
Synopsis
Synopsis
0
KDMF
0
VMF
0
FDA Orange Book
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
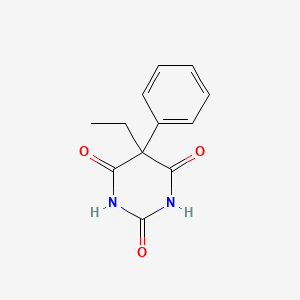

1. Acid, Phenylethylbarbituric
2. Gardenal
3. Hysteps
4. Luminal
5. Monosodium Salt Phenobarbital
6. Phenemal
7. Phenobarbital Sodium
8. Phenobarbital, Monosodium Salt
9. Phenobarbitone
10. Phenylbarbital
11. Phenylethylbarbituric Acid
12. Sodium, Phenobarbital
1. Phenobarbitone
2. Luminal
3. Phenobarbitol
4. 50-06-6
5. Phenylethylbarbiturate
6. Phenobarbituric Acid
7. Phenylethylmalonylurea
8. Fenobarbital
9. Gardenal
10. Phenemal
11. Adonal
12. Phenylethylbarbituric Acid
13. Nunol
14. Gardepanyl
15. Neurobarb
16. Phenaemal
17. Dormiral
18. Hysteps
19. Aphenylbarbit
20. Aphenyletten
21. Dezibarbitur
22. Lepinaletten
23. Lumofridetten
24. Aephenal
25. Agrypnal
26. Amylofene
27. Barbenyl
28. Barbiphenyl
29. Barbipil
30. Barbivis
31. Barbonal
32. Barbophen
33. Bialminal
34. Cabronal
35. Calmetten
36. Calminal
37. Cardenal
38. Codibarbita
39. Coronaletta
40. Cratecil
41. Doscalun
42. Ensobarb
43. Ensodorm
44. Episedal
45. Epsylone
46. Eskabarb
47. Fenbital
48. Fenylettae
49. Glysoletten
50. Haplopan
51. Hennoletten
52. Hypnaletten
53. Hypnette
54. Hypnogen
55. Hypnolone
56. Hypnoltol
57. Liquital
58. Lixophen
59. Lubergal
60. Lubrokal
61. Lumesettes
62. Luphenil
63. Nirvonal
64. Parkotal
65. Pharmetten
66. Phenemalum
67. Phenobal
68. Phenobarbyl
69. Phenoluric
70. Phenolurio
71. Phenomet
72. Phenonyl
73. Phenoturic
74. Phenyletten
75. Phenyral
76. Polcominal
77. Promptonal
78. Sedizorin
79. Sedonettes
80. Sedophen
81. Solfoton
82. Sombutol
83. Somnolens
84. Somnoletten
85. Somnosan
86. Spasepilin
87. Starifen
88. Starilettae
89. Teolaxin
90. Versomnal
91. Barbita
92. Bardorm
93. Bartol
94. Chinoin
95. Duneryl
96. Epanal
97. Epidorm
98. Epilol
99. Etilfen
100. Euneryl
101. Fenemal
102. Fenosed
103. Haplos
104. Henotal
105. Leonal
106. Lepinal
107. Linasen
108. Lumesyn
109. Luramin
110. Molinal
111. Noptil
112. Sedabar
113. Sedicat
114. Sedlyn
115. Sedofen
116. Sedonal
117. Sevenal
118. Somonal
119. Lumen
120. Seda-tablinen
121. 5-ethyl-5-phenylbarbituric Acid
122. Blu-phen
123. Nova-pheno
124. Solu-barb
125. Hypno-tablinetten
126. Stental Extentabs
127. Phen-bar
128. Phob
129. Phenobarb
130. Talpheno
131. Triabarb
132. Triphenatol
133. 5-phenyl-5-ethylbarbituric Acid
134. Phenobarbitalum
135. Tridezibarbitur
136. Zadoletten
137. Barbinal
138. Barbiphen
139. Damoral
140. Dormina
141. Lefebar
142. Lephebar
143. Stental
144. Teoloxin
145. Theoloxin
146. Zadonal
147. 5-ethyl-5-phenylpyrimidine-2,4,6(1h,3h,5h)-trione
148. Sk-phenobarbital
149. Thenobarbital
150. 2,4,6(1h,3h,5h)-pyrimidinetrione, 5-ethyl-5-phenyl-
151. 5-ethyl-5-phenyl-1,3-diazinane-2,4,6-trione
152. Dormital
153. Tedral
154. Phenylethylbarbitursaeure
155. Phenylaethylbarbitursaeure
156. 5-ethyl-5-phenyl-2,4,6(1h,3h,5h)-pyrimidinetrione
157. Barbituric Acid, 5-ethyl-5-phenyl-
158. 5-ethyl-5-phenyl-pyrimidine-2,4,6-trione
159. Barbilehae (barbilettae)
160. Component Of Tedral
161. Sedonal (sedative)
162. Component Of Slowten
163. Chebi:8069
164. Component Of Antrocol
165. Component Of Hecadrol
166. Component Of Bronkotabs
167. 5-ethyl-5-phenyl-2,4,6-(1h,3h,5h)pyrimidinetrione
168. Phenobarbital Civ
169. Component Of Primatene P
170. Nsc-9848
171. Chembl40
172. Phenobarbital (in Methanol Solution)
173. Nsc-128143
174. Component Of Valpin 50-pb
175. Yqe403bp4d
176. Austrominal
177. Phenobarbitonum
178. Ncgc00159493-02
179. Fenobarbitale
180. Fenobarbitale [dcit]
181. Elixir Of Phenobarbital
182. Phenobarbitalum [inn]
183. Dsstox_cid_1122
184. Chardonna-2
185. Dsstox_rid_75953
186. Dsstox_gsid_21122
187. Phenylethyl Barbituric Acid
188. Fenobarbital [inn-spanish]
189. Phenyl-ethyl-barbituric Acid
190. Phenobarbitalum [inn-latin]
191. Wln: T6vmvmv Fhj F2 Fr
192. Barbinol
193. Fenemal Recip
194. Cas-50-06-6
195. Levsin Pb Drops And Tablets
196. Ccris 502
197. Luminal (tn)
198. Phenobarbital (pb)
199. Acido 5-fenil-5-etilbarbiturico
200. Hsdb 3157
201. Acido 5-fenil-5-etilbarbiturico [italian]
202. 5-ethyl-5-phenyl-2,6(1h,3h,5h)-pyrimidinetrione
203. Einecs 200-007-0
204. Unii-yqe403bp4d
205. 2,6(1h,3h,5h)-pyrimidinetrione, 5-ethyl-5-phenyl-
206. Nsc 128143
207. Phenobar
208. Ai3-02726
209. Phenobarbital [usp:inn:ban:jan]
210. Tedral (salt/mix)
211. Primidone Impurity B
212. Tedrigen
213. Theophed
214. Kinesed (salt/mix)
215. Antrocol (salt/mix)
216. Donnatal (salt/mix)
217. Donnazyme (salt/mix)
218. Quadrinal (salt/mix)
219. Mephobarbital M (nor)
220. Barbidonna (salt/mix)
221. Bronkotabs (salt/mix)
222. Tedral Suspension
223. Chardonna-2 (salt/mix)
224. Phenobarbital [mi]
225. Epitope Id:116048
226. Phenobarbital [inn]
227. Phenobarbital [jan]
228. Phenobarbital [hsdb]
229. Phenobarbital [iarc]
230. Bidd:pxr0061
231. Oprea1_384816
232. Schembl16583
233. 5-ethyl-5-phenyl-hexahydropyrimidine-2,4,6-trione
234. Methylphenobarbital, M(nor-)
235. Mls001240232
236. Divk1c_000987
237. Phenobarbital [mart.]
238. Phenobarbitalum [hpus]
239. Phenobarbital [who-dd]
240. Phenobarbital [who-ip]
241. Gtpl2804
242. Dtxsid5021122
243. Schembl11114624
244. Hms503e15
245. Kbio1_000987
246. Nsc9848
247. Phenobarbital (jp17/usp/inn)
248. Ninds_000987
249. Hms2272g06
250. Phenobarbital [ep Impurity]
251. Phenobarbital Civ [usp-rs]
252. Phenobarbital [ep Monograph]
253. Tox21_111713
254. Tox21_200510
255. Bdbm50021437
256. Nsc128143
257. Phenobarbital [usp Monograph]
258. Stl367898
259. Zinc95588079
260. Component Of Primatene P (salt/mix)
261. Phenobarbital 0.1 Mg/ml In Methanol
262. Phenobarbital 1.0 Mg/ml In Methanol
263. Akos000605404
264. Akos015964976
265. Barbituric Acid, 5-ethyl-5-phenyl-,
266. Phenobarbitalum [who-ip Latin]
267. Ab02704
268. Db01174
269. Component Of Valpin 50-pb (salt/mix)
270. Idi1_000987
271. Nsc-128143-
272. Levsin Pb Drops And Tablets (salt/mix)
273. Ncgc00159493-03
274. Ncgc00159493-04
275. Ncgc00258064-01
276. Smr000058986
277. Primidone Impurity B [ep Impurity]
278. Db-051722
279. C07434
280. D00506
281. Phenobarbital Solution, 1.0 Mg/ml In Methanol
282. A827956
283. Q407241
284. Sr-01000313151
285. Methylphenobarbital Impurity A [ep Impurity]
286. Sr-01000313151-1
287. Phenobarbital, United States Pharmacopeia (usp) Reference Standard
288. Phenobarbital Solution, 1 Mg/ml In Methanol, Ampule Of 1 Ml, Certified Reference Material
289. 11097-06-6
290. Uqa
| Molecular Weight | 232.23 g/mol |
|---|---|
| Molecular Formula | C12H12N2O3 |
| XLogP3 | 1.5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Exact Mass | 232.08479225 g/mol |
| Monoisotopic Mass | 232.08479225 g/mol |
| Topological Polar Surface Area | 75.3 Ų |
| Heavy Atom Count | 17 |
| Formal Charge | 0 |
| Complexity | 339 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anticonvulsants; Carcinogens; Central Nervous System Depressants; Excitatory Amino Acid Antagonists; GABA Modulators; Sedatives, Barbiturate
National Library of Medicine's Medical Subject Headings online file (MeSH, 2009)
Phenobarbital is indicated for use as a sedative or as and anticonvulsant for the treatment of generalized and partial seizures.
Novak, K.M. (ed.). Drug Facts and Comparisons2008 Edition. Wolters Kluwer Health. St. Louis, Missouri 2008., p. 1443
/Phenobarbital is indicated/ as a hypnotic, for the short term management of of insomnia.
Novak, K.M. (ed.). Drug Facts and Comparisons2008 Edition. Wolters Kluwer Health. St. Louis, Missouri 2008., p. 1443
/Phenobarbital is indicated/ as a sedative, for the relief of anxiety, tension,and apprehension.
Novak, K.M. (ed.). Drug Facts and Comparisons2008 Edition. Wolters Kluwer Health. St. Louis, Missouri 2008., p. 1443
For more Therapeutic Uses (Complete) data for Phenobarbital (7 total), please visit the HSDB record page.
Barbiturates have been shown to cause an incr incidence of fetal abnormalities. Risk-benefit must be carefully considered when the medication is required in life-threatening situations or in serious diseases for which other medications cannot be used or are ineffective. /Barbiturates/
USP Convention. USPDI - Drug Information for the Health Care Professional. 17th ed. Volume I. Rockville, MD: Convention, Inc., 1997. (Plus Updates)., p. 502
Use of barbiturates throughout the last trimester of pregnancy may cause physical dependence with resulting withdrawal symptoms in the neonate. In infants suffering from long-term exposure in utero, the acute withdrawal syndrome of seizures and hyperirritability has been reported to occur from birth to a delayed onset of up to 14 days. /Barbiturates/
USP Convention. USPDI - Drug Information for the Health Care Professional. 17th ed. Volume I. Rockville, MD: Convention, Inc., 1997. (Plus Updates)., p. 502
Use of barbiturates during labor may cause respiratory depression in the neonate, esp the premature neonate, because of immature hepatic function. /Barbiturates/
USP Convention. USPDI - Drug Information for the Health Care Professional. 17th ed. Volume I. Rockville, MD: Convention, Inc., 1997. (Plus Updates)., p. 503
The risk of barbiturate-induced hypothermia may be increased in elderly patients, especially with high doses or in acute overdoses of barbiturates. In addition, elderly patients are more likely to have age-related hepatic or renal function impairment, which may require a reduction of dosage in patients receiving a barbiturate. /Barbiturates/
USP Convention. USPDI - Drug Information for the Health Care Professional. 17th ed. Volume I. Rockville, MD: Convention, Inc., 1997. (Plus Updates)., p. 503
For more Drug Warnings (Complete) data for Phenobarbital (28 total), please visit the HSDB record page.
The toxic dose of barbiturates varies considerably but, in general, a severe reaction is likely to occur when the amount ingested is more than 10 times the usual oral hypnotic dose. Potentially lethal blood concentrations are those in excess of 80 ug/mL for phenobarbital, 50 ug/mL for amobarbital or butabarbital, and approximately 30 ug/mL for secobarbital or pentobarbital; however, some patients have survived much higher blood concentrations. /Barbiturates General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2578
For the treatment of all types of seizures except absence seizures.
Treatment of epilepsy
Phenobarbital, the longest-acting barbiturate, is used for its anticonvulsant and sedative-hypnotic properties in the management of all seizure disorders except absence (petit mal).
GABA Modulators
Substances that do not act as agonists or antagonists but do affect the GAMMA-AMINOBUTYRIC ACID receptor-ionophore complex. GABA-A receptors (RECEPTORS, GABA-A) appear to have at least three allosteric sites at which modulators act: a site at which BENZODIAZEPINES act by increasing the opening frequency of GAMMA-AMINOBUTYRIC ACID-activated chloride channels; a site at which BARBITURATES act to prolong the duration of channel opening; and a site at which some steroids may act. GENERAL ANESTHETICS probably act at least partly by potentiating GABAergic responses, but they are not included here. (See all compounds classified as GABA Modulators.)
Cytochrome P-450 CYP2B6 Inducers
Drugs and compounds that induce the synthesis of CYTOCHROME P-450 CYP2B6. (See all compounds classified as Cytochrome P-450 CYP2B6 Inducers.)
Cytochrome P-450 CYP3A Inducers
Drugs and compounds that induce the synthesis of CYTOCHROME P-450 CYP3A. (See all compounds classified as Cytochrome P-450 CYP3A Inducers.)
Anticonvulsants
Drugs used to prevent SEIZURES or reduce their severity. (See all compounds classified as Anticonvulsants.)
Excitatory Amino Acid Antagonists
Drugs that bind to but do not activate excitatory amino acid receptors, thereby blocking the actions of agonists. (See all compounds classified as Excitatory Amino Acid Antagonists.)
Hypnotics and Sedatives
Drugs used to induce drowsiness or sleep or to reduce psychological excitement or anxiety. (See all compounds classified as Hypnotics and Sedatives.)
N03AA02
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
N - Nervous system
N03 - Antiepileptics
N03A - Antiepileptics
N03AA - Barbiturates and derivatives
N03AA02 - Phenobarbital
Absorption
Absorbed in varying degrees following oral, rectal or parenteral administration. The salts are more rapidly absorbed than are the acids. The rate of absorption is increased if the sodium salt is ingested as a dilute solution or taken on an empty stomach.
About 70-90% of an oral dose of phenobarbital is absorbed slowly from the GI tract. Following rectal administration of phenobarbital sodium, the drug is readily absorbed from the colon. Following oral administration of phenobarbital, peak blood concentrations are reached in 8-12 hours and peak brain concentrations in 10-15 hours. ... When phenobarbital sodium is administered IV, the onset of action usually occurs within 5 minutes and maximum effects are achieved within 30 minutes. IM or subcutaneous administration of phenobarbital sodium results in a slightly slower onset of action. The duration of action of parenterally administered phenobarbital sodium is usually 4-6 hours.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2269
Barbiturates are absorbed in varying degrees following oral, rectal, or im administration. The sodium salts are more rapidly absorbed by all routes of administration than are the acids. The rate of oral absorption is increased when the sodium salt is ingested as a dilute solution or taken on an empty stomach. Alcohol also enhances the rate of absorption, possibly by increasing blood flow through the gastric mucosa. /Barbiturates General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2579
Following oral or rectal administration, the onset of action varies from 10-30 minutes for amobarbital, aprobarbital, butabarbital, pentobarbital, and secobarbital and from 20-60 minutes for metharbital, mephobarbital, and phenobarbital. IM administration results in a slightly faster onset of action. Following iv administration of the sodium salts of amobarbital, pentobarbital, phenobarbital, or secobarbital, the onset of action ranges from almost immediately for methohexital, pentobarbital, and thiopental to 5 minutes for phenobarbital. Maximum effects of thiopental or pentobarbital are achieved within about 1 minute while as much as 30 minutes may be required with administration of phenobarbital. /Barbiturates General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2579
The duration of sedative effects of all the barbiturates is usually 3-6 hours following iv administration and 6-8 hours when the drugs are administered by other routes. There appears to be very little difference in duration of hypnotic action among barbiturates used orally as hypnotics. Therefore, most authorities now believe that barbiturates should be classified according to their intended pharmacologic action (ie, sedative-hypnotic barbiturates and anesthetic barbiturates [methohexital, thiamylal (no longer commercially available in the US), thiopental]), rather than as long-acting (mephobarbital, metharbital, and phenobarbital), intermediate-acting (amobarbital and butabarbital), short-acting (aprobarbital, pentobarbital, and secobarbital), and ultrashort-acting (methohexital, thiopental). /Barbiturates General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2579
For more Absorption, Distribution and Excretion (Complete) data for Phenobarbital (15 total), please visit the HSDB record page.
Hepatic (mostly via CYP2C19).
Phenobarbital is hydroxylated by the liver to form p-hydroxyphenobarbital, an inactive metabolite. Phenobarbital is a potent inducer of the enzymes involved in the metabolism of other drugs, but there is no conclusive evidence that phenobarbital accelerates its own metabolism.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2584
The inactive metabolites of the barbiturates are excreted as conjugates of glucuronic acid.
Medical Economics Co; Physicians Desk Reference: Generics 2nd ed p.2458 (1996)
Biotransformation occurs primarily by the hepatic microsomal enzyme system. /Barbiturates/
USP Convention. USPDI - Drug Information for the Health Care Professional. 17th ed. Volume I. Rockville, MD: Convention, Inc., 1997. (Plus Updates)., p. 502
Metabolites of phenobarbital sodium produced in rats and guinea pigs are 5-(3,4-dihydroxy-1,5-cyclohexadien-1-yl)-5-ethylbarbituric acid; 5-(1-hydroxyethyl)-5-phenylbarbituric acid; 5-(3,4-dihydroxyphenyl)-5-ethylbarbituric acid; and 5-(4-hydroxyphenyl)-5-ethylbarbituric acid ...
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V13 166 (1977)
For more Metabolism/Metabolites (Complete) data for Phenobarbital (6 total), please visit the HSDB record page.
Phenobarbital has known human metabolites that include p-Hydroxyphenobarbital.
Phenobarbital is a known human metabolite of (s)-mephobarbital.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
53 to 118 hours (mean 79 hours)
The plasma half-life for phenobarbital in adults ranges between 53 and 118 hours with an mean of 79 hours.
Medical Economics Co; Physicians Desk Reference: Generics 2nd ed p.2458 (1996)
The plasma half-life for phenobarbital in children and newborns (less than 48 hours old) ranges between 60 to 180 hours with a mean of 110 hours.
Medical Economics Co; Physicians Desk Reference: Generics 2nd ed p.2458 (1996)
Phenobarbital acts on GABAA receptors, increasing synaptic inhibition. This has the effect of elevating seizure threshold and reducing the spread of seizure activity from a seizure focus. Phenobarbital may also inhibit calcium channels, resulting in a decrease in excitatory transmitter release. The sedative-hypnotic effects of phenobarbital are likely the result of its effect on the polysynaptic midbrain reticular formation, which controls CNS arousal.
/Phenobarbital/ produces anticonvulsant effects in subhypnotic doses. The drug lowers serum bilirubin concentrations in neonates and patients with congenital nonhemolytic unconjugated hyperbilirubinemia and patients with chronic intrahepatic cholestasis, presumably by induction of glucuronyl transferase, the enzyme which conjugates bilirubin.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2584
The exact mechanism(s) by which barbiturates exert their effect on the CNS, has not been fully elucidated. However, it is believed that such effects are related, at least partially, to the drugs' ability to enhance the activity of gamma-aminobutyric acid (GABA), the principal inhibitory neurotransmitter in the CNS, by altering inhibitory synaptic transmissions that are mediated by GABAA receptors. /Barbiturates General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2579
Although the drugs act throughout the CNS, a site of particular sensitivity is the polysynaptic midbrain reticular formation which is concerned with the arousal mechanism. Barbiturates induce an imbalance in central inhibitory and facilitatory mechanisms influencing the cerebral cortex and the reticular formation. The significance of the effect of barbiturates on neurotransmitters is unclear. It appears that the drugs decrease the excitability of both presynaptic and postsynaptic membranes. It has not been determined which of the various actions of barbiturates at cellular and synaptic levels are responsible for their sedative and hypnotic effects. /Barbiturates General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2579
Relatively low doses of the barbiturates depress the sensory cortex, decrease motor activity, and produce sedation and drowsiness. In some patients, however, drowsiness may be preceded by a period of transient elation, confusion, euphoria, or excitement, especially after subhypnotic doses of aprobarbital, pentobarbital, or secobarbital. /Barbiturates General Statement/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2579
For more Mechanism of Action (Complete) data for Phenobarbital (12 total), please visit the HSDB record page.

Certificate Number : CEP 2022-432 - Rev 00
Issue Date : 2023-11-09
Type : Chemical
Substance Number : 201
Status : Valid

NDC Package Code : 65319-1450
Start Marketing Date : 2019-01-14
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27827
Submission : 2013-12-01
Status : Active
Type : II

Certificate Number : CEP 2017-269 - Rev 01
Issue Date : 2023-11-03
Type : Chemical
Substance Number : 201
Status : Valid

NDC Package Code : 58624-0520
Start Marketing Date : 1997-01-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 31321
Submission : 2017-02-09
Status : Active
Type : II

Certificate Number : R1-CEP 2005-118 - Rev 02
Issue Date : 2016-10-04
Type : Chemical
Substance Number : 201
Status : Valid

NDC Package Code : 59348-0011
Start Marketing Date : 2015-11-02
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 17823
Submission : 2004-11-10
Status : Active
Type : II

Certificate Number : CEP 2003-017 - Rev 04
Issue Date : 2024-08-22
Type : Chemical
Substance Number : 201
Status : Valid

Date of Issue : 2025-11-12
Valid Till : 2028-11-11
Written Confirmation Number : WC-0045
Address of the Firm :

NDC Package Code : 66064-2001
Start Marketing Date : 2005-01-20
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (25kg/25kg)
Marketing Category : BULK INGREDIENT



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 4642
Submission : 1982-09-01
Status : Active
Type : II

Certificate Number : R1-CEP 2009-132 - Rev 03
Issue Date : 2021-12-08
Type : Chemical
Substance Number : 201
Status : Valid

NDC Package Code : 50095-001
Start Marketing Date : 1982-11-05
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (25kg/25kg)
Marketing Category : BULK INGREDIENT


USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 1003
Submission : 1966-10-19
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 4714
Submission : 1982-11-10
Status : Inactive
Type : II

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3756
Submission : 1980-02-28
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
47
PharmaCompass offers a list of Phenobarbital API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Phenobarbital manufacturer or Phenobarbital supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Phenobarbital manufacturer or Phenobarbital supplier.
A Solfoton talpheno manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Solfoton talpheno, including repackagers and relabelers. The FDA regulates Solfoton talpheno manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Solfoton talpheno API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Solfoton talpheno manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Solfoton talpheno supplier is an individual or a company that provides Solfoton talpheno active pharmaceutical ingredient (API) or Solfoton talpheno finished formulations upon request. The Solfoton talpheno suppliers may include Solfoton talpheno API manufacturers, exporters, distributors and traders.
click here to find a list of Solfoton talpheno suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Solfoton talpheno DMF (Drug Master File) is a document detailing the whole manufacturing process of Solfoton talpheno active pharmaceutical ingredient (API) in detail. Different forms of Solfoton talpheno DMFs exist exist since differing nations have different regulations, such as Solfoton talpheno USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Solfoton talpheno DMF submitted to regulatory agencies in the US is known as a USDMF. Solfoton talpheno USDMF includes data on Solfoton talpheno's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Solfoton talpheno USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Solfoton talpheno suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Solfoton talpheno Drug Master File in Japan (Solfoton talpheno JDMF) empowers Solfoton talpheno API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Solfoton talpheno JDMF during the approval evaluation for pharmaceutical products. At the time of Solfoton talpheno JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Solfoton talpheno suppliers with JDMF on PharmaCompass.
A Solfoton talpheno CEP of the European Pharmacopoeia monograph is often referred to as a Solfoton talpheno Certificate of Suitability (COS). The purpose of a Solfoton talpheno CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Solfoton talpheno EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Solfoton talpheno to their clients by showing that a Solfoton talpheno CEP has been issued for it. The manufacturer submits a Solfoton talpheno CEP (COS) as part of the market authorization procedure, and it takes on the role of a Solfoton talpheno CEP holder for the record. Additionally, the data presented in the Solfoton talpheno CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Solfoton talpheno DMF.
A Solfoton talpheno CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Solfoton talpheno CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Solfoton talpheno suppliers with CEP (COS) on PharmaCompass.
A Solfoton talpheno written confirmation (Solfoton talpheno WC) is an official document issued by a regulatory agency to a Solfoton talpheno manufacturer, verifying that the manufacturing facility of a Solfoton talpheno active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Solfoton talpheno APIs or Solfoton talpheno finished pharmaceutical products to another nation, regulatory agencies frequently require a Solfoton talpheno WC (written confirmation) as part of the regulatory process.
click here to find a list of Solfoton talpheno suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Solfoton talpheno as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Solfoton talpheno API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Solfoton talpheno as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Solfoton talpheno and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Solfoton talpheno NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Solfoton talpheno suppliers with NDC on PharmaCompass.
Solfoton talpheno Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Solfoton talpheno GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Solfoton talpheno GMP manufacturer or Solfoton talpheno GMP API supplier for your needs.
A Solfoton talpheno CoA (Certificate of Analysis) is a formal document that attests to Solfoton talpheno's compliance with Solfoton talpheno specifications and serves as a tool for batch-level quality control.
Solfoton talpheno CoA mostly includes findings from lab analyses of a specific batch. For each Solfoton talpheno CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Solfoton talpheno may be tested according to a variety of international standards, such as European Pharmacopoeia (Solfoton talpheno EP), Solfoton talpheno JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Solfoton talpheno USP).

