
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Cvt 3146
2. Cvt-3146
3. Cvt3146
4. Lexiscan
1. 313348-27-5
2. Lexiscan
3. Rapiscan
4. Cvt-3146
5. Regadenoson Anhydrous
6. 2-[4-[(methylamino)carbonyl]-1h-pyrazol-1-yl]adenosine
7. 7axv542lz4
8. Chembl317052
9. 1-(6-amino-9-((2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-9h-purin-2-yl)-n-methyl-1h-pyrazole-4-carboxamide
10. Adenosine, 2-[4-[(methylamino)carbonyl]-1h-pyrazol-1-yl]-
11. Regadenoson Monohydrate
12. Lexiscan (tn)
13. Cvt 3146
14. Regadenoson [usan:inn]
15. Unii-7axv542lz4
16. Adenosine, 2-(4-((methylamino)carbonyl)-1h-pyrazol-1-yl)-
17. Regadenoson [mi]
18. Regadenoson [inn]
19. Regadenoson (usan/inn)
20. Regadenoson; Cvt-3146
21. Dsstox_cid_31501
22. Dsstox_rid_97386
23. Regadenoson [mart.]
24. Dsstox_gsid_57712
25. Schembl678893
26. Gtpl5596
27. Dtxsid4057712
28. Chebi:135613
29. Hms3886o21
30. Amy27715
31. Ex-a2148
32. Hy-a0168
33. Tox21_113668
34. Bdbm50119132
35. S5358
36. Zinc13818943
37. Akos026750593
38. Ccg-268525
39. Cs-5612
40. Cv-3146
41. Db06213
42. 1-(6-amino-9-beta-d-ribofuranosyl-9h-purin-2-yl)-n-methyl-1h-pyrazole-4-carboxamide
43. Ncgc00249892-01
44. (1-(9-(3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl)-6-aminopurin-2-yl)pyrazol-4-yl)-n-methylcarboxamide
45. Ac-35838
46. As-56292
47. B5904
48. Cas-313348-27-5
49. Cvt-3146;cvt3146;cvt 3146
50. D05711
51. 348r275
52. J-018384
53. Q7307897
54. (1-{9-[(4s,2r,3r,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-6-aminopurin-2-yl}pyrazol-4-yl)-n-methylcarboxamide
55. 1-[6-amino-9-((2r,3r,4s,5r)-3,4-dihydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-9h-purin-2-yl]-1h-pyrazole-4-carboxylic Acid Methylamide
56. 1-{6-amino-9-[(2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-9h-purin-2-yl}-n-methyl-1h-pyrazole-4-carboxamide
57. 6-amino-2-[4-(methylcarbamoyl)-1h-pyrazol-1-yl]purine-9-yl-beta-d-ribofuranoside;1-(6-amino-9-((2s,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yloxy)-9h-purin-2-yl)-n-methyl-1h-pyrazole-4-carboxamide
| Molecular Weight | 390.35 g/mol |
|---|---|
| Molecular Formula | C15H18N8O5 |
| XLogP3 | -1.5 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 4 |
| Exact Mass | 390.14001570 g/mol |
| Monoisotopic Mass | 390.14001570 g/mol |
| Topological Polar Surface Area | 187 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 587 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Lexiscan |
| PubMed Health | Regadenoson (Injection) |
| Drug Classes | Diagnostic Agent, Cardiac Function |
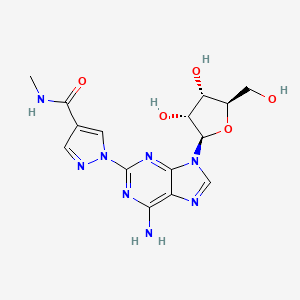
| Drug Label | Regadenoson is an A2A adenosine receptor agonist that is a coronary vasodilator [see Clinical Pharmacology (12.1)]. Regadenoson is chemically described as adenosine, 2-[4-[(methylamino)carbonyl]-1H-pyrazol-1-yl]-, monohydrate. Its structural formula... |
| Active Ingredient | Regadenoson |
| Dosage Form | Solution |
| Route | Intravenous |
| Strength | 0.4mg/5ml (0.08mg/ml) |
| Market Status | Prescription |
| Company | Astellas |
| 2 of 2 | |
|---|---|
| Drug Name | Lexiscan |
| PubMed Health | Regadenoson (Injection) |
| Drug Classes | Diagnostic Agent, Cardiac Function |
| Drug Label | Regadenoson is an A2A adenosine receptor agonist that is a coronary vasodilator [see Clinical Pharmacology (12.1)]. Regadenoson is chemically described as adenosine, 2-[4-[(methylamino)carbonyl]-1H-pyrazol-1-yl]-, monohydrate. Its structural formula... |
| Active Ingredient | Regadenoson |
| Dosage Form | Solution |
| Route | Intravenous |
| Strength | 0.4mg/5ml (0.08mg/ml) |
| Market Status | Prescription |
| Company | Astellas |
Diagnostic agent for radionuclide myocardial perfusion imaging (MPI)
FDA Label
This medicinal product is for diagnostic use only.
Rapiscan is a selective coronary vasodilator for use as a pharmacological stress agent for radionuclide myocardial perfusion imaging (MPI) in adult patients unable to undergo adequate exercise stress.
Regadenoson rapidly increases coronary blood flow (CBF) which is sustained for a short duration. Mean average peak velocity increased to greater than twice baseline by 30 seconds and decreased to less than twice the baseline level within 10 minutes. Myocardial uptake of the radiopharmaceutical is proportional to (CBF). Regadenoson increases blood flow in normal coronary arteries but not in stenotic (blocked) arteries. The significance of this finding is that stenotic arteries will take up less of the radiopharmaceutical than normal coronary arteries, resulting in a signal that is less intense in these areas.
Adenosine A2 Receptor Agonists
Compounds that selectively bind to and activate ADENOSINE A2 RECEPTORS. (See all compounds classified as Adenosine A2 Receptor Agonists.)
C01EB21
C01EB21
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
C - Cardiovascular system
C01 - Cardiac therapy
C01E - Other cardiac preparations
C01EB - Other cardiac preparations
C01EB21 - Regadenoson
Absorption
The pharmacokinetic profile of regadenoson is best described by a 3-compartment model. T max, injection = 1 to 3 minutes; Onset of pharmacodynamic response = 1 to 3 minutes; E max 12.3 ng/mL
Route of Elimination
58% of total regadenoson eliminate is via renal excretion
Volume of Distribution
Central compartment: 11.5 L; Steady state: 78.7 L
Clearance
Average plasma renal clearance = 450 mL/min. As this value is larger than the glomerular filtration rate, this suggests occurrence of renal tubular secretion.
The metabolism of regadenoson is unknown in humans. The cytochrome P450 enzyme system is not likely to be involved with the metabolism of regadenoson.
Initial phase: 2-4 minutes; Intermediate phase: 30 minutes (this phase coincides with a loss of the pharmacodynamic effect); Terminal phase: 2 hours
Regadenoson is an selective low-affinity (Ki= 1.3 M) A2A receptor agonist that mimics the effects of adenosine in causing coronary vasodilatation and increasing myocardial blood flow. It is a very weak agonist of the A1 adenosine receptor (Ki > 16.5 M). Furthermore, it has negligible affinity to A2B and A3 adenosine receptors. Regadenoson is undergoing trials for use in pharmacological stress tests. Adenosine slows conduction time through the A-V node, can interrupt the reentry pathways through the A-V node, and can restore normal sinus rhythm in patients with paroxysmal supraventricular tachycardia (PSVT), including PSVT associated with Wolff-Parkinson-White Syndrome.
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
25
PharmaCompass offers a list of Regadenoson Monohydrate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Regadenoson Monohydrate manufacturer or Regadenoson Monohydrate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Regadenoson Monohydrate manufacturer or Regadenoson Monohydrate supplier.
A Regadenoson manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Regadenoson, including repackagers and relabelers. The FDA regulates Regadenoson manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Regadenoson API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Regadenoson manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Regadenoson supplier is an individual or a company that provides Regadenoson active pharmaceutical ingredient (API) or Regadenoson finished formulations upon request. The Regadenoson suppliers may include Regadenoson API manufacturers, exporters, distributors and traders.
click here to find a list of Regadenoson suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Regadenoson DMF (Drug Master File) is a document detailing the whole manufacturing process of Regadenoson active pharmaceutical ingredient (API) in detail. Different forms of Regadenoson DMFs exist exist since differing nations have different regulations, such as Regadenoson USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Regadenoson DMF submitted to regulatory agencies in the US is known as a USDMF. Regadenoson USDMF includes data on Regadenoson's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Regadenoson USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Regadenoson suppliers with USDMF on PharmaCompass.
A Regadenoson written confirmation (Regadenoson WC) is an official document issued by a regulatory agency to a Regadenoson manufacturer, verifying that the manufacturing facility of a Regadenoson active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Regadenoson APIs or Regadenoson finished pharmaceutical products to another nation, regulatory agencies frequently require a Regadenoson WC (written confirmation) as part of the regulatory process.
click here to find a list of Regadenoson suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Regadenoson as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Regadenoson API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Regadenoson as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Regadenoson and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Regadenoson NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Regadenoson suppliers with NDC on PharmaCompass.
Regadenoson Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Regadenoson GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Regadenoson GMP manufacturer or Regadenoson GMP API supplier for your needs.
A Regadenoson CoA (Certificate of Analysis) is a formal document that attests to Regadenoson's compliance with Regadenoson specifications and serves as a tool for batch-level quality control.
Regadenoson CoA mostly includes findings from lab analyses of a specific batch. For each Regadenoson CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Regadenoson may be tested according to a variety of international standards, such as European Pharmacopoeia (Regadenoson EP), Regadenoson JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Regadenoson USP).

