
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDA Orange Book
0
Canada
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Demerol
2. Dolantin
3. Dolargan
4. Dolcontral
5. Dolin
6. Dolosal
7. Dolsin
8. Isonipecain
9. Lidol
10. Lydol
11. Meperidine Hydrochloride
12. Operidine Epj I
13. Operidine Epj-i
14. Pethidine
1. Pethidine
2. Isonipecaine
3. Meperidol
4. Pethanol
5. Pethidineter
6. Demerol
7. Pethidin
8. Phetidine
9. Ethyl 1-methyl-4-phenylpiperidine-4-carboxylate
10. 57-42-1
11. Pipersal
12. Nemerol
13. Petydyna
14. Pethidinum
15. Petidina
16. Ethyl 1-methyl-4-phenylisonipecotate
17. 4-piperidinecarboxylic Acid, 1-methyl-4-phenyl-, Ethyl Ester
18. Renaudin
19. Pethidine Dbl
20. Methyl Phenylpiperidine Carbonic Acid Ethyl Ester
21. Isonipecotic Acid, 1-methyl-4-phenyl-, Ethyl Ester
22. N-methyl-4-phenyl-4-carbethoxypiperidine
23. 1-methyl-4-phenylpiperidine-4-carboxylic Acid Ethyl Ester
24. 1-methyl-4-phenylisonipecotic Acid, Ethyl Ester
25. Hsdb 3116
26. Pethidine (inn)
27. Ids-np-001
28. 1-methyl-4-phenyl-4-piperidinecarboxylic Acid Ethyl Ester
29. Chembl607
30. 1-methyl-4-phenyl-piperidin-4-carbon-saeure-aethylester
31. Chebi:6754
32. 9e338qe28f
33. Pethidine [inn]
34. Meperidina
35. Mepridine
36. Petydyna [polish]
37. Demarol
38. Piperosal
39. Pethidinum [inn-latin]
40. Petidina [inn-spanish]
41. 1-methyl-4-phenyl-piperidine-4-carboxylic Acid Ethyl Ester
42. Pethidine Dbl (tn)
43. Pethidine [inn:ban]
44. Einecs 200-329-1
45. Unii-9e338qe28f
46. Dea No. 9230
47. 1-methyl-4-phenyl-piperidin-4-carbon-saeure-aethylester [german]
48. Lidol (salt/mix)
49. Lydol (salt/mix)
50. Dolsin (salt/mix)
51. Dolaren (salt/mix)
52. Dolosal (salt/mix)
53. Dispadol (salt/mix)
54. Dolantin (salt/mix)
55. Petantin (salt/mix)
56. Operidine (salt/mix)
57. Piridosal (salt/mix)
58. 4-carbethoxy-1-methyl-4-phenylpiperidine
59. Centralgin (salt/mix)
60. Dolcontral (salt/mix)
61. Meperidine [mi]
62. Meperidine [hsdb]
63. Meperidine [vandf]
64. 1-methyl-4-phenylisonipecotic Acid Ethyl Ester
65. Pethidine [who-dd]
66. Ethyl 1-methyl-4-phenyl-4-piperidinecarboxylate
67. Schembl25431
68. Divk1c_000983
69. Gtpl7221
70. Zinc1681
71. Dtxsid9023253
72. Kbio1_000983
73. Ninds_000983
74. Yisonipecotic Acid, 1-methyl-4-phenyl-, Ethyl Ester (8ci)
75. Bdbm50026752
76. Db00454
77. Idi1_000983
78. Db-053051
79. C07128
80. D08343
81. Pethidine (meperidine) 0.1 Mg/ml In Methanol
82. Pethidine (meperidine) 1.0 Mg/ml In Methanol
83. Q55434
84. 4-ethoxycarbonyl-1-methyl-4-phenyl-piperidinium
85. L000908
86. Sr-01000944492
87. Sr-01000944492-1
88. Meperidine Solution, 1 Mg/ml In Methanol, Drug Standard
89. 1-methyl-4-phenyl-piperidine-4-carboxylic Acid Ethyl Ester(meperidine)
90. Meperidine Solution, 1.0 Mg/ml In Methanol, Ampule Of 1 Ml, Certified Reference Material
| Molecular Weight | 247.33 g/mol |
|---|---|
| Molecular Formula | C15H21NO2 |
| XLogP3 | 2.5 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Exact Mass | 247.157228913 g/mol |
| Monoisotopic Mass | 247.157228913 g/mol |
| Topological Polar Surface Area | 29.5 Ų |
| Heavy Atom Count | 18 |
| Formal Charge | 0 |
| Complexity | 276 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Demerol |
| PubMed Health | Meperidine (Injection) |
| Drug Classes | Analgesic, Anesthetic Adjunct |
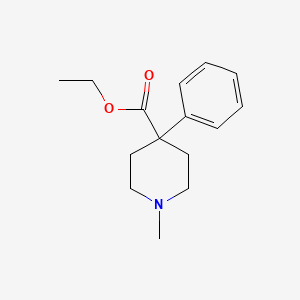
| Drug Label | Meperidine hydrochloride is ethyl 1-methyl-4-phenylisonipecotate hydrochloride, a white crystalline substance with a melting point of 186C to 189C. It is readily soluble in water and has a neutral reaction and a slightly bitter taste. The solutio... |
| Active Ingredient | Meperidine hydrochloride |
| Dosage Form | Injectable; Tablet |
| Route | Injection; Oral |
| Strength | 75mg/ml; 50mg/ml; 25mg/ml; 100mg; 100mg/ml; 50mg |
| Market Status | Prescription |
| Company | Sanofi Aventis Us; Hospira |
| 2 of 2 | |
|---|---|
| Drug Name | Demerol |
| PubMed Health | Meperidine (Injection) |
| Drug Classes | Analgesic, Anesthetic Adjunct |
| Drug Label | Meperidine hydrochloride is ethyl 1-methyl-4-phenylisonipecotate hydrochloride, a white crystalline substance with a melting point of 186C to 189C. It is readily soluble in water and has a neutral reaction and a slightly bitter taste. The solutio... |
| Active Ingredient | Meperidine hydrochloride |
| Dosage Form | Injectable; Tablet |
| Route | Injection; Oral |
| Strength | 75mg/ml; 50mg/ml; 25mg/ml; 100mg; 100mg/ml; 50mg |
| Market Status | Prescription |
| Company | Sanofi Aventis Us; Hospira |
Analgesics, Opioid; Adjuvants, Anesthesia; Narcotics
National Library of Medicine's Medical Subject Headings. Meperidine. Online file (MeSH, 2017). Available from, as of September 1, 2017: https://www.nlm.nih.gov/mesh/2017/mesh_browser/MBrowser.html
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM's MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Meperidine is included in the database.
NIH/NLM; ClinicalTrials.Gov. Available from, as of September 1, 2017: https://clinicaltrials.gov/
Meperidine is a strong analgesic used in the relief of moderate to severe pain. The drug has been used to relieve the pain of myocardial infarction, although it is probably not as effective as morphine sulfate. Meperidine also is used parenterally for preoperative sedation, as a supplement to anesthesia, and to provide analgesia during labor. Meperidine is used in patients with acute pulmonary edema for its cardiovascular effects and to allay anxiety. The drug should not be used in the treatment of pulmonary edema resulting from a chemical respiratory irritant. /Included in US product label/
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2256
Use of meperidine hydrochloride as first-line opiate therapy is discouraged because of the central excitatory toxicity of its metabolite, normeperidine. Use of meperidine hydrochloride for management of chronic pain is discouraged because of its short duration of effect and the risk of normeperidine accumulation and resultant central excitatory toxicity with repeated or large doses. Some experts also discourage use in children.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2256
For more Therapeutic Uses (Complete) data for Meperidine (12 total), please visit the HSDB record page.
/BOXED WARNING/ RISK OF MEDICATION ERRORS. Ensure accuracy when prescribing, dispensing, and administering Meperidine Hydrochloride Oral Solution. Dosing errors due to confusion between mg and mL, and other Meperidine Hydrochloride Oral Solutions of different concentrations can result in accidental overdose and death.
NIH; DailyMed. Current Medication Information for Meperidine Hydrochloride Tablet; Meperidine Hydrochloride Solution (Updated: September 2017). Available from, as of November 17, 2017: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=15bc51a8-1e24-4d33-9a2f-05a41c29acf0
/BOXED WARNING/ ADDICTION, ABUSE, AND MISUSE. Meperidine Hydrochloride Tablets and Oral Solution expose patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death. Assess each patient's risk prior to prescribing meperidine, and monitor all patients regularly for the development of these behaviors and conditions.
NIH; DailyMed. Current Medication Information for Meperidine Hydrochloride Tablet; Meperidine Hydrochloride Solution (Updated: September 2017). Available from, as of November 17, 2017: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=15bc51a8-1e24-4d33-9a2f-05a41c29acf0
/BOXED WARNING/ LIFE-THREATENING RESPIRATORY DEPRESSION. Serious, life-threatening, or fatal respiratory depression may occur with use of Meperidine Hydrochloride Tablets or Oral Solution. Monitor for respiratory depression, especially during initiation of Meperidine Hydrochloride Tablets or Oral Solution or following a dose increase.
NIH; DailyMed. Current Medication Information for Meperidine Hydrochloride Tablet; Meperidine Hydrochloride Solution (Updated: September 2017). Available from, as of November 17, 2017: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=15bc51a8-1e24-4d33-9a2f-05a41c29acf0
/BOXED WARNING/ ACCIDENTAL INGESTION. Accidental ingestion of Meperidine Hydrochloride Tablets or Oral Solution, especially by children, can result in a fatal overdose of meperidine.
NIH; DailyMed. Current Medication Information for Meperidine Hydrochloride Tablet; Meperidine Hydrochloride Solution (Updated: September 2017). Available from, as of November 17, 2017: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=15bc51a8-1e24-4d33-9a2f-05a41c29acf0
For more Drug Warnings (Complete) data for Meperidine (29 total), please visit the HSDB record page.
Toxic meperidine blood concentration: 500 ug/dL; Lethal meperidine blood concentration: 1-3 mg/dL /From table/
Gossel, T.A., J.D. Bricker. Principles of Clinical Toxicology. 3rd ed. New York, NY: Raven Press, Ltd., 1994., p. 298
Used to control moderate to severe pain.
FDA Label
Meperidine is a synthetic opiate agonist belonging to the phenylpiperidine class. Meperidine may produce less smooth muscle spasm, constipation, and depression of the cough reflex than equivalent doses of morphine. The onset of action is lightly more rapid than with morphine, and the duration of action is slightly shorter. The chemical structure of meperidine is similar to local anesthetics. Meperidine is recommended for relief of moderate to severe acute pain and has the unique ability to interrupt postoperative shivering and shaking chills induced by amphotericin B. Meperidine has also been used for intravenous regional anesthesia, peripheral nerve blocks and intraarticular, epidural and spinal analgesia. Meperidine is considered a second-line agent for the treatment of acute pain.
Adjuvants, Anesthesia
Agents that are administered in association with anesthetics to increase effectiveness, improve delivery, or decrease required dosage. (See all compounds classified as Adjuvants, Anesthesia.)
Analgesics, Opioid
Compounds with activity like OPIATE ALKALOIDS, acting at OPIOID RECEPTORS. Properties include induction of ANALGESIA or NARCOSIS. (See all compounds classified as Analgesics, Opioid.)
Narcotics
Agents that induce NARCOSIS. Narcotics include agents that cause somnolence or induced sleep (STUPOR); natural or synthetic derivatives of OPIUM or MORPHINE or any substance that has such effects. They are potent inducers of ANALGESIA and OPIOID-RELATED DISORDERS. (See all compounds classified as Narcotics.)
N - Nervous system
N02 - Analgesics
N02A - Opioids
N02AB - Phenylpiperidine derivatives
N02AB02 - Pethidine
Absorption
The oral bioavailability of meperidine in patients with normal hepatic function is 50-60% due to extensive first-pass metabolism. Bioavailability increases to 80-90% in patients with hepatic impairment (e.g. liver cirrhosis). Meperidine is less than half as effective when administered orally compared to parenteral administration. One study reported that 80-85% of the drug administered intramuscularly was absorbed within 6 hours of intragluteal injection in health adults; however, inter-individual variation and patient-specific variable appear to cause considerable variations in absorption upon IM injection.
Route of Elimination
Excreted in the urine. The proportion of drug that is excreted unchanged or as metabolites is dependent on pH. When urine pH is uncontrolled, 5-30% of the meperidine dose is excreted as normeperidine and approximately 5% is excreted unchanged. Meperidine and normeperidine are found in acidic urine, while the free and conjugated forms of meperidinic and normperidinic acids are found in alkaline urine.
Volume of Distribution
Meperidine crosses the placenta and is distributed into breast milk.
Following oral administration, meperidine undergoes extensive metabolism on first pass through the liver, with approximately 50-60% of a dose reaching systemic circulation unchanged. In patients with hepatic impairment (e.g., liver cirrhosis), oral bioavailability of meperidine increases to approximately 80-90%. Meperidine is less than one-half as effective when given orally as when given parenterally. Approximately 80-85% of an IM dose of the drug reportedly was absorbed within 6 hours after intragluteal injection in healthy adults in one study; however, absorption from the IM injection site appears to show considerable interindividual variation and may depend on the site of injection, dose, and patient-specific variables. Meperidine appears to have a more rapid onset and shorter duration of action than does morphine. Following oral administration of meperidine, peak analgesia occurs within one hour and gradually declines over 2-4 hours. Peak analgesia occurs about 40-60 minutes after subcutaneous administration and 30-50 minutes after IM administration. Analgesia may be maintained for 2-4 hours following subcutaneous or IM administration.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2257
Meperidine is approximately 60-80% bound to plasma proteins, principally albumin and alpha1-acid glycoprotein (alpha1-AGP). There is some evidence that the ratio of bound to free drug is correlated with plasma alpha1-AGP concentrations. In patients with cirrhosis or active viral hepatitis, the extent of protein binding does not appear to be affected.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2257
/MILK/ Meperidine crosses the placenta and is distributed into milk.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2257
Plasma meperidine concentrations decline in a biphasic manner, with a half-life in the initial distribution phase (t1/2alpha) of 2-11 minutes and a half-life in the terminal elimination phase (t1/2beta) of 3-5 hours in individuals with normal renal and hepatic function. The elimination half-life is prolonged in patients with hepatic dysfunction, averaging about 7-11 hours in patients with liver cirrhosis or active viral hepatitis.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2257
For more Absorption, Distribution and Excretion (Complete) data for Meperidine (14 total), please visit the HSDB record page.
Meperidine is metabolized in the liver by hydrolysis to meperidinic acid followed by partial conjugation with glucuronic acid. Meperidine also undergoes N-demethylation to normeperidine, which then undergoes hydrolysis and partial conjugation. Normeperidine is about half as potent as meperidine, but it has twice the CNS stimulation effects.
Meperidine is metabolized principally in the liver. The drug is biotransformed mainly by hydrolysis to meperidinic acid followed by partial conjugation with glucuronic acid. Meperidine may also undergo N-demethylation to normeperidine followed by hydrolysis and partial conjugation. Other metabolites also have been identified, but only normeperidine has been detected in blood or plasma. When urine pH is uncontrolled, approximately 5-30% of a dose of meperidine is excreted in urine as the N-demethylated derivative and about 5% is excreted unchanged; however, the relative proportion of the drug excreted in urine unchanged and as metabolites is pH dependent. Meperidine and normeperidine are found in acid urine whereas meperidinic and normeperidinic acids in the free and conjugated form are present in alkaline urine. Excretion of the unchanged drug and normeperidine is enhanced by acidifying the urine. Normeperidine is pharmacologically active, reportedly exhibiting about half the analgesic potency of meperidine but twice the CNS stimulant (e.g., seizure-inducing) potency. Various toxic effects secondary to CNS stimulation (e.g., seizures, agitation, irritability, nervousness, tremors, twitches, myoclonus) have been attributed to accumulation of this metabolite. The elimination half-life of normeperidine is substantially longer than that of meperidine, reportedly ranging from 8-21 hours, and may be prolonged (e.g., to longer than 30 hours) in patients with renal impairment. Accumulation of this metabolite may occur with repeated, high doses of the drug and in patients with renal or hepatic impairment.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2257
Meperidine (Demerol) is a mu- and kappa-opiate receptor agonist used for moderate to severe pain. Overdose can result in respiratory depression, hypotension and coma, while accumulation of its toxic metabolite, normeperidine, can cause delirium and seizures. Little data exist examining the inter- and intrasubject variability of the normeperidine-to-meperidine metabolic ratio (MR) in urine. This retrospective data analysis examined meperidine and normeperidine urine concentrations collected from chronic pain patients. In 98 subjects with multiple visits, the geometric mean urinary MR = 6.1 (coefficient of variation, %CV = 68%). From single specimens obtained from 799 subjects, the geometric mean urinary MR = 6.2 (%CV = 212%). The urinary MR increased in young subjects compared with elderly (P = 0.004) and middle-aged subjects (P = 0.01). A 27% difference was found between the male and female urinary MR (male geometric mean MR = 5.1, female geometric mean MR = 7.0, P = 0.02). Intersubject variability in meperidine metabolism was 3-fold greater than intrasubject variability. A significant difference in the urinary MR was found between males and females. The substantial variability in meperidine metabolism and the serious side effects of its metabolite normeperidine require greater vigilance in patient medication monitoring.
PMID:24133175 Moy KV et al; J Anal Toxicol 38 (1): 1-7 (2014)
Human liver carboxylesterases catalyze the hydrolysis of apolar drug or xenobiotic esters into more soluble acid and alcohol products for elimination. Two carboxylesterases, hCE-1 and hCE-2, have been purified and characterized with respect to their role in cocaine and heroin hydrolysis. The binding of meperidine (Demerol) and propoxyphene (Darvon) was examined in a competitive binding, spectrophotometric assay. The hCE-1 and hCE-2 bound both drugs, with Ki values in the 0.4- to 1.3-mM range. Meperidine was hydrolyzed to meperidinic acid and ethanol by hCE-1 but not hCE-2. The Km of hCE-1 for meperidine was 1.9 mM and the kcat (catalytic rate constant) was 0.67 min-1. Hydrolysis of meperidine by hCE-1 was consistent with its specificity for hydrolysis of esters containing simple aliphatic alcohol substituents. Hence, hCE-1 in human liver microsomes may play an important role in meperidine elimination. Propoxyphene was not hydrolyzed by hCE-1 or hCE-2. This observation is consistent with the absence of a major hydrolytic pathway for propoxyphene metabolism in humans.
PMID:10381793 Zhang J et al; J Pharmacol Exp Ther 290 (1): 314-8 (1999)
Meperidine is metabolized to N-methyl-4-phenylpiperidine-4-carboxylic acid in rat. /From table/
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. M-4
For more Metabolism/Metabolites (Complete) data for Meperidine (13 total), please visit the HSDB record page.
Meperidine has known human metabolites that include normeperidine.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
Initial distribution phase (t1/2 α) = 2-11 minutes; terminal elimination phase (t1/2 β) = 3-5 hours. In patients with hepatic dysfunction (e.g. liver cirrhosis or active viral hepatitis) the t1/2 β is prolonged to 7-11 hours.
Plasma meperidine concentrations decline in a biphasic manner, with a half-life in the initial distribution phase (t1/2alpha) of 2-11 minutes and a half-life in the terminal elimination phase (t1/2beta) of 3-5 hours in individuals with normal renal and hepatic function. The elimination half-life is prolonged in patients with hepatic dysfunction, averaging about 7-11 hours in patients with liver cirrhosis or active viral hepatitis.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 2257
After single rapid iv injection in man, 0.8 mg/kg, plasma meperidine concentration declined biexponentially with fast (alpha) and slow (beta) phases having half-lives in normal group of 0.19 hr and 3.2 hr (mean values), respectively. In cirrhotics half-life-alpha was not changed but half-life-beta increased to 7.0 hr.
PMID:4419525 Klotz U et al; Clin Pharmacol Ther 16: 667-75 (1974)
Pharmacokinetics of morphine, buprenorphine and pethidine were determined in 10 cats. ... Six received pethidine (5 mg/kg) intramuscularly. Jugular venous blood samples were collected at time points to 24 hr, and plasma morphine concentrations were measured by high performance liquid chromatograpy (HPLC), buprenorphine by radioimmunoassay (RIA) and pethidine by gas chromatography. ... For i.m. pethidine, elimination half-life /was/ 216.4 min ... .
PMID:11903869 Taylor PM et al; J Vet Pharmacol Ther 24 (6): 391-8 (2001)
Following intravenous administration of meperidine in healthy individuals, the volume of distribution at steady state was 269 L (range, 198 to 333 L); plasma clearance was 1.06 L/min (range, 0.71 to 1.32), and the elimination half-life was 3.6 hours (range, 3.1 to 4.1). Liver disease, eg, cirrhosis, acute viral hepatitis, doubles the half-life. There is evidence that the disposition of meperidine varies between day and night, with the elimination half-life being shorter and the plasma clearance greater at night. This suggests that larger doses might be required at night. Bioavailability after oral administration is about 50% due to first-pass metabolism in the liver, but increases to 80% to 90% in patients with cirrhosis. The elimination half-life in the neonate is 22.7 hours.
American Medical Association. AMA Drug Evaluations Annual 1991. Chicago, IL: American Medical Association, 1991., p. 94
For more Biological Half-Life (Complete) data for Meperidine (7 total), please visit the HSDB record page.
Meperidine is primarily a kappa-opiate receptor agonist and also has local anesthetic effects. Meperidine has more affinity for the kappa-receptor than morphine. Opiate receptors are coupled with G-protein receptors and function as both positive and negative regulators of synaptic transmission via G-proteins that activate effector proteins. Binding of the opiate stimulates the exchange of GTP for GDP on the G-protein complex. As the effector system is adenylate cyclase and cAMP located at the inner surface of the plasma membrane, opioids decrease intracellular cAMP by inhibiting adenylate cyclase. Subsequently, the release of nociceptive neurotransmitters such as substance P, GABA, dopamine, acetylcholine and noradrenaline is inhibited. Opioids also inhibit the release of vasopressin, somatostatin, insulin and glucagon. Opioids close N-type voltage-operated calcium channels (OP2-receptor agonist) and open calcium-dependent inwardly rectifying potassium channels (OP3 and OP1 receptor agonist). This results in hyperpolarization and reduced neuronal excitability.
Meperidine is a mu (OP3)-receptor (MOR) agonist opioid with approximately 20-25% the potency of morphine. Receptors for opiate analgesics are found in high concentrations in the limbic system, spinal cord, thalamus, hypothalamus, striatum, and midbrain. They are also found in tissues such as the gastrointestinal tract, urinary track, and in other smooth muscle. The morphine-like agonists (morphine, meperidine, oxymorphone) have primary activity in the mu receptors, with some activity possible in the delta receptor. ... Meperidine produces equivalent respiratory depression at equi-analgesic doses as morphine. Like morphine, it can cause histamine release. It does not have antitussive activity at doses lower than those causing analgesia. Meperidine is the only used opioid that has vagolytic and negative inotropic properties at clinically used doses. One study in ponies demonstrated jejunal activity after meperidine administration, but no effects on transit time or colonic electrical activity were noted.
Plumb D.C. Veterinary Drug Handbook. 8th ed. (pocket). Ames, IA: Wiley-Blackwell, 2015., p. 925
The purpose of the present study was to investigate the effect of meperidine on rat ventricular muscle. Cardiac function was assessed in Langendorff-perfused rat hearts and intracellular calcium level was recorded in enzymatically isolated rat ventricular myocytes using spectrofluorometric techniques. To explore the underlying mechanism, whole-cell configuration of patch-clamp technique was used to record L-type Ca(2+) current. The results showed that meperidine decreased the product of heart rate and left ventricular developed pressure (LVDP HR), maximal rate of the left ventricular pressure increase (LV +dP/dt(max)) and decrease (LV -dP/dt(max)), but increased left ventricular end-diastolic pressure in a dose-dependent manner (0-1000 umol/L). Meperidine also produced a dose-dependent reduction in electrically induced [Ca(2+)](i) transient amplitude and an increase in diastolic [Ca(2+)](i) baseline level, but did not alter the caffeine (20 mmol/L) induced Ca(2+) release from intracellular ryanodine-sensitive Ca(2+) stores. Meperidine at 100 umol/L inhibited L-type Ca(2+) current to 67.4 10.1% of control but did not affect the voltage dependency of activation and inactivation. The inhibitory effect of meperidine on Ca(2+) current could not be prevented by pretreatment with the opioid receptor antagonist naloxone. These data suggest that meperidine exerts a negative inotropic effect by inhibiting L-type Ca(2+) current. The lack of effect of naloxone implies that the action is independent of the opioid receptor.
PMID:12715111 Zhang X et al; Sheng Li Xue Bao 55 (2): 197-200 (2003)
In addition to local anesthetics, meperidine has been successfully used for local anesthesia. When applied intrathecally, the dorsal horn neurons of the superficial laminae are exposed to high concentrations of meperidine. These cells represent an important point for the transmission of pain information. This study investigated the blocking effects of meperidine on different ionic currents of spinal dorsal horn neurons and, in particular, its impact on the generation of action potentials. Using a combination of the patch clamp technique and the entire soma isolation method, the action of meperidine on voltage-gated Na+ and K+ currents in spinal dorsal horn neurons of rats was described. Current clamp recordings from intact neurons showed the functional relevance of the ion current blockade for the generation of action potentials. Externally applied meperidine reversibly blocked voltage-gated Na+ currents with a half-maximum inhibiting concentration (IC50) of 112 uM. During repetitive stimulation, a slight phasic block occurred. In addition, A-type K+ currents and delayed-rectifier K+ currents were affected in a dose-dependent manner, with IC50 values of 102 and 52 uM, respectively. In the current clamp mode, single action potentials were suppressed by meperidine. The firing frequency was lowered to 54% at concentrations (100 uM) insufficient for the suppression of a single action potential. Meperidine inhibits the complex mechanism of generating action potentials in spinal dorsal horn neurons by the blockade of voltage-gated Na+ and K+ channels. This can contribute to the local anesthetic effect of meperidine during spinal anesthesia.
PMID:15087632 Wolff M et al; Anesthesiology 100 (4): 947-55 (2004)
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
41
PharmaCompass offers a list of Meperidine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Meperidine manufacturer or Meperidine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Meperidine manufacturer or Meperidine supplier.
A Pethidine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Pethidine, including repackagers and relabelers. The FDA regulates Pethidine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Pethidine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Pethidine supplier is an individual or a company that provides Pethidine active pharmaceutical ingredient (API) or Pethidine finished formulations upon request. The Pethidine suppliers may include Pethidine API manufacturers, exporters, distributors and traders.
Pethidine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Pethidine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Pethidine GMP manufacturer or Pethidine GMP API supplier for your needs.
A Pethidine CoA (Certificate of Analysis) is a formal document that attests to Pethidine's compliance with Pethidine specifications and serves as a tool for batch-level quality control.
Pethidine CoA mostly includes findings from lab analyses of a specific batch. For each Pethidine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Pethidine may be tested according to a variety of international standards, such as European Pharmacopoeia (Pethidine EP), Pethidine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Pethidine USP).

