
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDA Orange Book
0
Canada
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
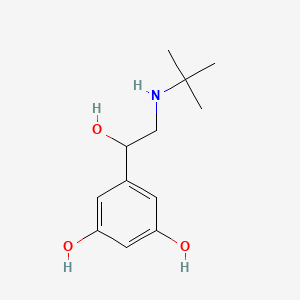

1. Arubendol
2. Asthmoprotect
3. Brethaire
4. Brethine
5. Bricanyl
6. Bricanyl Sa
7. Butaliret
8. Butalitab
9. Contimit
10. Kwd 2019
11. Kwd-2019
12. Kwd2019
13. Monovent
14. Taziken
15. Tedipulmo
16. Terbasmin
17. Terbul
18. Terbutalin Al
19. Terbutalin Ratiopharm
20. Terbutalin Stada
21. Terbutalin Von Ct
22. Terbutalin-ratiopharm
23. Terbutaline Sulfate
24. Terbuturmant
1. 23031-25-6
2. Terbutalin
3. Brican
4. Brethine
5. Bricaril
6. Bricar
7. Bricyn
8. Brethaire
9. Bricanyl
10. Terbutalinum
11. Terbutalina [dcit]
12. Terbutalina
13. Terbutalinum [inn-latin]
14. 5-[2-(tert-butylamino)-1-hydroxyethyl]benzene-1,3-diol
15. Terbutalino [inn-spanish]
16. (+-)-terbutaline
17. Asthmasian
18. Terbutaline Hemisulfate
19. 5-(2-(tert-butylamino)-1-hydroxyethyl)benzene-1,3-diol
20. Terbutaline (inn)
21. 46719-29-3
22. 1,3-benzenediol, 5-[2-[(1,1-dimethylethyl)amino]-1-hydroxyethyl]-
23. Kwd-2019
24. Aerodur; Brican; Bricar
25. N8onu3l3pg
26. 5-(2-((1,1-dimethylethyl)amino)-1-hydroxyethyl)-1,3-benzenediol
27. Chebi:9449
28. 1,3-benzenediol, 5-(2-((1,1-dimethylethyl)amino)-1-hydroxyethyl)-
29. 23031-25-6 (free Base)
30. (+-)-5-(2-((1,1-dimethylethyl)amino)-1-hydroxyethyl)-1,3-benzenediol
31. Terbutalino
32. 5-{2-[(1,1-dimethylethyl)amino]-1-hydroxyethyl}benzene-1,3-diol
33. Terbutaline [inn]
34. 1,3-benzenediol, 5-(2-((1,1-dimethylethyl)amino)-1-hydroxyethyl)-, (+-)-
35. Terbutaline [inn:ban]
36. Asthmasian (tn)
37. Einecs 245-385-8
38. Unii-n8onu3l3pg
39. Brn 2370513
40. Sr-01000000174
41. Bricanyl (salt/mix)
42. Terbutaline,(+/-)
43. Brethaire (salt/mix)
44. Spectrum_001030
45. Terbutaline [mi]
46. Prestwick0_000391
47. Prestwick1_000391
48. Prestwick2_000391
49. Prestwick3_000391
50. Spectrum2_001327
51. Spectrum3_000593
52. Spectrum4_000870
53. Spectrum5_001262
54. Terbutaline [vandf]
55. Schembl4391
56. Chembl1760
57. Lopac0_001126
58. Oprea1_823972
59. Bspbio_000601
60. Bspbio_002066
61. Gtpl560
62. Kbiogr_001539
63. Kbioss_001510
64. Terbutaline [who-dd]
65. Divk1c_000078
66. Spbio_001453
67. Spbio_002522
68. Bpbio1_000663
69. Terbutaline Sulfate (salt/mix)
70. Dtxsid7021310
71. Bdbm25770
72. Hy-b0802a
73. Kbio1_000078
74. Kbio2_001510
75. Kbio2_004078
76. Kbio2_006646
77. Kbio3_001566
78. Ninds_000078
79. Benzyl Alcohol, Alpha-((t-butylamino)methyl)-3,5-dihydroxy-
80. Pdsp1_000158
81. Pdsp2_000157
82. Akos015889885
83. Ccg-205201
84. Db00871
85. Sdccgsbi-0051094.p005
86. Idi1_000078
87. Ncgc00016010-03
88. Ncgc00016010-04
89. Ncgc00016010-06
90. Ncgc00016010-16
91. Ncgc00089821-02
92. Sbi-0051094.p004
93. Ab00053548
94. Cs-0013822
95. Ft-0674864
96. C07129
97. D08570
98. Ab00053548_15
99. 031t256
100. A816514
101. L000594
102. Q424340
103. J-014950
104. Sr-01000000174-2
105. 1-(3',5'-dihydroxyphenyl)-2-(t-butylamino)-ethanol
106. Brd-a50157456-065-02-4
107. Brd-a50157456-065-12-3
108. 5-[2-(tert-butylamino)-1-hydroxyethyl]-1,3-benzenediol #
109. 5-[2-(tert-butylamino)-1-oxidanyl-ethyl]benzene-1,3-diol
110. 5-[2-(tert-butylamino)-1-hydroxyethyl]benzene-1,3-diol, 5
111. Benzyl Alcohol, .alpha.-((t-butylamino)methyl)-3,5-dihydroxy-
112. (rs)-5-(2-(tert-butylamino)-1-hydroxyethyl)benzene-1,3-diol
113. 1164563-86-3
| Molecular Weight | 225.28 g/mol |
|---|---|
| Molecular Formula | C12H19NO3 |
| XLogP3 | 0.9 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 225.13649347 g/mol |
| Monoisotopic Mass | 225.13649347 g/mol |
| Topological Polar Surface Area | 72.7 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 205 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Terbutaline sulfate |
| Drug Label | Terbutaline Sulfate, USP, the active ingredient of Terbutaline Sulfate Injection, USP, is a betaadrenergic agonist bronchodilator available as a sterile, nonpyrogenic, aqueous solution in vials, for subcutaneous administration. Each milliliter of... |
| Active Ingredient | Terbutaline sulfate |
| Dosage Form | Tablet; Injectable |
| Route | Injection; Oral |
| Strength | 2.5mg; 1mg/ml; 5mg |
| Market Status | Prescription |
| Company | Bedford; Hikma Farmaceutica; United Biomedcl; Fresenius Kabi Usa; Lannett; Impax Labs; Akorn |
| 2 of 2 | |
|---|---|
| Drug Name | Terbutaline sulfate |
| Drug Label | Terbutaline Sulfate, USP, the active ingredient of Terbutaline Sulfate Injection, USP, is a betaadrenergic agonist bronchodilator available as a sterile, nonpyrogenic, aqueous solution in vials, for subcutaneous administration. Each milliliter of... |
| Active Ingredient | Terbutaline sulfate |
| Dosage Form | Tablet; Injectable |
| Route | Injection; Oral |
| Strength | 2.5mg; 1mg/ml; 5mg |
| Market Status | Prescription |
| Company | Bedford; Hikma Farmaceutica; United Biomedcl; Fresenius Kabi Usa; Lannett; Impax Labs; Akorn |
Terbutaline is indicated for prevention and reversal of bronchospasm in patients at least 12 years old, with asthma and reversible bronchospasm associated with bronchitis and emphysema.
Terbutaline is a beta-2 adrenergic receptor agonist indicated to treat reversibly bronchospasm in asthmatic patients with bronchitis and emphysema. It has a short duration as the inhaled form is taken up to three times daily, and the therapeutic window is wide.
Bronchodilator Agents
Agents that cause an increase in the expansion of a bronchus or bronchial tubes. (See all compounds classified as Bronchodilator Agents.)
Adrenergic beta-2 Receptor Agonists
Compounds bind to and activate ADRENERGIC BETA-2 RECEPTORS. (See all compounds classified as Adrenergic beta-2 Receptor Agonists.)
Sympathomimetics
Drugs that mimic the effects of stimulating postganglionic adrenergic sympathetic nerves. Included here are drugs that directly stimulate adrenergic receptors and drugs that act indirectly by provoking the release of adrenergic transmitters. (See all compounds classified as Sympathomimetics.)
Tocolytic Agents
Drugs that prevent preterm labor and immature birth by suppressing uterine contractions (TOCOLYSIS). Agents used to delay premature uterine activity include magnesium sulfate, beta-mimetics, oxytocin antagonists, calcium channel inhibitors, and adrenergic beta-receptor agonists. The use of intravenous alcohol as a tocolytic is now obsolete. (See all compounds classified as Tocolytic Agents.)
R - Respiratory system
R03 - Drugs for obstructive airway diseases
R03A - Adrenergics, inhalants
R03AC - Selective beta-2-adrenoreceptor agonists
R03AC03 - Terbutaline
R - Respiratory system
R03 - Drugs for obstructive airway diseases
R03C - Adrenergics for systemic use
R03CC - Selective beta-2-adrenoreceptor agonists
R03CC03 - Terbutaline
Absorption
A 0.5 mg subcutaneous dose of terbutaline reaches a mean Cmax of 9.6 ng/mL, with a median Tmax of 0.5 hours, and a mean AUC of 29.4 14.2 h\*ng/mL. A 5 mg oral terbutaline tablet reaches a mean Cmax of 8.3 3.9 ng/mL with a median Tmax of 2 hours, and a mean AUC of 54.6 26.8 h\*ng/mL. A 5 mg oral terbutaline solution reaches a mean Cmax of 8.6 3.6 ng/mL, with a median Tmax of 1.5 hours, and a mean AUC of 53.1 23.5 h\*ng/mL. Oral terbutaline has an oral bioavailability of 14-15%.
Route of Elimination
An oral dose of terbutaline is 40% eliminated in the urine after 72 hours. The major metabolite in the urine was the sulphate conjugated form of terbutaline. Parenteral doses of terbutaline are 90% eliminated in the urine, with approximately 2/3 as the unchanged parent drug. Less than 1% of a dose of terbutaline is eliminated in the feces.
Volume of Distribution
Terbutaline has a mean volume of distribution of 1.6 L/kg.
Clearance
The average clearance of terbutaline is 3.0 mL/min/kg.
Terbutaline is sulphated or glucuronidated prior to elimination.
An oral dose of terbutaline has an elimination half life of 3.4 hours, while a subcutaneous dose has an elimination half life of 2.9 hours.
Terbutaline is a selective beta-2 adrenergic receptor agonist. Agonism of these receptors in bronchioles activates adenylyl cyclase, increasing intracellular cyclic adenosine monophosphate (cAMP). Increased cAMP decreases intracellular calcium, activating protein kinase A, inactivating myosin light-chain kinase, activating myosin light-chain phosphatase, and finally relaxing smooth muscle in the bronchiole.
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
30
PharmaCompass offers a list of Terbutaline API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Terbutaline manufacturer or Terbutaline supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Terbutaline manufacturer or Terbutaline supplier.
A Terbutaline manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Terbutaline, including repackagers and relabelers. The FDA regulates Terbutaline manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Terbutaline API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Terbutaline supplier is an individual or a company that provides Terbutaline active pharmaceutical ingredient (API) or Terbutaline finished formulations upon request. The Terbutaline suppliers may include Terbutaline API manufacturers, exporters, distributors and traders.
Terbutaline Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Terbutaline GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Terbutaline GMP manufacturer or Terbutaline GMP API supplier for your needs.
A Terbutaline CoA (Certificate of Analysis) is a formal document that attests to Terbutaline's compliance with Terbutaline specifications and serves as a tool for batch-level quality control.
Terbutaline CoA mostly includes findings from lab analyses of a specific batch. For each Terbutaline CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Terbutaline may be tested according to a variety of international standards, such as European Pharmacopoeia (Terbutaline EP), Terbutaline JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Terbutaline USP).

