2. Tetramisole
1. 5086-74-8
2. Anthelvet
3. Tetramisole Hcl
4. Citarin
5. Concurat
6. Tetramisole (hydrochloride)
7. Ascarotrat
8. Nilverom
9. Ripercol
10. Nilverm
11. Pig Wormer
12. Solaskil Hydrochloride
13. Mcn-jr-8299-11
14. Mcn Jr 8299
15. R 8299
16. 6-phenyl-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole Hydrochloride
17. Tetramisole Hydrochloride [usan]
18. Dl-tetramisole Hydrochloride
19. Spartakon
20. Tetramisol
21. Ashitetra; Duxamintic
22. 6-phenyl-2,3,5,6-tetrahydroimidazo[2,1-b]thiazole Hydrochloride
23. Nsc-170985
24. 0ndk265mcv
25. Tetramisole Hydrochloride, Dl-
26. Chebi:9496
27. Levamisole Dl-form Hydrochloride
28. Orovermol
29. 4641-34-3
30. Citarin (van)
31. Tetramisole Hydrochloride (usan)
32. Bayer 9051
33. (+/-)-tetramisole Hydrochloride;dl-tetramisole Hydrochloride;r-829
34. 6-phenyl-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole;hydrochloride
35. Nsc 170985
36. Mcn-jr-8299
37. Sr-01000076148
38. Einecs 225-799-5
39. Unii-0ndk265mcv
40. Ripereol
41. Ncgc00094446-03
42. Prestwick_854
43. Ergamisol Hydrochloride
44. Tetramisol Hydrochloride
45. (.+-.)-anthelvet
46. Dl-6-phenyl-2,3,5,6-tetrahydroimidazole(2,1-b)thiazole-hydrochloride
47. (+-)-2,3,5,6-tetrahydro-6-phenylimidazo(2,1-b)thiazole Monohydrochloride
48. Dl Tetramisole Hydrochloride
49. Mcnjr8299
50. Schembl44230
51. Bmk1-h7
52. Imidazo[2, 2,3,5,6-tetrahydro-6-phenyl-, Monohydrochloride
53. Mls000028416
54. Chembl554891
55. (+-)-tetramisole Hydrochloride
56. (.+-.)-tetramisole Hydrochloride
57. Dtxsid101017245
58. Hms1569n11
59. Pharmakon1600-01503245
60. Pharmakon1600-01505679
61. Amy40516
62. Hy-b1194
63. Tox21_501194
64. Mfcd00005536
65. Nsc170985
66. Nsc215179
67. Nsc758437
68. Nsc759175
69. S4094
70. Akos015900432
71. Ccg-213508
72. Ccg-220545
73. Ccg-222498
74. Cs-4801
75. Hs-0026
76. Lp01194
77. Methyl4-methoxy-3-nitrobenzoate
78. Nsc-215179
79. Nsc-758437
80. Nsc-759175
81. Sb18744
82. Imidazo(2,1-b)thiazole, 2,3,5,6-tetrahydro-6-phenyl-, Monohydrochloride, (+-)-
83. Ncgc00094446-01
84. Ncgc00094446-02
85. Ncgc00094446-04
86. Ncgc00094446-05
87. Ncgc00261879-01
88. Tetramisole Hydrochloride [mart.]
89. Ac-22590
90. Rp 16535
91. Smr000058395
92. Tetramisole Hydrochloride [who-dd]
93. Db-043639
94. Eu-0101194
95. Ft-0625516
96. Ft-0659999
97. Ft-0670770
98. Levamisole Dl-form Hydrochloride [mi]
99. R-829
100. T3663
101. Wln: T55 An Dn Fs Dutj Cr &gh -r
102. D06091
103. D70698
104. T 1512
105. Tetramisole Hydrochloride, Phosphatase Inhibitor
106. 5086-74-8, 5036-02-2(free Base)
107. A828325
108. Sr-01000076148-1
109. Sr-01000076148-5
110. W-105926
111. Q27108410
112. ( Inverted Exclamation Marka)-tetramisole Hydrochloride
113. 2,5,6-tetrahydro-6-phenylimidazo[2,1-b]thiazole Hydrochloride
114. Tetramisole Hydrochloride, Vetranal(tm), Analytical Standard
115. 6-phenyl-2,3,5,6-tetrahydroimidazo[2,1-b]thiazolehydrochloride
116. Dl-6-phenyl-2,5,6-tetrahydroimidazole[2,1-b]thiazole-hydrochloride
117. (+/-)-2,3,5,6-tetrahydro-6-phenylimidazo[2,1-b]thiazole Hydrochloride
118. (.+-.)-2,5,6-tetrahydro-6-phenylimidazo[2,1-b]thiazole Monohydrochloride
119. Imidazo[2, 2,3,5,6-tetrahydro-6-phenyl-, Monohydrochloride, (.+-.)-
120. ( Inverted Exclamation Marka)-tetramisole Hydrochloride;dl-tetramisole Hydrochloride;r-8299
121. ( Inverted Question Mark)-2,3,5,6-tetrahydro-6-phenylimidazo[2,1-b]thiazole Hydrochloride
122. (+/-)-2,3,5,6-tetrahydro-6-phenylimidazo(2,1-b)thiazole Monohydrochloride
123. Imidazo(2,1-b)thiazole, 2,3,5,6-tetrahydro-6-phenyl-, Monohydrochloride, (+/-)-
124. Imidazo[2,1-b]thiazole, 2,3,5,6-tetrahydro-6-phenyl-, Hydrochloride (1:1)
| Molecular Weight | 240.75 g/mol |
|---|---|
| Molecular Formula | C11H13ClN2S |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Exact Mass | 240.0487973 g/mol |
| Monoisotopic Mass | 240.0487973 g/mol |
| Topological Polar Surface Area | 40.9 Ų |
| Heavy Atom Count | 15 |
| Formal Charge | 0 |
| Complexity | 246 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Market Place

ABOUT THIS PAGE
19
PharmaCompass offers a list of Tetramisole Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tetramisole Hydrochloride manufacturer or Tetramisole Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Tetramisole Hydrochloride manufacturer or Tetramisole Hydrochloride supplier.
A Tetramisole Hydrochloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Tetramisole Hydrochloride, including repackagers and relabelers. The FDA regulates Tetramisole Hydrochloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Tetramisole Hydrochloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Tetramisole Hydrochloride manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Tetramisole Hydrochloride supplier is an individual or a company that provides Tetramisole Hydrochloride active pharmaceutical ingredient (API) or Tetramisole Hydrochloride finished formulations upon request. The Tetramisole Hydrochloride suppliers may include Tetramisole Hydrochloride API manufacturers, exporters, distributors and traders.
click here to find a list of Tetramisole Hydrochloride suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Tetramisole Hydrochloride DMF (Drug Master File) is a document detailing the whole manufacturing process of Tetramisole Hydrochloride active pharmaceutical ingredient (API) in detail. Different forms of Tetramisole Hydrochloride DMFs exist exist since differing nations have different regulations, such as Tetramisole Hydrochloride USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Tetramisole Hydrochloride DMF submitted to regulatory agencies in the US is known as a USDMF. Tetramisole Hydrochloride USDMF includes data on Tetramisole Hydrochloride's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Tetramisole Hydrochloride USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Tetramisole Hydrochloride suppliers with USDMF on PharmaCompass.
Tetramisole Hydrochloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Tetramisole Hydrochloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Tetramisole Hydrochloride GMP manufacturer or Tetramisole Hydrochloride GMP API supplier for your needs.
A Tetramisole Hydrochloride CoA (Certificate of Analysis) is a formal document that attests to Tetramisole Hydrochloride's compliance with Tetramisole Hydrochloride specifications and serves as a tool for batch-level quality control.
Tetramisole Hydrochloride CoA mostly includes findings from lab analyses of a specific batch. For each Tetramisole Hydrochloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Tetramisole Hydrochloride may be tested according to a variety of international standards, such as European Pharmacopoeia (Tetramisole Hydrochloride EP), Tetramisole Hydrochloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Tetramisole Hydrochloride USP).

