
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
DRUG PRODUCT COMPOSITIONS
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Jevtana
2. Kabazitaxel
1. 183133-96-2
2. Jevtana
3. Taxoid Xrp6258
4. Txd 258
5. Xrp-6258
6. Cabazitaxelum
7. Xrp6258
8. Xrp 6258
9. Jevtana (tn)
10. Nsc-761432
11. Chebi:63584
12. Txd-258
13. 51f690397j
14. Kabazitaxel
15. Jevtana Kit
16. Cabazitaxel (jevtana)
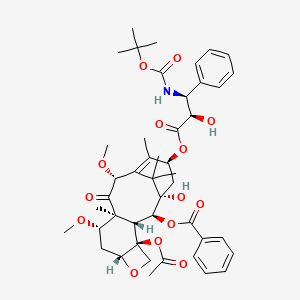
17. (2ar,4s,4as,6r,9s,11s,12s,12ar,12bs)-12b-acetoxy-9-(((2r,3s)-3-((tert-butoxycarbonyl)amino)-2-hydroxy-3-phenylpropanoyl)oxy)-11-hydroxy-4,6-dimethoxy-4a,8,13,13-tetramethyl-5-oxo-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-1h-7,11-methanocyclodeca[3,4]benzo[1,2-b]oxet-12-yl Benzoate.
18. Cabazitaxel Acetonate
19. Cabazitaxel Acetonate [jan]
20. Cabazitaxel Injection
21. Cabazitaxel [usan:inn]
22. Jevanta
23. Txd258
24. Unii-51f690397j
25. Rpr 116258a
26. Rpr-116258a
27. Cabazitaxel [mi]
28. Cabazitaxel [inn]
29. Cabazitaxel (usan/inn)
30. Cabazitaxel [usan]
31. Cabazitaxel [vandf]
32. Cabazitaxel [mart.]
33. Cabazitaxel [who-dd]
34. Schembl179674
35. Cabazitaxel [ema Epar]
36. Gtpl6798
37. Chembl1201748
38. Amy4317
39. Cabazitaxel [orange Book]
40. Dtxsid40171389
41. Ex-a838
42. Mfcd18827611
43. Nsc761432
44. Nsc794609
45. S3022
46. Zinc85536932
47. Akos032947285
48. Ccg-270519
49. Cs-0972
50. Db06772
51. Nsc 761432
52. Nsc-794609
53. Ncgc00346704-01
54. Ncgc00346704-03
55. As-75355
56. Hy-15459
57. A25044
58. D09755
59. Ab01273971-01
60. Ab01273971_02
61. Q412963
62. Sr-01000941585
63. J-011721
64. J-519981
65. Sr-01000941585-1
66. (((tertbutoxy)carbonyl)amino)-2-hydroxy-3-phenylpropanoate1-hydroxy-7beta,10beta-dimethoxy-9-oxo-5beta,20-epoxytax-11-ene-2alpha,4,13alpha-triyl 4-acetate 2-benzoate 13-((2r,3s)-3-
67. (1s)-5beta,20-epoxy-9-oxo-7beta,10beta-dimethoxytaxa-11-ene-1,2alpha,4alpha,13alpha-tetraol 2-benzoate 4-acetate 13-[(2r,3s)-2-hydroxy-3-(tert-butoxycarbonylamino)-3-phenylpropionate]
68. (1s,2s,3r,4s,7r,9s,10s,12r,15s)-4-(acetyloxy)-15-{[(2r,3s)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-3-phenylpropanoyl]oxy}-1-hydroxy-9,12-dimethoxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0(3),(1)?.0?,?]heptadec-13-en-2-yl Benzoate
69. (1s,2s,3r,4s,7r,9s,10s,12r,15s)-4-(acetyloxy)-15-{[(2r,3s)-3-{[(tert-butoxy)carbonyl]amino}-2-hydroxy-3-phenylpropanoyl]oxy}-1-hydroxy-9,12-dimethoxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.0^{3,10}.0^{4,7}]heptadec-13-en-2-yl Benzoate
70. (2alpha,5beta,7beta,10beta,13alpha)-4-acetoxy-13-({(2r,3s)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl}oxy)-1-hydroxy-7,10-dimethoxy-9-oxo-5,20-epoxytax-11-en-2-yl Benzoate
71. (2ar,4s,4as,6r,9s,11s,12s,12ar,12bs)-12b-acetoxy-9-(((2r,3s)-3-((tert-butoxycarbonyl)amino)-2-hydroxy-3-phenylpropanoyl)oxy)-11-hydroxy-4,6-dimethoxy-4a,8,13,1
72. (2ar,4s,4as,6r,9s,11s,12s,12ar,12bs)-12b-acetoxy-9-(((2r,3s)-3-((tert-butoxycarbonyl)amino)-2-hydroxy-3-phenylpropanoyl)oxy)-11-hydroxy-4,6-dimethoxy-4a,8,13,13-tetramethyl-5-oxo-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-1h-7,11-methanocyclodeca[3,4]benzo[1,2-b]oxet-12-yl Benzoate
73. [(1s,2s,3r,4s,7r,9s,10s,12r,15s)-4-acetyloxy-1-hydroxy-15-[(2r,3s)-2-hydroxy-3-[(2-methylpropan-2-yl)oxycarbonylamino]-3-phenylpropanoyl]oxy-9,12-dimethoxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7]heptadec-13-en-2-yl] Benzoate
74. 1-hydroxy-7,10-dimethoxy-9-oxo-5,20-epoxytax-11-ene-2,4,13-triyl 4-acetate 2-benzoate 13-((2r,3s)-3-(((tertbutoxy)carbonyl)amino)-2-hydroxy-3-phenylpropanoate)
75. 1-hydroxy-7.beta.,10.beta.-dimethoxy-9-oxo-5.beta.,20-epoxytax-11-ene-2.alpha.,4,13.alpha.-triyl 4-acetate 2-benzoate 13-((2r,3s)-3-(((tert-butoxy)carbonyl)amino)-2-hydroxy-3-phenylpropanoate)
76. Benzenepropanoic Acid, Beta-[[(1,1-dimethylethoxy)carbonyl]amino]-alpha-hydroxy-, (2ar,4s,4as,6r,9s,11s,12s,12ar,12bs)-12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-11-hydroxy-4,6-dimethoxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1h-cyclodeca[3,4]benz[1,2-b]oxet-9-yl Ester, (alphar,betas)-
| Molecular Weight | 835.9 g/mol |
|---|---|
| Molecular Formula | C45H57NO14 |
| XLogP3 | 2.7 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 15 |
| Exact Mass | 835.37790549 g/mol |
| Monoisotopic Mass | 835.37790549 g/mol |
| Topological Polar Surface Area | 202 Ų |
| Heavy Atom Count | 60 |
| Formal Charge | 0 |
| Complexity | 1690 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 11 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Jevtana kit |
| Active Ingredient | Cabazitaxel |
| Dosage Form | Solution |
| Route | Iv (infusion) |
| Strength | 60mg/1.5ml (40mg/ml) |
| Market Status | Prescription |
| Company | Sanofi Aventis Us |
| 2 of 2 | |
|---|---|
| Drug Name | Jevtana kit |
| Active Ingredient | Cabazitaxel |
| Dosage Form | Solution |
| Route | Iv (infusion) |
| Strength | 60mg/1.5ml (40mg/ml) |
| Market Status | Prescription |
| Company | Sanofi Aventis Us |
For treatment of patients with hormone-refractory metastatic prostate cancer previously treated with a docetaxel-containing treatment regimen.
FDA Label
Jevtana in combination with prednisone or prednisolone is indicated for the treatment of patients with hormone-refractory metastatic prostate cancer previously treated with a docetaxel-containing regimen.
Treatment of prostate cancer
Treatment of patients with hormone refractory metastatic prostate cancer previously treated with a docetaxel-containing regimen.
Cabaitaxel has anti-tumour properties and is effective against docetaxel-sensitive and -insensitive tumours.
L01CD
L01CD04
L01CD04
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01C - Plant alkaloids and other natural products
L01CD - Taxanes
L01CD04 - Cabazitaxel
Absorption
After an intravenous dose of cabazitaxel 25 mg/m2 every three weeks to a population of 170 patients with solid tumors, the mean Cmax in patients with metastatic prostate cancer was 226 ng/mL (CV 107%) and was reached at the end of the one-hour infusion (Tmax). The mean AUC in patients with metastatic prostate cancer was 991 ng.h/mL (CV 34%). Administration with prednisone or prednisolone do not effect the pharmacokinetic profile of cabazitaxel.
Route of Elimination
After a one-hour intravenous infusion [14C]-cabazitaxel 25 mg/m2, approximately 80% of the administered dose was eliminated within 2 weeks. Cabazitaxel is mainly excreted in the feces as numerous metabolites (76% of the dose); while renal excretion of cabazitaxel and metabolites account for 3.7% of the dose (2.3% as unchanged drug in urine).
Volume of Distribution
The volume of distribution (Vss) was 4,864 L (2,643 L/m2 for a patient with a median BSA of 1.84 m2) at steady state. Compared to other taxanes, penetrates the CNS to a greater extent.
Clearance
Cabazitaxel has a plasma clearance of 48.5 L/h (CV 39%; 26.4 L/h/m2 for a patient with a median BSA of 1.84 m2) in patients with metastatic prostate cancer.
Cabazitaxel is extensively metabolized in the liver (>95%), mainly by the CYP3A4/5 isoenzyme (80% to 90%), and to a lesser extent by CYP2C8 which results in 20 different metabolites. Two of these metabolites are active demethylated derivatives of cabaxitaxel and referred to as RPR112698 and RPR123142 respectively. Docetaxel is another metabolite of cabazitaxel. Cabazitaxel is the main circulating moiety in human plasma.
Following a one-hour intravenous infusion, plasma concentrations of cabazitaxel can be described by a three-compartment pharmacokinetic model with -, -, and - half-lives of 4 minutes, 2 hours, and 95 hours, respectively.
Cabazitaxel is a microtubule inhibitor. Cabazitaxel binds to tubulin and promotes its assembly into microtubules while simultaneously inhibiting disassembly. This leads to the stabilization of microtubules, which results in the interference of mitotic and interphase cellular functions. The cell is then unable to progress further into the cell cycle, being stalled at metaphase, thus triggering apoptosis of the cancer cell.
 Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 41055
Submission : 2025-01-27
Status : Active
Type : II
 Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
GDUFA
DMF Review : Complete
Rev. Date : 2021-09-16
Pay. Date : 2020-12-10
DMF Number : 34809
Submission : 2020-04-17
Status : Active
Type : II
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23496
Submission : 2010-01-29
Status : Active
Type : II
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
GDUFA
DMF Review : Complete
Rev. Date : 2014-06-17
Pay. Date : 2014-03-20
DMF Number : 27895
Submission : 2014-02-18
Status : Active
Type : II
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
GDUFA
DMF Review : Complete
Rev. Date : 2014-06-02
Pay. Date : 2014-03-19
DMF Number : 28101
Submission : 2014-04-01
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2014-03-28
Pay. Date : 2014-02-19
DMF Number : 27978
Submission : 2014-02-20
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-03-28
Pay. Date : 2014-03-04
DMF Number : 27831
Submission : 2014-03-03
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25769
Submission : 2012-02-07
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27339
Submission : 2013-07-18
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-02-08
Pay. Date : 2013-09-06
DMF Number : 27441
Submission : 2013-09-26
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Date of Issue : 2025-07-15
Valid Till : 2028-07-07
Written Confirmation Number : WC-0067
Address of the Firm : Chemical Technical Operation
Date of Issue : 2025-11-12
Valid Till : 2028-11-11
Written Confirmation Number : WC-0115
Address of the Firm : D-35, Industrial Area, Kalyani, Dist-Nadia-741235, West Bengal

Date of Issue : 2025-09-03
Valid Till : 2028-08-08
Written Confirmation Number : WC-0041
Address of the Firm : Unit-I, Sy. No. 10, I.D.A, Gaddapotharam (V), Jinnaram (M), Sangareddy District,...

Date of Issue : 2025-09-19
Valid Till : 2028-05-05
Written Confirmation Number : WC-0349
Address of the Firm : Unit-II, Sy. Nos. 50, 53, 53/A, 54 & 54/A,Kardanur (Village), Patancheru (Mandal...

Date of Issue : 2026-01-01
Valid Till : 2028-07-02
Written Confirmation Number : WC-0147
Address of the Firm : 100% EOU, Plot No.33, 33A, 40 to 47,Block C, D, E, H, I& AM,Raichur Industrial G...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
Indena - Advancing botanical science with a century of innovation, quality, and trusted global expertise.
About the Company : Indena is a global leader in the identification, development, and production of high-quality plant-derived active ingredients for the pharmaceutical and health-food industries. Fou...
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing APIs, including hard-to-find drug substances, for pharmaceutical and biotech industries. LGM also operates as a full-service drug product ...
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
About the Company : Founded in 1984, DRL is well-known for its generic APIs and its track record in drug product development. It is one of the earliest pharma API manufacturers with a diverse portfoli...
About the Company : HRV Pharma is a global manufacturer, seller, and exporter of APIs, intermediates, pellets, food-grade chemicals, food additives, and food ingredients. The company provides sourcing...
 TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
TAPI, a leading global supplier of APIs, provides over 350 products and customized CDMO solutions for every stage of development.
About the Company : Founded in 1935, TAPI Technology & API Services has a long legacy of advancing health through innovation. Today, we offer one of the industry’s most comprehensive API portfolios ...
About the Company : Guangzhou Tosun Pharmaceutical was founded in 1999, which mainly focuses on importation & exportation of Active Pharmaceutical Ingrediants, Chemical Raw Materials, Intermediate, Ex...

About the Company : Laurus Labs is a leading research and development-driven pharmaceutical company in India. The company has grown consistently to become one of the leading manufacturers of Active Ph...

About the Company : Established in 2010, Mac-Chem is a NMM Group company. Mac-Chem is focused on oncology and other niche specialty APIs. It supplies quality APIs to the top 15 oncology players in Ind...

About the Company : Founded in 1980, Sichuan Xieli Pharmaceutical Co., Ltd. operates with the guiding principle of "collaborating to build a healthy enterprise." The company specializes in the cultiva...

About the Company : Synthland is an integrated pharmaceutical company that develops, produce and supplies Active Pharmaceutical Ingredients (APIs) and Intermediates. We also represent some well-known...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
99
PharmaCompass offers a list of Cabazitaxel API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Cabazitaxel manufacturer or Cabazitaxel supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cabazitaxel manufacturer or Cabazitaxel supplier.
A RPR 116258A manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of RPR 116258A, including repackagers and relabelers. The FDA regulates RPR 116258A manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. RPR 116258A API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of RPR 116258A manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A RPR 116258A supplier is an individual or a company that provides RPR 116258A active pharmaceutical ingredient (API) or RPR 116258A finished formulations upon request. The RPR 116258A suppliers may include RPR 116258A API manufacturers, exporters, distributors and traders.
click here to find a list of RPR 116258A suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A RPR 116258A DMF (Drug Master File) is a document detailing the whole manufacturing process of RPR 116258A active pharmaceutical ingredient (API) in detail. Different forms of RPR 116258A DMFs exist exist since differing nations have different regulations, such as RPR 116258A USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A RPR 116258A DMF submitted to regulatory agencies in the US is known as a USDMF. RPR 116258A USDMF includes data on RPR 116258A's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The RPR 116258A USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of RPR 116258A suppliers with USDMF on PharmaCompass.
A RPR 116258A written confirmation (RPR 116258A WC) is an official document issued by a regulatory agency to a RPR 116258A manufacturer, verifying that the manufacturing facility of a RPR 116258A active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting RPR 116258A APIs or RPR 116258A finished pharmaceutical products to another nation, regulatory agencies frequently require a RPR 116258A WC (written confirmation) as part of the regulatory process.
click here to find a list of RPR 116258A suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing RPR 116258A as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for RPR 116258A API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture RPR 116258A as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain RPR 116258A and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a RPR 116258A NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of RPR 116258A suppliers with NDC on PharmaCompass.
RPR 116258A Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of RPR 116258A GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right RPR 116258A GMP manufacturer or RPR 116258A GMP API supplier for your needs.
A RPR 116258A CoA (Certificate of Analysis) is a formal document that attests to RPR 116258A's compliance with RPR 116258A specifications and serves as a tool for batch-level quality control.
RPR 116258A CoA mostly includes findings from lab analyses of a specific batch. For each RPR 116258A CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
RPR 116258A may be tested according to a variety of international standards, such as European Pharmacopoeia (RPR 116258A EP), RPR 116258A JP (Japanese Pharmacopeia) and the US Pharmacopoeia (RPR 116258A USP).

