
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
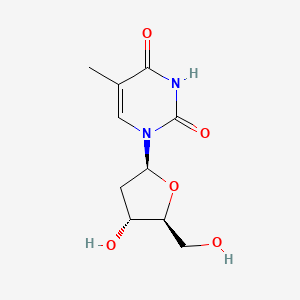

1. 1-(2-deoxy-beta-l-erythropentafuranosyl)-5-methyl-2,4(1h,3h)-pyrimidinedione
2. Beta L 2' Deoxythymidine
3. Beta-l-2'-deoxythymidine
4. Telbivudin
5. Tyzeka
1. 3424-98-4
2. 2'-deoxy-l-thymidine
3. Epavudine
4. L-thymidine
5. Beta-l-thymidine
6. Tyzeka
7. L-dt
8. Telbivudin
9. Sebivo
10. L-deoxythymidine
11. Nv-02b
12. Ldt600
13. Beta-l-2'-deoxythymidine
14. 26879-47-0
15. Ldt
16. Nv 02b
17. Ldt-600
18. Chebi:63624
19. 1-(2-deoxy-beta-l-erythro-pentofuranosyl)-5-methylpyrimidine-2,4(1h,3h)-dione
20. 2oc4hkd3sf
21. 1-((2s,4r,5s)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1h,3h)-dione
22. 1-(2-deoxy-beta-l-ribofuranosyl)-5-methyluracil
23. Telbivudine [usan]
24. 1-[(2s,4r,5s)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-methyl-1,2,3,4-tetrahydropyrimidine-2,4-dione
25. Mfcd02683612
26. Llt
27. Tyzeka (tn)
28. Unii-2oc4hkd3sf
29. Telbivudine (usan/inn)
30. Telbivudine [usan:inn:ban]
31. 1-[(2s,4r,5s)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione
32. Telbivudine [mi]
33. B-l-2'-deoxy-thymidine
34. Telbivudine [inn]
35. Telbivudine [vandf]
36. Telbivudine [mart.]
37. Telbivudine [who-dd]
38. Schembl124279
39. Telbivudine (sebivo, Tyzeka)
40. Telbivudine [ema Epar]
41. Chembl374731
42. Epavudine;l-thymidine;nv 02b
43. Thymine, 1-(2-deoxy-beta-l-erythro-pentofuranosyl)-
44. Zinc2159
45. Telbivudine, >=98% (hplc)
46. Dtxsid30187813
47. Telbivudine [orange Book]
48. Amy23971
49. Hy-b0017
50. Bdbm50088372
51. S1651
52. Akos025117349
53. Ac-5632
54. Ccg-266887
55. Cs-1934
56. Db01265
57. 2,4(1h,3h)-pyrimidinedione, 1-(2-deoxy-b-l-erythro-pentofuranosyl)-5-methyl-
58. 2,4(1h,3h)-pyrimidinedione, 1-(2-deoxy-beta-l-erythro-pentofuranosyl)-5-methyl-
59. Ncgc00346560-01
60. Ncgc00346560-06
61. Ncgc00346560-12
62. 1-[(2s,4r,5s)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-5-methyl-pyrimidine-2,4-dione
63. As-35209
64. Sw220236-1
65. D06675
66. Ab01274717-01
67. Ab01274717_02
68. Q413621
69. Brd-k15976406-001-01-7
70. Thymine, 1-(2-deoxy-.beta.-l-erythro-pentofuranosyl)-
71. Z2574360267
72. 1-[(2r,4r,5s)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-methylpyrimidine-2,4-dione
73. 1-[(2s,4r,5s)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-methyl-pyrimidine-2,4-dione
74. 2,4(1h,3h)-pyrimidinedione,1-(2-deoxy-.beta.-l-erythro-pentofuranosyl)-5-methyl-
75. 1-((2s,4s,5s)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)-5-methylpyrimidine-2,4(1h,3h)-dione
76. L-thymidine; Beta-l-thymidine; L-dt; 2'-deoxy-l-thymidine; 1-(2-deoxy-beta-l-erythro-pentofuranosyl)-5-methylpyrimidine-2,4(1h,3h)-dione
| Molecular Weight | 242.23 g/mol |
|---|---|
| Molecular Formula | C10H14N2O5 |
| XLogP3 | -1.2 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 242.09027155 g/mol |
| Monoisotopic Mass | 242.09027155 g/mol |
| Topological Polar Surface Area | 99.1 Ų |
| Heavy Atom Count | 17 |
| Formal Charge | 0 |
| Complexity | 381 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Tyzeka |
| PubMed Health | Telbivudine (By mouth) |
| Drug Classes | Antiviral |
| Drug Label | Tyzeka is the trade name for telbivudine, a synthetic thymidine nucleoside analogue with activity against hepatitisB virus (HBV). The chemical name for telbivudine is 1-((2S,4R,5S)-4-hydroxy-5-hydroxymethyltetrahydrofuran-2-y1)-5-methyl-1H-pyrimidi... |
| Active Ingredient | Telbivudine |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 600mg |
| Market Status | Prescription |
| Company | Novartis |
| 2 of 2 | |
|---|---|
| Drug Name | Tyzeka |
| PubMed Health | Telbivudine (By mouth) |
| Drug Classes | Antiviral |
| Drug Label | Tyzeka is the trade name for telbivudine, a synthetic thymidine nucleoside analogue with activity against hepatitisB virus (HBV). The chemical name for telbivudine is 1-((2S,4R,5S)-4-hydroxy-5-hydroxymethyltetrahydrofuran-2-y1)-5-methyl-1H-pyrimidi... |
| Active Ingredient | Telbivudine |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 600mg |
| Market Status | Prescription |
| Company | Novartis |
For the treatment of chronic hepatitis B in adult and adolescent patients 16 years of age with evidence of viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
Sebivo is indicated for the treatment of chronic hepatitis B in adult patients with compensated liver disease and evidence of viral replication, persistently elevated serum alanine aminotransferase (ALT) levels and histological evidence of active inflammation and/or fibrosis.
Initiation of Sebivo treatment should only be considered when the use of an alternative antiviral agent with a higher genetic barrier to resistance is not available or appropriate.
Telbivudine is a synthetic thymidine nucleoside analogue with activity against hepatitis B virus (HBV). Telbivudine is the unmodified L enantiomer of the naturally occurring nucleoside, thymidine. It undergoes phosphorylation via interaction with cellular kinases to form the active metabolite, telbivudine 5'-triphosphate.
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)
Nucleic Acid Synthesis Inhibitors
Compounds that inhibit cell production of DNA or RNA. (See all compounds classified as Nucleic Acid Synthesis Inhibitors.)
J05AF11
J - Antiinfectives for systemic use
J05 - Antivirals for systemic use
J05A - Direct acting antivirals
J05AF - Nucleoside and nucleotide reverse transcriptase inhibitors
J05AF11 - Telbivudine
Absorption
Absorbed following oral administration. Telbivudine absorption and exposure were unaffected when a single 600mg dose was administered with a highfat (~55 g), highcalorie (~950 kcal) meal.
Route of Elimination
Telbivudine is eliminated primarily by urinary excretion of unchanged drug.
Clearance
7.6 +/- 2.9 L/h [Normal renal function (Clcr>80 mL/min)]
5.0 +/- 1.2 L/h [Mild renal function impairement (Clcr=50-80 mL/min)]
2.6 +/- 1.2 L/h [Moderate renal function impairement (Clcr=30-49 mL/min)]
0.7 +/- 0.4 L/h [Severe renal function impairement (Clcr<30 mL/min)]
No metabolites of telbivudine were detected following administration of [14C]telbivudine in humans. Telbivudine is not a substrate, or inhibitor of the cytochrome P450 (CYP450) enzyme system.
Approximately 15 hours.
Telbivudine 5'triphosphate inhibits HBV DNA polymerase (reverse transcriptase) by competing with the natural substrate, thymidine 5'triphosphate. This leads to the chain termination of DNA synthesis, thereby inhibiting viral replication. Incorporation of telbivudine 5'triphosphate into viral DNA also causes DNA chain termination, resulting in inhibition of HBV replication. Telbivudine inhibits anticompliment or second-strand DNA.
Related Excipient Companies

Excipients by Applications
Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
33
PharmaCompass offers a list of Telbivudine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Telbivudine manufacturer or Telbivudine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Telbivudine manufacturer or Telbivudine supplier.
A Telbivudine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Telbivudine, including repackagers and relabelers. The FDA regulates Telbivudine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Telbivudine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Telbivudine supplier is an individual or a company that provides Telbivudine active pharmaceutical ingredient (API) or Telbivudine finished formulations upon request. The Telbivudine suppliers may include Telbivudine API manufacturers, exporters, distributors and traders.
click here to find a list of Telbivudine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Telbivudine DMF (Drug Master File) is a document detailing the whole manufacturing process of Telbivudine active pharmaceutical ingredient (API) in detail. Different forms of Telbivudine DMFs exist exist since differing nations have different regulations, such as Telbivudine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Telbivudine DMF submitted to regulatory agencies in the US is known as a USDMF. Telbivudine USDMF includes data on Telbivudine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Telbivudine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Telbivudine suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Telbivudine Drug Master File in Korea (Telbivudine KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Telbivudine. The MFDS reviews the Telbivudine KDMF as part of the drug registration process and uses the information provided in the Telbivudine KDMF to evaluate the safety and efficacy of the drug.
After submitting a Telbivudine KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Telbivudine API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Telbivudine suppliers with KDMF on PharmaCompass.
Telbivudine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Telbivudine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Telbivudine GMP manufacturer or Telbivudine GMP API supplier for your needs.
A Telbivudine CoA (Certificate of Analysis) is a formal document that attests to Telbivudine's compliance with Telbivudine specifications and serves as a tool for batch-level quality control.
Telbivudine CoA mostly includes findings from lab analyses of a specific batch. For each Telbivudine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Telbivudine may be tested according to a variety of international standards, such as European Pharmacopoeia (Telbivudine EP), Telbivudine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Telbivudine USP).

