
Synopsis
Synopsis
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
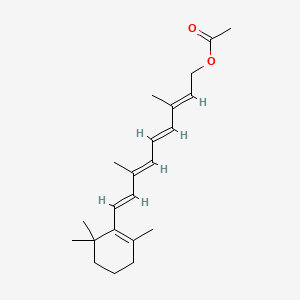

1. 9-cis-retinyl Acetate
2. All-trans-retinyl Acetate
3. Dagravit A Forte
4. Dif Vitamin A Masivo
5. Qlt091001
6. Retinit
7. Retinol Acetate
8. Retinol Acetate, (9,13-cis)-isomer
9. Vitamin A Acetate
10. Vitamin A Dispersa
11. Vitamin-a-saar
1. Vitamin A Acetate
2. Retinol Acetate
3. 127-47-9
4. Retinol, Acetate
5. All-trans-retinyl Acetate
6. Crystalets
7. Vitamin A1 Acetate
8. All-trans-retinol Acetate
9. Vitamin A Alcohol Acetate
10. Davitan A 650
11. Vitamin A Ester
12. All-trans-vitamin A Acetate
13. Retinol, Acetate, All-trans-
14. O~15~-acetylretinol
15. Nsc 122045
16. Trans-retinyl Acetate
17. [(2e,4e,6e,8e)-3,7-dimethyl-9-(2,6,6-trimethylcyclohexen-1-yl)nona-2,4,6,8-tetraenyl] Acetate
18. Ro 1-5275
19. Retinol Acetate [jan]
20. Trans-vitamin A Acetate
21. Myvak (van)
22. Myvax (van)
23. 3le3d9d6oy
24. All-trans-retinylacetate
25. (2e,4e,6e,8e)-3,7-dimethyl-9-(2,6,6-trimethylcyclohex-1-en-1-yl)nona-2,4,6,8-tetraen-1-yl Acetate
26. Chebi:32095
27. Vitamin A Acetate (van)
28. Acetic Acid, Retinyl Ester
29. Nsc-122045
30. Nsc-122760
31. Ncgc00090756-09
32. Retinol Acetate/all-trans-retinyl Acetate
33. Dsstox_cid_1240
34. Dsstox_rid_76032
35. Dsstox_gsid_21240
36. Vitaminaacetate
37. Vitamin A Acetate (tritiated)
38. Vitamin A, Acetate
39. Retinol, Acetate, Labeled With Tritium
40. O(15)-acetylretinol
41. 9-cis Retinol Acetate
42. Retinyl Acetate, All-trans-
43. Cas-127-47-9
44. Ccris 1907
45. Trans-retinol Acetate
46. Sr-05000001431
47. Einecs 204-844-2
48. Unii-3le3d9d6oy
49. Brn 1915439
50. Retinyl-acetate
51. Wln: L6utj A1 B1u1y1 & U2u1y1 & U2ov1 C1 C1
52. Vitamin A Acetat
53. 34356-31-5
54. Acetic Acid Retinyl
55. Mfcd00019413
56. Oristar Ra
57. (13z)-retinol Acetate
58. Spectrum5_001195
59. Spectrum5_002001
60. Retinol Acetate (jp17)
61. Retinyl (retinol) Acetate
62. Ec 204-844-2
63. Retinol, O~15~-acetyl-
64. Bspbio_002833
65. Spectrum1503051
66. Retinyl Acetate [inci]
67. C22h32o2 (retinol Acetate)
68. Chembl486193
69. Vitamin A Acetate [mi]
70. Dtxsid6021240
71. Chebi:94695
72. Hms501k04
73. Retinol Acetate [who-dd]
74. Retinyl Acetate [usp-rs]
75. Vitamin A Acetate [vandf]
76. Hms1922a19
77. Hms2089g20
78. Pharmakon1600-01503051
79. Amy13865
80. Hy-n0679
81. Zinc3874857
82. Retinyl Acetate, Analytical Standard
83. Tox21_113549
84. Tox21_201423
85. Tox21_302737
86. Bdbm50442911
87. Ccg-39564
88. Lmpr01090012
89. Nsc122045
90. Nsc122760
91. Nsc758220
92. S4083
93. Akos015914999
94. Tox21_113549_1
95. Cs-8187
96. Nsc-758220
97. Idi1_000522
98. Ncgc00090756-01
99. Ncgc00090756-02
100. Ncgc00090756-03
101. Ncgc00090756-05
102. Ncgc00090756-06
103. Ncgc00090756-07
104. Ncgc00090756-08
105. Ncgc00090756-10
106. Ncgc00090756-11
107. Ncgc00090756-12
108. Ncgc00256509-01
109. Ncgc00258974-01
110. Vitamin A (as Acetate) [vandf]
111. 64536-04-5
112. Ac-19999
113. All-(e)-retinol Acetate [who-ip]
114. Sbi-0051756.p002
115. Vitamin A Acetate 1.5 M.i.u./g
116. Ro-1-5275
117. A16783
118. D01621
119. H12041
120. Vitamin A Acetate 10 Microg/ml In Acetonitrile
121. Ab00052305-02
122. Ab00052305_03
123. Vitamin A Acetate In Gelatin, 500,000 I.u./g
124. Q7316808
125. Sr-05000001431-1
126. Sr-05000001431-3
127. W-108382
128. Brd-k65331431-001-01-3
129. Vitamin A (acetate), Meets Usp Testing Specifications
130. Vitamin A (as Acetate & Beta Carotene) [vandf]
131. Retinol Acetate, European Pharmacopoeia (ep) Reference Standard
132. Retinyl Acetate, Synthetic, Crystalline Solid Or Supercooled Liquid
133. Retinyl Acetate, United States Pharmacopeia (usp) Reference Standard
134. Retinyl Acetate, Concentrate (~50% (w/w) In Peanut Oil), ~1500 U/mg
135. Retinyl Acetate, Solid Or Viscous Liquid, Bioreagent, Synthetic, Suitable For Cell Culture
136. Retinyl Acetate, Synthetic, Matrix Dispersion, 475,000-650,000 Usp Units/g
137. (2e,4e,6e,8e)-3,7-dimethyl-9-(2,6,6-trimethylcyclohex-1-enyl)nona-2,4,6,8-tetraenyl Acetate
138. 3,7-dimethyl-9-(2,6,6,-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraen-1-ol Acetate, (all Trans)-
139. Retinyl Acetate (vitamin A Acetate), Pharmaceutical Secondary Standard; Certified Reference Material
140. Retinyl Acetate Solution, 100 Mug/ml In Ethanol (with 0.1% (w/v) Bht), Ampule Of 1 Ml, Certified Reference Material
141. Retinyl Acetate Solution, 100 Mug/ml In Ethanol (with 0.1% (w/v) Bht; Degassed), Certified Reference Material
| Molecular Weight | 328.5 g/mol |
|---|---|
| Molecular Formula | C22H32O2 |
| XLogP3 | 6.3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 7 |
| Exact Mass | 328.240230259 g/mol |
| Monoisotopic Mass | 328.240230259 g/mol |
| Topological Polar Surface Area | 26.3 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 596 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 4 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anticarcinogenic Agents
Agents that reduce the frequency or rate of spontaneous or induced tumors independently of the mechanism involved. (See all compounds classified as Anticarcinogenic Agents.)
Adjuvants, Immunologic
Substances that augment, stimulate, activate, potentiate, or modulate the immune response at either the cellular or humoral level. The classical agents (Freund's adjuvant, BCG, Corynebacterium parvum, et al.) contain bacterial antigens. Some are endogenous (e.g., histamine, interferon, transfer factor, tuftsin, interleukin-1). Their mode of action is either non-specific, resulting in increased immune responsiveness to a wide variety of antigens, or antigen-specific, i.e., affecting a restricted type of immune response to a narrow group of antigens. The therapeutic efficacy of many biological response modifiers is related to their antigen-specific immunoadjuvanticity. (See all compounds classified as Adjuvants, Immunologic.)
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
46
PharmaCompass offers a list of Vitamin A Acetate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Vitamin A Acetate manufacturer or Vitamin A Acetate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Vitamin A Acetate manufacturer or Vitamin A Acetate supplier.
A Retinyl Acetate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Retinyl Acetate, including repackagers and relabelers. The FDA regulates Retinyl Acetate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Retinyl Acetate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Retinyl Acetate manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Retinyl Acetate supplier is an individual or a company that provides Retinyl Acetate active pharmaceutical ingredient (API) or Retinyl Acetate finished formulations upon request. The Retinyl Acetate suppliers may include Retinyl Acetate API manufacturers, exporters, distributors and traders.
click here to find a list of Retinyl Acetate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Retinyl Acetate DMF (Drug Master File) is a document detailing the whole manufacturing process of Retinyl Acetate active pharmaceutical ingredient (API) in detail. Different forms of Retinyl Acetate DMFs exist exist since differing nations have different regulations, such as Retinyl Acetate USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Retinyl Acetate DMF submitted to regulatory agencies in the US is known as a USDMF. Retinyl Acetate USDMF includes data on Retinyl Acetate's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Retinyl Acetate USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Retinyl Acetate suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Retinyl Acetate Drug Master File in Japan (Retinyl Acetate JDMF) empowers Retinyl Acetate API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Retinyl Acetate JDMF during the approval evaluation for pharmaceutical products. At the time of Retinyl Acetate JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Retinyl Acetate suppliers with JDMF on PharmaCompass.
Retinyl Acetate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Retinyl Acetate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Retinyl Acetate GMP manufacturer or Retinyl Acetate GMP API supplier for your needs.
A Retinyl Acetate CoA (Certificate of Analysis) is a formal document that attests to Retinyl Acetate's compliance with Retinyl Acetate specifications and serves as a tool for batch-level quality control.
Retinyl Acetate CoA mostly includes findings from lab analyses of a specific batch. For each Retinyl Acetate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Retinyl Acetate may be tested according to a variety of international standards, such as European Pharmacopoeia (Retinyl Acetate EP), Retinyl Acetate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Retinyl Acetate USP).

