
Synopsis
Synopsis
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. Advantage S
2. Advantage-s
3. Delfen Cream
4. Delfen Creams
5. Emulgen 911
6. Emulgin 913
7. Nonoxinol
8. Nonoxinols
9. Nonoxynol
10. Nonoxynol 9
11. Nonoxynol, 1(4)-sulfate, Sodium Salt
12. Nonoxynol, 4-sulfate, Ammonium Salt
13. Nonoxynols
14. Nonylphenoxypolyethoxyethanol
15. Nonylphenoxypolyethoxyethanols
16. Oval, Patentex
17. Patentex Oval
1. Nonoxynol 9
2. 14409-72-4
3. Delfen
4. Nonoxynol, N=9
5. 26571-11-9
6. Tergitol Np-9
7. Peg-9 Nonyl Phenyl Ether
8. Nonoxynol9
9. Tergitol Np9
10. Nonaethylene Glycol Nonylphenyl Ether
11. Nonylphenol Octa(oxyethylene) Ethanol
12. Nonaethylene Glycol Mono(nonylphenyl) Ether
13. Nonoxinol-9
14. 2-[2-[2-[2-[2-[2-[2-[2-[2-(4-nonylphenoxy)ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethoxy]ethanol
15. Nonylphenoxypolyethoxyethanol
16. Polyoxyethylene (9) Nonyl Phenyl Ether
17. 26-(4-nonylphenoxy)-3,6,9,12,15,18,21,24-octaoxahexacosan-1-ol
18. 26-(nonylphenoxy)-3,6,9,12,15,18,21,24-octaoxahexacosan-1-ol
19. Chebi:53775
20. Nsc-758941
21. P-nonylphenyl Polyethylene Glycol Ether
22. N-9
23. Conceptrol
24. Intercept
25. Semicid
26. 3,6,9,12,15,18,21,24-octaoxahexacosan-1-ol, 26-(4-nonylphenoxy)-
27. 2-[2-[2-[2-[2-[2-[2-[2-[2-(4-nonylphenoxy)ethoxy]ethoxy]ethoxy]ethoxy]-ethoxy]ethoxy]ethoxy]ethoxy]ethanol
28. Staycept
29. Emko
30. Today Sponge
31. Nonoxinolum 9
32. Hsdb 8094
33. Gynol Ii
34. C-film
35. Norfox Np-9
36. Tergitol Tp-9
37. Nonoxynol-9.5
38. Einecs 247-816-5
39. Conco Ni-90
40. K-y Plus Nonoxynol-9
41. Spectrum2_001247
42. Spectrum3_001946
43. Alosetronhydrochloride
44. Chembl1410
45. Schembl36844
46. Bspbio_003546
47. Spectrum1505292
48. Spbio_001154
49. Dowfax 9n9, Neutronyx 611
50. Co-630sp
51. Kbio3_002833
52. Dtxsid00858720
53. Hms2093o22
54. Pharmakon1600-01505292
55. Zinc8214629
56. 3,6,9,12,15,18,21,24-octaoxahexacosan-1-ol, 26-(nonylphenoxy)-
57. Bdbm50442874
58. Ccg-39124
59. Nsc758941
60. Akos015910808
61. Db06804
62. Nsc 758941
63. Ncgc00095907-02
64. Ncgc00095907-03
65. N 9
66. Sbi-0206761.p001
67. Polyethylene Glycol 450 Nonyl Phenyl Ether
68. Ab01563056_01
69. Q420039
70. Sr-05000001885
71. J-016227
72. Q-201490
73. Sr-05000001885-1
74. Brd-k88625236-001-01-8
75. 1-(4-nonylphenyl)-1,4,7,10,13,16,19,22,25-nonaoxaheptacosan-27-ol
76. N9
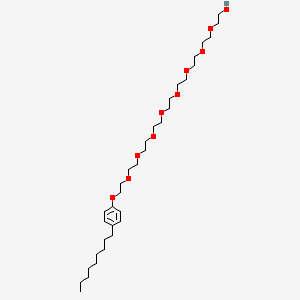
| Molecular Weight | 616.8 g/mol |
|---|---|
| Molecular Formula | C33H60O10 |
| XLogP3 | 4.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 35 |
| Exact Mass | 616.41864811 g/mol |
| Monoisotopic Mass | 616.41864811 g/mol |
| Topological Polar Surface Area | 103 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 520 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Today |
| PubMed Health | Nonoxynol 9 (Vaginal) |
| Drug Classes | Contraceptive, Local |
| Active Ingredient | Nonoxynol-9 |
| Dosage Form | Sponge |
| Route | Vaginal |
| Strength | 1gm |
| Market Status | Over the Counter |
| Company | Mayer Labs |
| 2 of 2 | |
|---|---|
| Drug Name | Today |
| PubMed Health | Nonoxynol 9 (Vaginal) |
| Drug Classes | Contraceptive, Local |
| Active Ingredient | Nonoxynol-9 |
| Dosage Form | Sponge |
| Route | Vaginal |
| Strength | 1gm |
| Market Status | Over the Counter |
| Company | Mayer Labs |
In January, 2003, FDA proposed new warning statements and other labeling information for these products after results from a major clinical study in Africa and Thailand showed that women using a contraceptive gel product containing N9 were not protected against HIV and other STDs and were, in fact, at higher risk for HIV infection than women using a placebo gel. Because these and other studies have shown that use of products containing N9 cause vaginal and rectal irritation that can heighten the chance of becoming infected with HIV from an infected partner, FDA believes the warning will empower consumers to make better informed decisions about the use of these products, and better protect the public health.
DHHS/FDA; For Consumers: New Warning for Nonoxynol 9 OTC Contraceptive Products re: STDS and HIV/AIDS (June18, 2009). Available from, as of October 16, 2012: https://www.fda.gov/ForConsumers/ByAudience/ForPatientAdvocates/HIVandAIDSActivities/ucm124023.htm
The FDA issued a final rule on December 18, 2007 requiring manufacturers of over-the-counter (OTC) stand-alone vaginal contraceptive and spermicidal products containing the chemical ingredient nonoxynol 9 (N9) include a warning stating that the chemical N9 does not provide protection against infection from HIV (the virus that causes AIDS) or other sexually transmitted diseases (STDs). Stand-alone spermicides include gels, foams, films, or inserts containing N9 that are used by themselves for contraception. Consumers can protect themselves from the transmission of STDs and HIV by practicing abstinence, being in a monogamous relationship where neither partner is infected, and using condoms consistently and correctly. FDA is issuing the rule in an effort to correct misconceptions that N9 protects against sexually transmitted diseases, including HIV infection.
DHHS/FDA; For Consumers: New Warning for Nonoxynol 9 OTC Contraceptive Products re: STDS and HIV/AIDS (June18, 2009). Available from, as of October 16, 2012: https://www.fda.gov/ForConsumers/ByAudience/ForPatientAdvocates/HIVandAIDSActivities/ucm124023.htm
Sexually transmitted diseases (STDs) alert: This product does not protect against HIV/AIDS or other STDs and may increase the risk of getting HIV from an infected partner.
DHHS/FDA; For Consumers: New Warning for Nonoxynol 9 OTC Contraceptive Products re: STDS and HIV/AIDS (June18, 2009). Available from, as of October 16, 2012: https://www.fda.gov/ForConsumers/ByAudience/ForPatientAdvocates/HIVandAIDSActivities/ucm124023.htm
Do not use if you or your sex partner has HIV/AIDS. If you do not know if you or your sex partner is infected, choose another form of birth control.
DHHS/FDA; For Consumers: New Warning for Nonoxynol 9 OTC Contraceptive Products re: STDS and HIV/AIDS (June18, 2009). Available from, as of October 16, 2012: https://www.fda.gov/ForConsumers/ByAudience/ForPatientAdvocates/HIVandAIDSActivities/ucm124023.htm
For more Drug Warnings (Complete) data for Nonoxynol-9 (7 total), please visit the HSDB record page.
Nonoxynol 9 is a surfactant spermicide used for contraception in spermicidal creams, jellies, foams, gel, and lubricants. It is also used in conjuction with other methods of contraception, including condoms, cervical caps and diaphragms.
Surface-Active Agents
Agents that modify interfacial tension of water; usually substances that have one lipophilic and one hydrophilic group in the molecule; includes soaps, detergents, emulsifiers, dispersing and wetting agents, and several groups of antiseptics. (See all compounds classified as Surface-Active Agents.)
Spermatocidal Agents
Chemical substances that are destructive to spermatozoa used as topically administered vaginal contraceptives. (See all compounds classified as Spermatocidal Agents.)
In mice intravenously injected with (14)C-Nonoxynol-9, the highest amounts of radioactivity were reported for the small and large intestines. (14)C radioactivity was excreted mainly in the urine and feces. An HPLC analysis of the urine and bile indicated that no intact Nonoxynol-9 was present in the bile or urine, and that Nonoxynol-9 was metabolized to highly polar species.
Alan AF ed.; Cosmetic Ingredient Review: Amended final report on the safety assessment of Nonoxynol -1, -2, -3, -4, -5, -6, -7 and -8. Int J Toxicol 18 (Suppl 1):11-31 (1999)
In in vitro skin penetration studies using cadaver skin (rinse off and leave-on protocols), the total skin penetration of Nonoxynol-2, -4, and -9 was less than 1 % over a period of 48 hours.
Alan AF ed.; Cosmetic Ingredient Review: Amended final report on the safety assessment of Nonoxynol -1, -2, -3, -4, -5, -6, -7 and -8. Int J Toxicol 18 (Suppl 1):11-31 (1999)
The disposition of nonoxynol-9 labeled with (14)C at the ethylene oxide units was studied following an iv or vaginal administration to female Sprague-Dawley rats. The results from the vaginal administration studies indicate 12.8% absorption of (14)C radioactivity in 6.0 hr and 37.7% in 24.0 hr. Tissue distribution studies showed that the small and large intestines, including their contents, had the highest (14)C activity by either route of administration. Radiomonitored HPLC of bile collected at 6.0 hr and urine at 6.0, 24.0, and 48.0 hr following an iv injection of (14)C nonoxynol-9 showed that the compound was completely metabolized in the body of the rat. The metabolites were primarily excreted in the feces and secondarily in the urine. Analysis of urinary metabolites containing the (14)C label, 6.0 hr following an iv dose, indicated the presence of highly polar neutral (53.27%) and acidic (39.23%) species.
PMID:2848334 Walter BA et al; Toxicol Appl Pharmacol 96 (2): 258-68 (1988)
Nonoxynol-9 interacts with the lipids in the membranes of the acrosome and the midpiece of the sperm. The sperm membranes are lysed; the acrosome, neck and midpiece of the spermatozoa are loosened and then detached which results in their immobilization and death.
Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
87
PharmaCompass offers a list of NONOXYNOL-9 API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right NONOXYNOL-9 manufacturer or NONOXYNOL-9 supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred NONOXYNOL-9 manufacturer or NONOXYNOL-9 supplier.
A Nonoxynol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Nonoxynol, including repackagers and relabelers. The FDA regulates Nonoxynol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Nonoxynol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Nonoxynol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Nonoxynol supplier is an individual or a company that provides Nonoxynol active pharmaceutical ingredient (API) or Nonoxynol finished formulations upon request. The Nonoxynol suppliers may include Nonoxynol API manufacturers, exporters, distributors and traders.
click here to find a list of Nonoxynol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Nonoxynol DMF (Drug Master File) is a document detailing the whole manufacturing process of Nonoxynol active pharmaceutical ingredient (API) in detail. Different forms of Nonoxynol DMFs exist exist since differing nations have different regulations, such as Nonoxynol USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Nonoxynol DMF submitted to regulatory agencies in the US is known as a USDMF. Nonoxynol USDMF includes data on Nonoxynol's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Nonoxynol USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Nonoxynol suppliers with USDMF on PharmaCompass.
A Nonoxynol CEP of the European Pharmacopoeia monograph is often referred to as a Nonoxynol Certificate of Suitability (COS). The purpose of a Nonoxynol CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Nonoxynol EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Nonoxynol to their clients by showing that a Nonoxynol CEP has been issued for it. The manufacturer submits a Nonoxynol CEP (COS) as part of the market authorization procedure, and it takes on the role of a Nonoxynol CEP holder for the record. Additionally, the data presented in the Nonoxynol CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Nonoxynol DMF.
A Nonoxynol CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Nonoxynol CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Nonoxynol suppliers with CEP (COS) on PharmaCompass.
Nonoxynol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Nonoxynol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Nonoxynol GMP manufacturer or Nonoxynol GMP API supplier for your needs.
A Nonoxynol CoA (Certificate of Analysis) is a formal document that attests to Nonoxynol's compliance with Nonoxynol specifications and serves as a tool for batch-level quality control.
Nonoxynol CoA mostly includes findings from lab analyses of a specific batch. For each Nonoxynol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Nonoxynol may be tested according to a variety of international standards, such as European Pharmacopoeia (Nonoxynol EP), Nonoxynol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Nonoxynol USP).

