
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
0
Europe
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
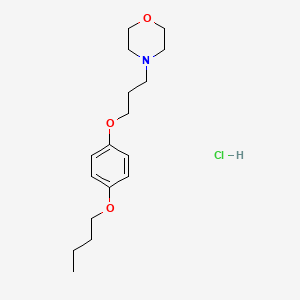

1. 4-(3-(4-butoxyphenoxy)propyl)morpholine
2. 4-(3-(p-butoxyphenoxy)propyl)morpholine
3. Balsabit
4. Fleet Pain Relief
5. Itch-x
6. Pramegel
7. Pramocaine
8. Pramocaine Hydrochloride
9. Pramox
10. Pramoxine
11. Prax
12. Proctofoam
13. Tronolane
14. Tronothane
1. 637-58-1
2. Pramoxine Hcl
3. Pramocaine Hydrochloride
4. Prax Lotion
5. Tronothane
6. Pramocaine (hydrochloride)
7. Morpholine, 4-[3-(4-butoxyphenoxy)propyl]-, Hydrochloride
8. 4-[3-(4-butoxyphenoxy)propyl]morpholine Hydrochloride
9. Nsc-25573
10. 4-(3-(4-butoxyphenoxy)propyl)morpholine Hydrochloride
11. Pramoxine Hydrochloride [usp]
12. Mls000028604
13. Tronolane
14. 88ayb867l5
15. 4-(3-(p-butoxyphenoxy)propyl)morpholine Hydrochloride
16. 4-[3-(4-butoxyphenoxy)propyl]morpholine;hydrochloride
17. 637-58-1 (hcl)
18. Morpholine, 4-(3-(4-butoxyphenoxy)propyl)-, Hydrochloride
19. Smr000058670
20. Dsstox_cid_27757
21. Dsstox_rid_82534
22. Dsstox_gsid_47777
23. Pramoxine Hydrochloride (usp)
24. Tronothane Hydrochloride
25. Proctofoam-ns
26. Hsdb 7220
27. Ncgc00016529-01
28. Cas-637-58-1
29. Einecs 211-293-1
30. Nsc 25573
31. 4-[3-(p-butoxyphenoxy)propyl]morpholine Hydrochloride
32. Unii-88ayb867l5
33. Sr-01000000243
34. Pramoxine, Hcl
35. Tronothane (tn)
36. Prestwick_1044
37. P-butoxyphenyl Gamma-morpholinopropyl Ether Hydrochloride
38. Gamma-morpholinopropyl 4-n-butoxyphenyl Ether Hydrochloride
39. Opera_id_1593
40. Pramoxine Hcl [inci]
41. Mls002222295
42. Schembl180542
43. Spectrum1501139
44. Amy585
45. Chebi:8358
46. Chembl1201171
47. Dtxsid2047777
48. Morpholine, 4-(3-(p-butoxyphenoxy)propyl)-, Hydrochloride
49. Hms1570o13
50. Hms1921j07
51. Pharmakon1600-01501139
52. Hy-b1319
53. Nsc25573
54. Pramoxine Hydrochloride [mi]
55. Tox21_110477
56. Mfcd00054323
57. Nsc757847
58. S4092
59. Pramoxine Hydrochloride [hsdb]
60. Akos015895041
61. Tox21_110477_1
62. Ac-2094
63. Ccg-212937
64. Cs-4643
65. Nsc-757847
66. Pramoxine Hydrochloride [vandf]
67. Wln: T6n Dotj A3or Do4 &gh
68. Pramocaine Hydrochloride [mart.]
69. Pramoxine Hydrochloride [usp-rs]
70. Ncgc00016529-07
71. Ncgc00094907-01
72. Ncgc00094907-02
73. Ncgc00094907-03
74. Pramocaine Hydrochloride [who-dd]
75. As-12691
76. Db-054541
77. Ft-0603501
78. Pramoxine Hydrochloride [orange Book]
79. Sw197114-3
80. Vu0239834-5
81. D00739
82. D97624
83. Epifoam Component Pramoxine Hydrochloride
84. Pramoxine Hydrochloride [usp Monograph]
85. A834509
86. Pramosone Component Pramoxine Hydrochloride
87. Pramoxine Hydrochloride Component Of Epifoam
88. Pramoxine Hydrochloride, Analytical Standard, >=98%
89. Q-100732
90. Sr-01000000243-3
91. Pramoxine Hydrochloride Component Of Pramosone
92. Proctofoam Hc Component Pramoxine Hydrochloride
93. Q27269918
94. 4-[3-(4-butoxyphenoxy)propyl]morpholin-4-ium;chloride
95. Pramoxine Hydrochloride Component Of Proctofoam Hc
96. P-butoxyphenyl .gamma.-morpholinopropyl Ether Hydrochloride
97. .gamma.-morpholinopropyl 4-n-butoxyphenyl Ether Hydrochloride
98. 4-[3-(4-butoxyphenoxy)propyl]morpholine Hydrochloride (1:1)
99. Morpholine, 4-[3-(p-n-butoxyphenoxy )propyl]-, Hydrochloride
100. 4-n-butoxyphenyl.gamma.-morpholinopropyl Ether Hydrochloride
101. Morpholine, 4-[3-(4-butoxyphenoxy)propyl]-, Hydrochloride (1:1)
102. 4-[3-(4-butoxyphenoxy)propyl]morpholine Chloride;pramocaine Hydrochloride
103. Pramoxine Hydrochloride, United States Pharmacopeia (usp) Reference Standard
104. Pramoxine Hydrochloride, Pharmaceutical Secondary Standard; Certified Reference Material
| Molecular Weight | 329.9 g/mol |
|---|---|
| Molecular Formula | C17H28ClNO3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 9 |
| Exact Mass | 329.1757714 g/mol |
| Monoisotopic Mass | 329.1757714 g/mol |
| Topological Polar Surface Area | 30.9 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 248 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Anesthetic (local)
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. 13th Edition, Whitehouse Station, NJ: Merck and Co., Inc., 2001., p. 1376
Indicated for the symptomatic relief of: hemorrhoids, anorectal inflammation and anorectal pain. (these medications /including pramoxine/ are effective when applied to the anal, periana, or anorectal areas. However, they are not likely to relieve symptoms associated with conditions confined to the rectum, which lacks sensory nerve fibers.) /Included in US product labeling/
Thomson.Micromedex. Drug Information for the Health Care Professional. 24th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2004., p. 153
Pramoxine preparations are used topically for the temporary relief of pain and itching associated with dermatoses; rashes due to poison ivy, poison oak, poison sumac, insect bites; minor burns; anogenital pruritus or irritation; and fissures; or hemorrhoids. The drug should not be used for bronchoscopy or gastroscopy since it is not sufficiently potent to abolish the gag reflex.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3414
Local effects, such as burning or stinging, may occur following topical application of pramoxine. The drug has a low index of sensitization, and cross-sensitization with other local anesthetics is unlikely.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3414
Topical pramoxine preparations should not be used in or near the eyes or nose.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3414
The preparations should not be applied to extensive areas of skin and are not for prolonged use. The aerosol foam for rectal administration should not be used longer than 4 consecutive weeks.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3414
If pramoxine preparations are used for self-medication and the condition worsens or symptoms persist for prolonged periods (e.g., for more than 7 days) or clear and occur again within a few days, the drug should be discontinued and a physician consulted.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3414
If rectal bleeding occurs during use of pramoxine for pain and itching of hemorrhoids or if erythema, irritation, swelling, or pain occurs during use of pramoxine preparations, the drug should be discontinued and a physician consulted.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2004. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2004 (Plus Supplements)., p. 3414
Anesthetics, Local
Drugs that block nerve conduction when applied locally to nerve tissue in appropriate concentrations. They act on any part of the nervous system and on every type of nerve fiber. In contact with a nerve trunk, these anesthetics can cause both sensory and motor paralysis in the innervated area. Their action is completely reversible. (From Gilman AG, et. al., Goodman and Gilman's The Pharmacological Basis of Therapeutics, 8th ed) Nearly all local anesthetics act by reducing the tendency of voltage-dependent sodium channels to activate. (See all compounds classified as Anesthetics, Local.)
... These agents are readily absorbed through mucous membranes into the systemic circulation. The rate of absorption is influenced by the vascularity or rate of blood flow at the site of application, the total dosage (concentration and volume) administered, and the duration of exposure. ... /Local anesthetics/
Thomson.Micromedex. Drug Information for the Health Care Professional. 24th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2004., p. 154
Local anesthetics block both the initiation and conduction of nerve impulses by decreasing the neuronal membrane's permeability to sodium ions. This reversibly stabilizes the membrane and inhibits depolarization, resulting in the failure of a propagated action potential and subsequent conduction blockade. /Local anesthetics/
Thomson.Micromedex. Drug Information for the Health Care Professional. 24th ed. Volume 1. Plus Updates. Content Reviewed by the United States Pharmacopeial Convention, Inc. Greenwood Village, CO. 2004., p. 154
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
79
PharmaCompass offers a list of Pramoxine Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Pramoxine Hydrochloride manufacturer or Pramoxine Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Pramoxine Hydrochloride manufacturer or Pramoxine Hydrochloride supplier.
A Pramoxine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Pramoxine, including repackagers and relabelers. The FDA regulates Pramoxine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Pramoxine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Pramoxine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Pramoxine supplier is an individual or a company that provides Pramoxine active pharmaceutical ingredient (API) or Pramoxine finished formulations upon request. The Pramoxine suppliers may include Pramoxine API manufacturers, exporters, distributors and traders.
click here to find a list of Pramoxine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Pramoxine DMF (Drug Master File) is a document detailing the whole manufacturing process of Pramoxine active pharmaceutical ingredient (API) in detail. Different forms of Pramoxine DMFs exist exist since differing nations have different regulations, such as Pramoxine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Pramoxine DMF submitted to regulatory agencies in the US is known as a USDMF. Pramoxine USDMF includes data on Pramoxine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Pramoxine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Pramoxine suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Pramoxine Drug Master File in Korea (Pramoxine KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Pramoxine. The MFDS reviews the Pramoxine KDMF as part of the drug registration process and uses the information provided in the Pramoxine KDMF to evaluate the safety and efficacy of the drug.
After submitting a Pramoxine KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Pramoxine API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Pramoxine suppliers with KDMF on PharmaCompass.
A Pramoxine written confirmation (Pramoxine WC) is an official document issued by a regulatory agency to a Pramoxine manufacturer, verifying that the manufacturing facility of a Pramoxine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Pramoxine APIs or Pramoxine finished pharmaceutical products to another nation, regulatory agencies frequently require a Pramoxine WC (written confirmation) as part of the regulatory process.
click here to find a list of Pramoxine suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Pramoxine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Pramoxine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Pramoxine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Pramoxine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Pramoxine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Pramoxine suppliers with NDC on PharmaCompass.
Pramoxine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Pramoxine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Pramoxine GMP manufacturer or Pramoxine GMP API supplier for your needs.
A Pramoxine CoA (Certificate of Analysis) is a formal document that attests to Pramoxine's compliance with Pramoxine specifications and serves as a tool for batch-level quality control.
Pramoxine CoA mostly includes findings from lab analyses of a specific batch. For each Pramoxine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Pramoxine may be tested according to a variety of international standards, such as European Pharmacopoeia (Pramoxine EP), Pramoxine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Pramoxine USP).

