
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
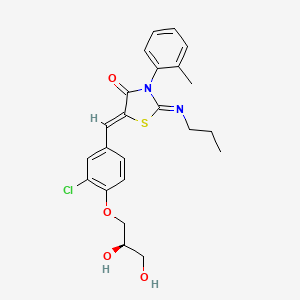

1. 5-(3-chloro-4-(2,3-dihydroxy-propoxy)benzylidene)-2-propylimino-3-o-tolyl-thiazolidin-4-one
2. Act 128800
3. Act-128800
4. Act128800
5. Ponvory
1. 854107-55-4
2. Act-128800
3. Ponvory
4. 5g7akv2mkp
5. Chembl1096146
6. Act128800
7. (2z,5z)-5-(3-chloro-4-((2r)-2,3-dihydroxypropoxy)phenylmethylidene)-3-(2-methylphenyl)-2-(propylimino)-1,3-thiazolidin-4-one
8. (5z)-5-[[3-chloro-4-[(2r)-2,3-dihydroxypropoxy]phenyl]methylidene]-3-(2-methylphenyl)-2-propylimino-1,3-thiazolidin-4-one
9. (z)-5-((z)-3-chloro-4-((r)-2,3-dihydroxypropoxy)benzylidene)-2-(propylimino)-3-(o-tolyl)thiazolidin-4-one
10. Unii-5g7akv2mkp
11. Ponesimod [inn]
12. (2z,5z)-5-(3-chloro-4-((r)-2,3-dihydroxypropoxy)benzylidene)-2-(propylimino)-3-(o-tolyl)thiazolidin-4-one
13. 4-thiazolidinone, 5-[[3-chloro-4-[(2r)-2,3-dihydroxypropoxy]phenyl]methylene]-3-(2-methylphenyl)-2-(propylimino)-, (2z,5z)-
14. Ponesimod [usan:inn]
15. 4-thiazolidinone, 5-((3-chloro-4-((2r)-2,3-dihydroxypropoxy)phenyl)methylene)-3-(2-methylphenyl)-2-(propylimino)-, (2z,5z)-
16. Act 128800
17. Ponesimod (usan/inn)
18. Ponesimod [usan]
19. Ponesimod [who-dd]
20. Ponesimod,act-128800
21. Gtpl9320
22. Ponesimod (act-128800)
23. Ponesimod [orange Book]
24. Schembl15477934
25. Schembl15477937
26. Dtxsid50234631
27. Amy23424
28. Enb-0040
29. Ex-a1417
30. Bdbm50316768
31. Mfcd18207776
32. S8241
33. Zinc34509627
34. At27979
35. Ccg-269345
36. Compound 8bo [pmid:20446681]
37. Db12016
38. Ac-30921
39. As-35140
40. Bp175283
41. Hy-10569
42. D11215
43. (2z,5z)-5-(3-chloro-4-((2r)-2,3-dihydroxypropoxy)benzylidene)-3-(2-methylphenyl)-2-(propylimino)-1,3-thiazolidin-4-one
44. (z,z)-5-[3-chloro-4-((2s)-2,3-dihydroxy-propoxy)-benzylidene]-2-propylimino-3-otolyl-thiazolidin-4-one
45. 4-thiazolidinone,5-[[3-chloro-4-[(2r)-2,3-dihydroxypropoxy]phenyl]methylene]-3-(2-methylphenyl)-2-(propylimino)-,(2z,5z)-
46. 5-(3-chloro-4-(((2r)-2,3-dihydroxypropyl)oxy)benz-(z)-ylidene)-2-((z)-propylimino)-3-(o-tolyl)thiazolidin-4-one
47. 5-(3-chloro-4-(2,3-dihydroxy-propoxy)benzylidene)-2-propylimino-3-o-tolyl-thiazolidin-4-one
| Molecular Weight | 461.0 g/mol |
|---|---|
| Molecular Formula | C23H25ClN2O4S |
| XLogP3 | 4.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 8 |
| Exact Mass | 460.1223562 g/mol |
| Monoisotopic Mass | 460.1223562 g/mol |
| Topological Polar Surface Area | 108 Ų |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 674 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Ponesimod is indicated to treat adults with relapsing forms of multiple sclerosis, including clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease.
Ponvory is indicated for the treatment of adult patients with relapsing forms of multiple sclerosis (RMS) with active disease defined by clinical or imaging features.
Ponesimod is a sphingosine 1-phosphate receptor 1 modulator indicated to treat adults with relapsing forms of multiple sclerosis. It has a long duration of action as it is given once daily. Patients should be counselled about the risk of infections, bradyarrhythmia, atrioventricular conduction delays, decreased respiratory function, liver injury, increased blood pressure, cutaneous malignancies, fetal harm, and macular edema.
Sphingosine 1 Phosphate Receptor Modulators
Agents that affect the function of G-protein coupled SPHINGOSINE 1-PHOSPHATE RECEPTORS. Their binding to the receptors blocks lymphocyte migration and are often used as IMMUNOSUPPRESSANTS. (See all compounds classified as Sphingosine 1 Phosphate Receptor Modulators.)
L04AA
L - Antineoplastic and immunomodulating agents
L04 - Immunosuppressants
L04A - Immunosuppressants
L04AA - Selective immunosuppressants
L04AA50 - Ponesimod
Absorption
A 10mg oral dose of ponesimod is 84% bioavailable. Ponesimod reaches a Cmax of 109 ng/mL, with a Tmax of 4.0 hours, and an AUC of 3872 h\*ng/mL.
Route of Elimination
57.3-79.6% of a radiolabelled oral dose is recovered in the feces, with 16-26% as the unmetabolized parent compound and 22% as the M12 metabolite. 10.3-18.4% of an oral dose is eliminated in the urine. 0.6-1.9% of a radiolabelled dose was recovered as expired CO2.
Volume of Distribution
The volume of distribution of ponesimod at steady state is 160 L.
Clearance
The clearance of ponesimod is 3.8 L/h.
Ponesimod can be sulfated to the M5 metabolite, oxidized to an undefined M27 metabolite, reduced to the M6 metabolite, dealkylated to the M32 metabolite, or oxidized and hydrolyzed to the M13 metabolite. Ponesimod can also be oxidized by CYP2J2, CYP3A4, CYP3A5, CYP4F3A, and CYP4F12 to the M12 metabolite. The undefined M27 metabolite can be glucuronidated by UGT1A1 and UGT 2B7 to the M38, M39, and M40 metabolites. The M12 metabolite is either dealkylated to the M32 metabolite or oxidized and hydrolyzed to M13. M13 is dealkylated to M32, which is reduced and oxidized to M48.
Ponesimod has an elimination half life of 33 hours.
The sphingosine 1-phosphate receptor 1 (S1P1R) is expressed on the surface of lymphocytes and detects sphingosine 1-phosphate (S1P) at nanomolar concentrations. S1P is a metabolite of the cell membrane component, sphingomyelin. As sphingomyelin degrades, lymphocytes respond to agonism of S1P1R by concentration gradients of S1P. Lymphocytes leave the lymphoid organs in response to higher concentrations of S1P in blood and lymph. Ponesimod modulates this response by stimulating and internalizing S1P1R on lymphocytes, effectively blinding them to concentration gradients of S1P, reducing the number of lymphocytes in blood. Ponesimod is roughly 650 times more selective for S1P1R than S1P.
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
92
PharmaCompass offers a list of Ponesimod API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ponesimod manufacturer or Ponesimod supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ponesimod manufacturer or Ponesimod supplier.
A Ponesimod manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ponesimod, including repackagers and relabelers. The FDA regulates Ponesimod manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ponesimod API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ponesimod manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ponesimod supplier is an individual or a company that provides Ponesimod active pharmaceutical ingredient (API) or Ponesimod finished formulations upon request. The Ponesimod suppliers may include Ponesimod API manufacturers, exporters, distributors and traders.
click here to find a list of Ponesimod suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
Ponesimod Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ponesimod GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ponesimod GMP manufacturer or Ponesimod GMP API supplier for your needs.
A Ponesimod CoA (Certificate of Analysis) is a formal document that attests to Ponesimod's compliance with Ponesimod specifications and serves as a tool for batch-level quality control.
Ponesimod CoA mostly includes findings from lab analyses of a specific batch. For each Ponesimod CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ponesimod may be tested according to a variety of international standards, such as European Pharmacopoeia (Ponesimod EP), Ponesimod JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ponesimod USP).

