
Synopsis
Synopsis
0
JDMF
0
VMF
0
Australia
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Afazol Grin
2. Ak Con
3. Ak-con
4. Albalon
5. All Clear
6. Clear Eyes
7. Colirio Alfa
8. Hydrochloride, Naphazoline
9. Idril
10. Miraclar
11. Monohydrochloride, Naphazoline
12. Nafazair
13. Naphazoline
14. Naphazoline Monohydrochloride
15. Naphazoline Nitrate
16. Naphcon
17. Naphcon Forte
18. Nitrate, Naphazoline
19. Optazine
20. Pensa, Vasoconstrictor
21. Privin
22. Privine
23. Proculin
24. Siozwo
25. Tele Stulln
26. Tele-stulln
27. Vasoclear
28. Vasocon
29. Vasocon Regular
30. Vasoconstrictor Pensa
31. Vasonit
1. 550-99-2
2. Naphazoline Hcl
3. Albalon
4. Rhinantin
5. Vasocon
6. Opcon
7. Albalon Liquifilm
8. Rhinoperd
9. Stricylon
10. Naphcon
11. Niazol
12. Rinofug
13. Naphcon Forte
14. 2-(1-naphthylmethyl)-2-imidazoline Hydrochloride
15. Clera Hydrochloride
16. Naphazoline Chloride
17. Privine Hydrochloride
18. Prizole Hydrochloride
19. Nafazair
20. Sanorin
21. 2-(1-naphthylmethyl)imidazoline Hydrochloride
22. Naphazoline (hydrochloride)
23. Nsc-35711
24. 2-(naphthalen-1-ylmethyl)-4,5-dihydro-1h-imidazole Hydrochloride
25. 2-(1-naphthylmethyl)-2-imidazoline Monohydrochloride
26. 1h-imidazole, 4,5-dihydro-2-(1-naphthalenylmethyl)-, Monohydrochloride
27. Mls000028434
28. Chebi:7470
29. Naphazoline Hydrochloride (naphcon)
30. Mz1131787d
31. Smr000058292
32. Vasoclear
33. Coldan
34. 2-(naphthalen-1-ylmethyl)-4,5-dihydro-1h-imidazole;hydrochloride
35. Comfort Eye Drops
36. Degest-2
37. Naphazoliniumchlorid
38. Naphazolini Chloridum
39. Naphthasolium Chloride
40. Chembl1706
41. Clear Eyes
42. Naphazoline Hydrochloride [jan]
43. Hsdb 2174
44. Naphthylmethylimidazolinhydrochlorid
45. Einecs 208-989-2
46. Mfcd00012554
47. Nsc 35711
48. Albacon
49. 2-(1-naphthylmethyl)-4,5-dihydro-1h-imidazole
50. Sr-01000000067
51. Unii-mz1131787d
52. Prestwick_507
53. Albalon (tn)
54. Vasocon (tn)
55. Component Of Nasocon
56. Naphazoline Hydrochloride [usp:jan]
57. 2-imidazoline, 2-(1-naphthylmethyl)-, Monohydrochloride
58. Opera_id_230
59. Ac1lcwb1
60. Surecn1649580
61. Schembl24153
62. Mls001148448
63. Mls002222217
64. Naphazolinehydrochloride
65. Naphazoline Hydrochloride,(s)
66. Spectrum1500424
67. 2-(1-naphthylmethyl)-2-imidazoline Hydro- Chloride
68. Naphazoline Hcl [inci]
69. 2-imidazoline, Monohydrochloride
70. Dtxsid7045788
71. Molport-000-703-064
72. Hms1568i13
73. Hms1920p11
74. Pharmakon1600-01500424
75. Amy23173
76. Ex-a4103
77. Hy-b0446
78. Nsc35711
79. Ccg-40122
80. Nsc757110
81. S2519
82. Naphazoline Hydrochloride [mi]
83. Akos015908797
84. Naphazoline Hydrochloride (jp17/usp)
85. Nc00523
86. Nsc-757110
87. Naphazoline Hydrochloride [hsdb]
88. Naphazoline Hydrochloride [mart.]
89. Naphazoline Hydrochloride [vandf]
90. Ncgc00094732-01
91. Ncgc00094732-02
92. Ncgc00094732-03
93. Ncgc00094732-04
94. As-16861
95. Naphazoline Hydrochloride [usp-rs]
96. Naphazoline Hydrochloride [who-dd]
97. 2-(naphthalen-1-ylmethyl)-4,5-dihydro-
98. Wln: L66j B1- Bt5m Cn Butj &gh
99. Ft-0603466
100. Lt00114215
101. Lt00233108
102. N0542
103. Sw196701-3
104. C07898
105. D00743
106. H12006
107. Naphazoline Hydrochloride [ep Impurity]
108. Naphazoline Hydrochloride [ep Monograph]
109. Naphazoline Hydrochloride [orange Book]
110. Naphazoline Hydrochloride [usp Monograph]
111. A830484
112. Opcon-a Component Naphazoline Hydrochloride
113. Visine-a Component Naphazoline Hydrochloride
114. Naphcon-a Component Naphazoline Hydrochloride
115. Sr-01000000067-3
116. Vasocon-a Component Naphazoline Hydrochloride
117. W-105587
118. Naphazoline Hydrochloride Component Of Opcon-a
119. Naphazoline Hydrochloride Component Of Visine-a
120. Q27887678
121. Naphazoline Hydrochloride Component Of Naphcon-a
122. Naphazoline Hydrochloride Component Of Vasocon-a
123. 4,5-dihydro-2-(1-naphthylmethyl)-1h-imidazole Hydrochloride
124. 1h-imidazole,5-dihydro-2-(1-naphthalenylmethyl)-, Monohydrochloride
125. Hydron;2-(naphthalen-1-ylmethyl)-4,5-dihydro-1h-imidazole;chloride
126. 1h-imidazole, 4,5-dihydro-2-(1-naphthalenylmethyl)-, Hydrochloride (1:1)
127. Naphazoline Hydrochloride, European Pharmacopoeia (ep) Reference Standard
128. Naphazoline Hydrochloride, United States Pharmacopeia (usp) Reference Standard
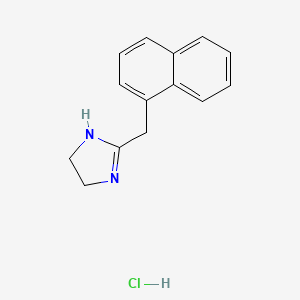
1. Naphazoline
| Molecular Weight | 246.73 g/mol |
|---|---|
| Molecular Formula | C14H15ClN2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 2 |
| Exact Mass | 246.0923762 g/mol |
| Monoisotopic Mass | 246.0923762 g/mol |
| Topological Polar Surface Area | 24.4 Ų |
| Heavy Atom Count | 17 |
| Formal Charge | 0 |
| Complexity | 272 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Naphazoline hydrochloride |
| Drug Label | Naphazoline Hydrochloride, an ocular vasoconstrictor, is an imidazoline derivative sympathomimetic amine. It occurs as a white, odorless crystalline powder having a bitter taste and is freely soluble in water and in alcohol. The active ingredient is... |
| Active Ingredient | Naphazoline hydrochloride |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | 0.1% |
| Market Status | Prescription |
| Company | Akorn |
| 2 of 2 | |
|---|---|
| Drug Name | Naphazoline hydrochloride |
| Drug Label | Naphazoline Hydrochloride, an ocular vasoconstrictor, is an imidazoline derivative sympathomimetic amine. It occurs as a white, odorless crystalline powder having a bitter taste and is freely soluble in water and in alcohol. The active ingredient is... |
| Active Ingredient | Naphazoline hydrochloride |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | 0.1% |
| Market Status | Prescription |
| Company | Akorn |
Adrenergic alpha-Agonists; Nasal Decongestants
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Naphazoline is applied topically to the conjunctiva to temporarily relieve congestion, itching, and minor irritation. Ocular decongestants are ineffective in the treatment of delayed hypersensitivity reactions such as contact dermatoconjunctivitis. The vasoconstrictor effects of naphazoline may be used during some ocular diagnostic procedures, but some clinicians prefer phenylephrine to naphazoline for this use.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2823
Ophthalmic solutions containing naphazoline in combination with antihistamines such as antazoline phosphate or pheniramine maleate and/or astringents such as zinc sulfate are commercially available. In the concentrations usually employed, zinc sulfate is a relatively ineffective antiseptic and may promote vasodilation.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2824
Adrenergic (vasoconstrictor); decongestant.
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. 13th Edition, Whitehouse Station, NJ: Merck and Co., Inc., 2001., p. 1142
Naphazoline hydrochloride (0.1%), an imidazole derivative with preferential alpha-2 activity, /was instilled/ in 17 eyes of 12 patients with myopathic ptosis due to involvement of the levator palpebrae superioris, in the attempt to selectively stimulate Muller's smooth muscle. Naphazoline significantly widened the palpebral fissure with little change in pupillary diameter and no significant change in ocular pressure, visual acuity and near point determination. However, a reduction of the effect, probably due to tachyphylaxis, was noticed when using naphazoline regularly several times a day for few weeks. In conclusion naphazoline has powerful cosmetical and functional effects in mild to moderate myopathic ptosis above all if taken occasionally.
PMID:8503264 Uncini A et al; Acta Neurol Scand 87 (4): 322-4 (1993)
Ophthalmic use of naphazoline may occasionally cause systemic sympathomimetic effects such as headache, hypertension, cardiac irregularities, nervousness, nausea, dizziness, weakness, and sweating.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2824
Use of naphazoline in the eye may cause blurred vision, mild transient stinging and/or irritation, mydriasis, and increased or decreased intraocular pressure. Conjunctival application of naphazoline, especially when high concentrations are used in geriatric patients, may liberate pigment granules, presumably from the iris. Rebound congestion, characterized by reactive hyperemia frequently occurs with prolonged use and may result in overuse of the drug. Prolonged use of the drug should be avoided for these reasons.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2824
The incidence of serious adverse effects is low in patients receiving therapeutic dosages of ophthalmic solutions of naphazoline hydrochloride. When naphazoline hydrochloride is used in combination preparations, the cautions applicable to each ingredient in the formulation must be observed. Excessive dosage and/or prolonged or too frequent use may irritate the conjunctiva and, especially in children, cause adverse systemic effects.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2824
Naphazoline hydrochloride ophthalmic solution should be used with caution in patients with hypertension, cardiovascular abnormalities, diabetes mellitus, hyperthyroidism, infection, or injury.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2824
For more Drug Warnings (Complete) data for NAPHAZOLINE HYDROCHLORIDE (14 total), please visit the HSDB record page.
Adrenergic alpha-Agonists
Drugs that selectively bind to and activate alpha adrenergic receptors. (See all compounds classified as Adrenergic alpha-Agonists.)
Nasal Decongestants
Drugs designed to treat inflammation of the nasal passages, generally the result of an infection (more often than not the common cold) or an allergy related condition, e.g., hay fever. The inflammation involves swelling of the mucous membrane that lines the nasal passages and results in inordinate mucus production. The primary class of nasal decongestants are vasoconstrictor agents. (From PharmAssist, The Family Guide to Health and Medicine, 1993) (See all compounds classified as Nasal Decongestants.)
Following topical application of naphazoline hydrochloride solutions to the conjunctiva, local vasoconstriction usually occurs within 10 minutes and may persist for 2-6 hours. Occasionally, enough naphazoline may be absorbed to produce systemic effects. Information on the distribution and elimination of the drug in humans is not available
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2824
Naphazoline constricts the vascular system of the conjunctiva. It is presumed that this effect is due to direct stimulation action of the drug upon the alpha-adrenergic receptors in the arterioles of the conjunctiva, resulting in decreased conjunctival congestion.
Physician's Desk Reference for Ophthalmic Medicines. 35rd Edition.Thomson PDR. Montvale, NJ. p. 219 (2006)
The mechanism of action of naphazoline has not been conclusively determined. Most pharmacologists believe that the drug directly stimulates alpha-adrenergic receptors of the sympathetic nervous system and exerts little or no effect on beta-adrenergic receptors. Following topical application of naphazoline to the conjunctiva, small arterioles are constricted and conjunctival congestion is temporarily relieved, but reactive hyperemia may occur. The drug also may produce mydriasis when applied to the conjunctiva, but this effect is usually minimal with the concentrations used as ocular decongestants.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2006., p. 2824
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
72
PharmaCompass offers a list of Naphazoline Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Naphazoline Hydrochloride manufacturer or Naphazoline Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Naphazoline Hydrochloride manufacturer or Naphazoline Hydrochloride supplier.
A Naphazoline HCl manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Naphazoline HCl, including repackagers and relabelers. The FDA regulates Naphazoline HCl manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Naphazoline HCl API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Naphazoline HCl manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Naphazoline HCl supplier is an individual or a company that provides Naphazoline HCl active pharmaceutical ingredient (API) or Naphazoline HCl finished formulations upon request. The Naphazoline HCl suppliers may include Naphazoline HCl API manufacturers, exporters, distributors and traders.
click here to find a list of Naphazoline HCl suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Naphazoline HCl DMF (Drug Master File) is a document detailing the whole manufacturing process of Naphazoline HCl active pharmaceutical ingredient (API) in detail. Different forms of Naphazoline HCl DMFs exist exist since differing nations have different regulations, such as Naphazoline HCl USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Naphazoline HCl DMF submitted to regulatory agencies in the US is known as a USDMF. Naphazoline HCl USDMF includes data on Naphazoline HCl's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Naphazoline HCl USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Naphazoline HCl suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Naphazoline HCl Drug Master File in Korea (Naphazoline HCl KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Naphazoline HCl. The MFDS reviews the Naphazoline HCl KDMF as part of the drug registration process and uses the information provided in the Naphazoline HCl KDMF to evaluate the safety and efficacy of the drug.
After submitting a Naphazoline HCl KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Naphazoline HCl API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Naphazoline HCl suppliers with KDMF on PharmaCompass.
A Naphazoline HCl CEP of the European Pharmacopoeia monograph is often referred to as a Naphazoline HCl Certificate of Suitability (COS). The purpose of a Naphazoline HCl CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Naphazoline HCl EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Naphazoline HCl to their clients by showing that a Naphazoline HCl CEP has been issued for it. The manufacturer submits a Naphazoline HCl CEP (COS) as part of the market authorization procedure, and it takes on the role of a Naphazoline HCl CEP holder for the record. Additionally, the data presented in the Naphazoline HCl CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Naphazoline HCl DMF.
A Naphazoline HCl CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Naphazoline HCl CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Naphazoline HCl suppliers with CEP (COS) on PharmaCompass.
A Naphazoline HCl written confirmation (Naphazoline HCl WC) is an official document issued by a regulatory agency to a Naphazoline HCl manufacturer, verifying that the manufacturing facility of a Naphazoline HCl active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Naphazoline HCl APIs or Naphazoline HCl finished pharmaceutical products to another nation, regulatory agencies frequently require a Naphazoline HCl WC (written confirmation) as part of the regulatory process.
click here to find a list of Naphazoline HCl suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Naphazoline HCl as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Naphazoline HCl API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Naphazoline HCl as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Naphazoline HCl and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Naphazoline HCl NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Naphazoline HCl suppliers with NDC on PharmaCompass.
Naphazoline HCl Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Naphazoline HCl GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Naphazoline HCl GMP manufacturer or Naphazoline HCl GMP API supplier for your needs.
A Naphazoline HCl CoA (Certificate of Analysis) is a formal document that attests to Naphazoline HCl's compliance with Naphazoline HCl specifications and serves as a tool for batch-level quality control.
Naphazoline HCl CoA mostly includes findings from lab analyses of a specific batch. For each Naphazoline HCl CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Naphazoline HCl may be tested according to a variety of international standards, such as European Pharmacopoeia (Naphazoline HCl EP), Naphazoline HCl JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Naphazoline HCl USP).

