
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Cns 7056
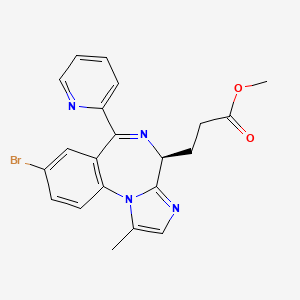
2. Methyl 3-(8-bromo-1-methyl-6-(2-pyridinyl)-4h-imidazo(1,2-a)(1,4)benzodiazepin-4-yl)propanoate
3. Ono 2745
4. Ono-2745
5. Ono2745
1. 308242-62-8
2. Cns 7056
3. Cns-7056
4. Ono-2745
5. 7v4a8u16mb
6. 308242-62-8 (freee Base)
7. 4h-imidazo(1,2-a)(1,4)benzodiazepine-4-propanoic Acid, 8-bromo-1-methyl-6-(2-pyridinyl)-, Methyl Ester, (4s)-
8. Methyl (s)-3-(8-bromo-1-methyl-6-(pyridin-2-yl)-4h-benzo[f]imidazo[1,2-a][1,4]diazepin-4-yl)propanoate
9. Remimazolam [inn]
10. Unii-7v4a8u16mb
11. Remimazolam [mi]
12. Remimazolam (usan/inn)
13. Remimazolam [usan:inn]
14. Remimazolam [usan]
15. Remimazolam [who-dd]
16. Schembl846435
17. Chembl4297526
18. Dtxsid20953024
19. Ono2745
20. Amy15524
21. Ex-a5536
22. Ono 2745
23. Who 9232
24. Zinc3927450
25. Akos025213215
26. Db12404
27. Hy-14867
28. 4523b
29. Cs-0003605
30. D11788
31. 4h-imidazol(1,2-a)(1,4)benzodiazepine-4-propionic Acid, (s)-
32. (s)-methyl 3-(8-bromo-1-methyl-6-(pyridin-2-yl)-4h-benzo[f]imidazo[1,2-a][1,4]diazepin-4-yl)propanoate
33. Methyl 3-((4s)-8-bromo-1-methyl-6-(pyridin-2-yl)-4h-imidazo(1,2-a)(1,4)benzodiazepin-4-yl)propanoate
34. Methyl 3-[(4s)-8-bromo-1-methyl-6-(2-pyridyl)-4h-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propanoate
35. Methyl 3-[(4s)-8-bromo-1-methyl-6-pyridin-2-yl-4h-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propanoate
36. Methyl 3-[8-bromo-1-methyl-6-(pyridin-2-yl)-4h-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propanoate
| Molecular Weight | 439.3 g/mol |
|---|---|
| Molecular Formula | C21H19BrN4O2 |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 5 |
| Exact Mass | 438.06914 g/mol |
| Monoisotopic Mass | 438.06914 g/mol |
| Topological Polar Surface Area | 69.4 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 601 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Remimazolam is indicated for the induction and maintenance of procedural sedation in adults undergoing procedures lasting 30 minutes or less.
FDA Label
Remimazolam is indicated in adults for procedural sedation.
Remimazolam modulates the effects of GABA(A) receptors in order to enhance the effects of GABA. It is considered an "ultra short-acting" benzodiazepine that achieves peak sedation within 3 to 3.5 minutes following intravenous administration, a property that makes it desirable for use during short procedures. Hepatic impairment can result in elevated serum levels of remimazolam - patients with severe hepatic impairment should be carefully titrated to effect. As of its approval date, remimazolam has not received a scheduling action by the DEA under the Controlled Substances Act. As benzodiazepines as a class have been implicated in the development of drug dependence and have a known potential for abuse, remimazolam should be used with caution in patients with a history of drug dependence or abuse.
N05CD
N - Nervous system
N05 - Psycholeptics
N05C - Hypnotics and sedatives
N05CD - Benzodiazepine derivatives
N05CD14 - Remimazolam
Absorption
The Cmax and AUC0-inf following intravenous administration of 0.01 to 0.5 mg/kg were 189 to 6,960 ng/mL and 12.1 to 452 ngh/mL, respectively, and appear to be relatively dose proportional. The Tmax of the inactive CNS7054 metabolite is approximately 20-30 minutes and its AUC0-inf ranges from 231 to 7,090 ngh/mL.
Route of Elimination
In patients undergoing colonoscopy, approximately 0.003% of the administered dose is excreted in the urine as unchanged parent drug and 50-60% is excreted in the urine as CNS7054. In healthy subjects, >80% of the administered dose is excreted in the urine as CNS7054.
Volume of Distribution
The volume of distribution is approximately 0.76 - 0.98 L/kg.
Clearance
The clearance of remimazolam is approximately 24 - 75 L/h and is independent of body weight.
Remimazolam does not appear to undergo biotransformation via hepatic cytochrome P450 enzymes, nor does it induce or inhibit these enzymes. Its primary route of metabolism is hydrolysis via hepatic carboxylesterase-1 (CES1) to yield the inactive CNS7054 metabolite, which then undergoes glucuronidation and hydroxylation prior to elimination. CNS7054 possesses a 300-fold lesser affinity for GABA(A) receptors as compared to the parent drug.
Following intravenous administration, the distribution half-life is of remimazolam is 0.5 - 2 minutes and the terminal elimination half-life is 37 - 53 minutes. Half-life is increased in patients with hepatic impairment necessitating careful dose titration in this population. The half-life of remimazolam's major inactive metabolite, CNS7054, is 2.4 - 3.8 hours.
Like other benzodiazepines, remimazolam exerts its therapeutic effects by potentiating the effect of gamma-aminobutyric acid (GABA) on GABA(A) receptors, the main inhibitory neurotransmitter receptors in the mammalian brain. GABA(A) receptors are a component of GABA-gated ionotropic chloride channels that produce inhibitory postsynaptic potentials - following activation by GABA, the channel undergoes a conformational change that allows the passage of chloride ions through the channel. The inhibitory potentials produced by GABA neurotransmission play an integral role in the suppression and control of epileptiform nerve firing such as that seen in epilepsy, which makes the GABA system a desirable target in the treatment of epilepsy. Benzodiazepines are positive allosteric modulators of GABA(A) function. They bind to the interface between alpha () and gamma () subunits on the receptor, commonly referred to as the benzodiazepine binding site, and modulate the receptor such that its inhibitory response to GABA binding is dramatically increased.
Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
87
PharmaCompass offers a list of Remimazolam Besylate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Remimazolam Besylate manufacturer or Remimazolam Besylate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Remimazolam Besylate manufacturer or Remimazolam Besylate supplier.
A Remimazolam manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Remimazolam, including repackagers and relabelers. The FDA regulates Remimazolam manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Remimazolam API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Remimazolam manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Remimazolam supplier is an individual or a company that provides Remimazolam active pharmaceutical ingredient (API) or Remimazolam finished formulations upon request. The Remimazolam suppliers may include Remimazolam API manufacturers, exporters, distributors and traders.
click here to find a list of Remimazolam suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Remimazolam DMF (Drug Master File) is a document detailing the whole manufacturing process of Remimazolam active pharmaceutical ingredient (API) in detail. Different forms of Remimazolam DMFs exist exist since differing nations have different regulations, such as Remimazolam USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Remimazolam DMF submitted to regulatory agencies in the US is known as a USDMF. Remimazolam USDMF includes data on Remimazolam's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Remimazolam USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Remimazolam suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Remimazolam Drug Master File in Korea (Remimazolam KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Remimazolam. The MFDS reviews the Remimazolam KDMF as part of the drug registration process and uses the information provided in the Remimazolam KDMF to evaluate the safety and efficacy of the drug.
After submitting a Remimazolam KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Remimazolam API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Remimazolam suppliers with KDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Remimazolam as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Remimazolam API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Remimazolam as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Remimazolam and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Remimazolam NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Remimazolam suppliers with NDC on PharmaCompass.
Remimazolam Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Remimazolam GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Remimazolam GMP manufacturer or Remimazolam GMP API supplier for your needs.
A Remimazolam CoA (Certificate of Analysis) is a formal document that attests to Remimazolam's compliance with Remimazolam specifications and serves as a tool for batch-level quality control.
Remimazolam CoA mostly includes findings from lab analyses of a specific batch. For each Remimazolam CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Remimazolam may be tested according to a variety of international standards, such as European Pharmacopoeia (Remimazolam EP), Remimazolam JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Remimazolam USP).

