
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF Dossiers
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
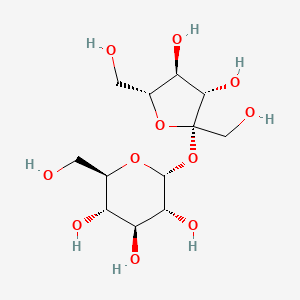

1. Saccharose
1. 57-50-1
2. Saccharose
3. Sugar
4. Table Sugar
5. Cane Sugar
6. White Sugar
7. D-sucrose
8. Saccharum
9. Rohrzucker
10. Microse
11. Beet Sugar
12. Granulated Sugar
13. Rock Candy
14. Amerfand
15. Amerfond
16. Confectioner's Sugar
17. D(+)-sucrose
18. Sucrose, Dust
19. Sucrose, Pure
20. D(+)-saccharose
21. Sacarosa
22. D-(+)-sucrose
23. Beta-d-fructofuranosyl Alpha-d-glucopyranoside
24. Beta-d-fructofuranosyl-alpha-d-glucopyranoside
25. D-saccharose
26. Sucraloxum [inn-latin]
27. D-(+)-saccharose
28. Ccris 2120
29. Hsdb 500
30. Sacharose
31. Alpha-d-glucopyranosyl Beta-d-fructofuranoside
32. Chebi:17992
33. Ai3-09085
34. (alpha-d-glucosido)-beta-d-fructofuranoside
35. Fructofuranoside, Alpha-d-glucopyranosyl, Beta-d
36. Glucopyranoside, Beta-d-fructofuranosyl, Alpha-d
37. (+)-sucrose
38. Alpha-d-glucopyranoside, Beta-d-fructofuranosyl-
39. Nci-c56597
40. Gne-410
41. S-67f
42. 1-alpha-d-glucopyranosyl-2-beta-d-fructofuranoside
43. Alpha-d-glucopyranoside, Beta-d-fructofuranosyl
44. C151h8m554
45. Nsc-406942
46. Beta-d-fruf-(2<->1)-alpha-d-glcp
47. Ncgc00164248-01
48. Sucraloxum
49. Dsstox_cid_1288
50. Nsc 406942
51. Dsstox_rid_76060
52. Dsstox_gsid_21288
53. Polysucrose
54. (2r,3r,4s,5s,6r)-2-[(2s,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)oxolan-2-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol
55. Sucrose [usan:jan]
56. Mfcd00006626
57. Cas-57-50-1
58. (2r,3r,4s,5s,6r)-2-{[(2s,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)oxolan-2-yl]oxy}-6-(hydroxymethyl)oxane-3,4,5-triol
59. Sucrose [jan:nf]
60. Einecs 200-334-9
61. Glc-(1-2)fru
62. Frost Sugar
63. Sucrose,ultrapure
64. Unii-c151h8m554
65. Compressible Sugar
66. Sucrose, Purified
67. Sucrose, Ar
68. Sucrose, Lr
69. Sucrose, Ultrapure
70. Sucrose, Usp
71. Sucrose Acs Grade
72. Sucrose (tn)
73. Sugar Spheres (nf)
74. Sugar,(s)
75. Refined Sugar
76. Sucrose, Acs Reagent
77. Sucrose, Reagent Grade
78. 1af6
79. Sugar, White
80. Sucrose [vandf]
81. Sucrose [hsdb]
82. Sucrose [inci]
83. Sucrose (jp17/nf)
84. Sucrose [fcc]
85. Sucrose [jan]
86. Sugar [vandf]
87. Glc(alpha1->2beta)fru
88. Sucrose [ii]
89. Sucrose [mi]
90. Sucrose [nf]
91. Sucrose Biochemical Grade
92. Sucrose [usp-rs]
93. Sucrose [who-dd]
94. Sucrose, Saj First Grade
95. Saccharum Officinale
96. Sugar, Compressible (nf)
97. Bmse000119
98. Bmse000804
99. Bmse000918
100. Epitope Id:153236
101. Sucrose, >=99.5%
102. Sucrose, Jis Special Grade
103. White Soft Sugar (jp17)
104. Sucrose, Analytical Standard
105. Sucrose, Cell Culture Tested
106. Sugar, Confectioner's (nf)
107. 1-alpha-d-glucopyranosyl-2-beta-d-fructofranoside
108. Sucrose [ep Impurity]
109. Sucrose, P.a., Acs Reagent
110. Sucrose [ep Monograph]
111. Chembl253582
112. Gtpl5411
113. Dtxsid2021288
114. Chebi:65313
115. Sucrose, 1.2m Aqueous Solution
116. Sucrose, Molecular Biology Grade
117. Sucrose, >=99.5% (gc)
118. Alpha-d-glc-(1-2)-beta-d-fru
119. Saccharum Officinale [hpus]
120. Hy-b1779
121. Zinc4217475
122. Tox21_112093
123. Tox21_201397
124. Tox21_300410
125. Bdbm50108105
126. S3598
127. Sucrose, For Electrophoresis, >99%
128. Akos024306988
129. Db02772
130. Sucrose, Bioxtra, >=99.5% (gc)
131. A-d-glucopyranosyl A-d-fructofuranoside
132. B -d-fructofuranosyl A-d-glucopyranoside
133. Ncgc00164248-02
134. Ncgc00164248-03
135. Ncgc00164248-05
136. Ncgc00254237-01
137. Ncgc00258948-01
138. 92004-84-7
139. Sucrose, Meets Usp Testing Specifications
140. Sucrose, Vetec(tm) Reagent Grade, 99%
141. D-saccharose 1000 Microg/ml In Methanol
142. Alpha-d-glucopyranosylbeta-d-fructofuranoside
143. Cs-0013810
144. S0111
145. Sucrose, Grade I, Plant Cell Culture Tested
146. Sucrose, Grade Ii, Plant Cell Culture Tested
147. C00089
148. D00025
149. D70407
150. Sucrose, For Molecular Biology, >=99.5% (gc)
151. Sucrose
152. ?-d-fructofuranosyl ?-d-glucopyranoside
153. Sr-01000883983
154. Sucrose, Nist(r) Srm(r) 17f, Optical Rotation
155. J-519846
156. Q4027534
157. Sr-01000883983-1
158. Sucrose, For Microbiology, Acs Reagent, >=99.0%
159. Alpha-d-glucopyranosyl-(1->2)-beta-d-fructofuranoside
160. Sucrose, British Pharmacopoeia (bp) Reference Standard
161. Sucrose, European Pharmacopoeia (ep) Reference Standard
162. Sucrose, Vetec(tm) Reagent Grade, Rnase And Dnase Free
163. Z1601554751
164. .beta.-d-fructofuranosyl-.alpha.-d-glucopyranoside
165. Beta-d-fructofuranosyl-(2↔1)-alpha-d-glucopyranoside
166. Sucrose, Analytical Standard, For Enzymatic Assay Kit Sca20
167. .alpha.-d-glucopyranoside, .beta.-d-fructofuranosyl-
168. Sucrose, Anhydrous, Free-flowing, Redi-dri(tm), Acs Reagent
169. Sucrose, Bioultra, For Molecular Biology, >=99.5% (hplc)
170. Sucrose, United States Pharmacopeia (usp) Reference Standard
171. Carbon Isotopes In Sucrose, Nist(r) Rm 8542, Iaea-ch-6 Sucrose
172. Sucrose (constituent Of Cranberry Liquid Preparation) [dsc]
173. Compressible Sugar, United States Pharmacopeia (usp) Reference Standard
174. Sucrose, Puriss., Meets Analytical Specification Of Ph. Eur., Bp, Nf
175. Wurcs=2.0/2,2,1/[a2122h-1a_1-5][ha122h-2b_2-5]/1-2/a1-b2
176. Wurcs=2.0/2,2,1/[ha122h-2b_2-5][a2122h-1a_1-5]/1-2/a2-b1
177. (2r,3r,4s,5s,6r)-2-(((2s,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)tetrahydrofuran-2-yl)oxy)-6-(hydroxymethyl)tetrahydro-2h-pyran-3,4,5-triol
178. (2r,3r,4s,5s,6r)-2-((2s,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)tetrahydrofuran-2-ylhydroxymethyl)tetrahydro-2h-pyran-3,4,5-triol
179. (2r,3r,4s,5s,6r)-2-((2s,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)tetrahydrofuran-2-yloxy)-6-(hydroxymethyl)tetrahydro-2h-pyran-3,4,5-triol
180. (2r,3r,4s,5s,6r)-2-[(2s,3s,4s,5r)-3,4-dihydroxy-2,5-bis(hydroxymethyl)tetrahydrofuran-2-yl]oxy-6-(hydroxymethyl)tetrahydropyran-3,4,5-triol
181. 8027-47-2
182. 8030-20-4
183. 85456-51-5
184. 86101-30-6
185. 87430-66-8
186. Sucrose, Bioreagent, Suitable For Cell Culture, Suitable For Insect Cell Culture, >=99.5% (gc)
187. Sucrose, Low Endotoxin, Pharmagrade, Usp/nf, Ph Eur, Manufactured Under Appropriate Gmp Controls For Pharma Or Biopharmaceutical Production.
| Molecular Weight | 342.30 g/mol |
|---|---|
| Molecular Formula | C12H22O11 |
| XLogP3 | -3.7 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 5 |
| Exact Mass | 342.11621151 g/mol |
| Monoisotopic Mass | 342.11621151 g/mol |
| Topological Polar Surface Area | 190 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 395 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 9 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
MEDICATION (VET): ITS ORAL USE IN ACETONEMIA OF CATTLE IS CONTROVERSIAL BUT STILL POPULAR. IT IS OFTEN USED ORALLY WITH VINEGAR...IN EMERGENCY TREATMENT OF ACUTE UREA TOXICITY IN CATTLE. DAILY ORAL USE CAN OCCASIONALLY REDUCE INCIDENCE OF BLOAT IN CATTLE ON SPRING PASTURE LOW IN CARBOHYDRATES.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 561
MEDICATION (VET): 20% ORAL SOLN CAUSE REFLEX CLOSURE OF ESOPHAGEAL GROOVE. ... SUGAR IS OCCASIONALLY USED...TO FACILITATE REDN OF EDEMA & EASE OF REPLACEMENT OF PROLAPSED UTERI IN COWS, EWES, BITCHES, ETC, & HAS BEEN USED LOCALLY ON WOUNDS, FOOT ROT, ETC BY LAYMEN & PROFESSIONALS.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 561
IV ADMIN OF HYPERTONIC SOLN OF SUCROSE HAS BEEN EMPLOYED CHIEFLY TO INITIATE OSMOTIC DIURESIS. SUCH PROCEDURE IS NOT COMPLETELY SAFE & RENAL TUBULAR DAMAGE MAY RESULT, PARTICULARLY IN PATIENTS WITH EXISTING RENAL PATHOLOGY. SAFER & MORE EFFECTIVE DIURETICS ARE AVAILABLE. /FORMER USE/
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 1235
VET: ORAL ADMIN TO YOUNG ANIMALS UNABLE TO DIGEST IT WILL CAUSE OR ACCENT DIARRHEAS.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 562
1. 1= PRACTICALLY NONTOXIC: PROBABLE ORAL LETHAL DOSE (HUMAN) ABOVE 15 G/KG, MORE THAN 1 QUART (2.2 LB) FOR 70 KG PERSON (150 LB).
Gosselin, R.E., H.C. Hodge, R.P. Smith, and M.N. Gleason. Clinical Toxicology of Commercial Products. 4th ed. Baltimore: Williams and Wilkins, 1976., p. II-151
Sweetening Agents
Substances that sweeten food, beverages, medications, etc., such as sugar, saccharine or other low-calorie synthetic products. (From Random House Unabridged Dictionary, 2d ed) (See all compounds classified as Sweetening Agents.)
 Pfanstiehl, a global leader in the manufacture of cGMP high purity, low endotoxin, low metals (HPLE-LM)TM injectable grade excipients.
Pfanstiehl, a global leader in the manufacture of cGMP high purity, low endotoxin, low metals (HPLE-LM)TM injectable grade excipients.
 Laboratorium Ofichem, offering a flexible, high-tech environment to produce broad range of APIs for Human & Vet pharmaceutical markets.
Laboratorium Ofichem, offering a flexible, high-tech environment to produce broad range of APIs for Human & Vet pharmaceutical markets.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26737
Submission : 2012-12-10
Status : Active
Type : IV


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 33289
Submission : 2018-10-26
Status : Active
Type : IV


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35209
Submission : 2020-09-14
Status : Active
Type : IV


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23812
Submission : 2010-05-12
Status : Active
Type : IV


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26713
Submission : 2012-11-08
Status : Active
Type : IV


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38742
Submission : 2023-08-18
Status : Active
Type : IV


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 35209
Submission : 2020-09-14
Status : Active
Type : IV

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 38742
Submission : 2023-08-18
Status : Active
Type : IV

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26737
Submission : 2012-12-10
Status : Active
Type : IV

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26713
Submission : 2012-11-08
Status : Active
Type : IV

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23812
Submission : 2010-05-12
Status : Active
Type : IV

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 33289
Submission : 2018-10-26
Status : Active
Type : IV

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
41
PharmaCompass offers a list of Sucrose API API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sucrose API manufacturer or Sucrose API supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Sucrose API manufacturer or Sucrose API supplier.
A Sucrose API manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Sucrose API, including repackagers and relabelers. The FDA regulates Sucrose API manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Sucrose API API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Sucrose API manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Sucrose API supplier is an individual or a company that provides Sucrose API active pharmaceutical ingredient (API) or Sucrose API finished formulations upon request. The Sucrose API suppliers may include Sucrose API API manufacturers, exporters, distributors and traders.
click here to find a list of Sucrose API suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Sucrose API DMF (Drug Master File) is a document detailing the whole manufacturing process of Sucrose API active pharmaceutical ingredient (API) in detail. Different forms of Sucrose API DMFs exist exist since differing nations have different regulations, such as Sucrose API USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Sucrose API DMF submitted to regulatory agencies in the US is known as a USDMF. Sucrose API USDMF includes data on Sucrose API's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Sucrose API USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Sucrose API suppliers with USDMF on PharmaCompass.
Sucrose API Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Sucrose API GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Sucrose API GMP manufacturer or Sucrose API GMP API supplier for your needs.
A Sucrose API CoA (Certificate of Analysis) is a formal document that attests to Sucrose API's compliance with Sucrose API specifications and serves as a tool for batch-level quality control.
Sucrose API CoA mostly includes findings from lab analyses of a specific batch. For each Sucrose API CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Sucrose API may be tested according to a variety of international standards, such as European Pharmacopoeia (Sucrose API EP), Sucrose API JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Sucrose API USP).

