
Synopsis
Synopsis
0
CEP/COS
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
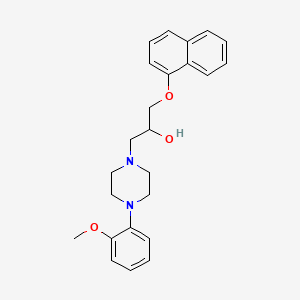

1. 1-(4-(2-methoxyphenyl)-1-piperazinyl)-3-(1-naphthyloxy)-2-propanol
1. 57149-07-2
2. Flivas
3. Kt-611
4. Naftopidil [inn]
5. Avishot
6. 1-(4-(2-methoxyphenyl)piperazin-1-yl)-3-(naphthalen-1-yloxy)propan-2-ol
7. Bm-15275
8. Naftopidil (flivas)
9. R9phw59sfn
10. Nsc-759293
11. Chembl142635
12. Ncgc00015718-06
13. 1-[4-(2-methoxyphenyl)piperazin-1-yl]-3-(naphthalen-1-yloxy)propan-2-ol
14. Dsstox_cid_25176
15. Dsstox_rid_80725
16. Dsstox_gsid_45176
17. Naftopidilum [latin]
18. Naftopidilum
19. 4-(2-methoxyphenyl)-alpha-[(1-naphthalenyloxy)methyl]-1-piperazineethanol
20. Naftopidil Hydrochloride Hydrate
21. Smr000466346
22. Cas-57149-07-2
23. Naftopidil (unspecified)
24. Unii-r9phw59sfn
25. Brn 0629965
26. Naftopidil,(s)
27. Flivas (tn)
28. Naftopidil [mi]
29. Naftopidil [jan]
30. Prestwick0_000975
31. Prestwick1_000975
32. Prestwick2_000975
33. Prestwick3_000975
34. (+-)-1-(4-(2-methoxyphenyl)piperazinyl)-3-(1-naphthyloxy)propan-2-ol
35. Naftopidil (jp17/inn)
36. (rs)-1-(4-(2-methoxyphenyl)-1-piperazinyl)-3-(1-naphthyloxy)-2-propanol
37. 4-(2-methoxyphenyl)-alpha-((1-naphthalenyloxy)methyl)-1-pioerazineethanol
38. Cid_4418
39. Naftopidil [mart.]
40. Naftopidil [who-dd]
41. Lopac0_000941
42. Regid_for_cid_4418
43. Bspbio_001009
44. Mls000759459
45. Mls001424117
46. Schembl113215
47. Spbio_002920
48. Bpbio1_001111
49. Dtxsid5045176
50. Bdbm50773
51. Chebi:31891
52. Cid_6603044
53. Hms2051b09
54. Hms2089g07
55. Hms3393b09
56. Hms3655h21
57. Hms3884p07
58. Pharmakon1600-01506024
59. Bcp21786
60. Hy-b0391
61. 3-(naphthalen-1-yloxy)propan-2-ol
62. Tox21_110205
63. Bbl028454
64. Mfcd00242741
65. Nsc759293
66. S2126
67. Stl372645
68. (+-)-4-(o-methoxyphenyl)-alpha-((1-naphthyloxy)methyl)-1-piperazineethanol
69. 1-[4-(2-methoxyphenyl)piperazin-1-yl]-3-naphthalen-1-yloxypropan-2-ol
70. 1-pioerazineethanol, 4-(2-methoxyphenyl)-alpha-((1-naphthalenyloxy)methyl)-
71. Akos002279617
72. Akos016328058
73. Tox21_110205_1
74. Bcp9000976
75. Ccg-100935
76. Ccg-205022
77. Db12092
78. Fe-0209
79. Nc00185
80. Nsc 759293
81. Sb19321
82. Sdccgsbi-0050915.p003
83. Ncgc00015718-04
84. Ncgc00015718-05
85. Ncgc00015718-08
86. Ncgc00015718-09
87. Ncgc00015718-10
88. Ncgc00015718-12
89. Ncgc00015718-25
90. Ncgc00024672-03
91. Ncgc00024672-04
92. Bn164631
93. 1-(4-(2-methoxyphenyl)piperazin-1-yl)-
94. Sbi-0050915.p002
95. Ab00514643
96. Ft-0630696
97. N0832
98. Sw196570-4
99. 49n072
100. D01674
101. Ab00489961-18
102. Ab00489961-19
103. Ab00489961_20
104. Ab00489961_21
105. A831328
106. Q6958243
107. Brd-a01787639-300-03-7
108. Brd-a01787639-300-04-5
109. 1-[4-(2-methoxyphenyl)piperazinyl]-3-(1-naphthyloxy)propan-2-ol
110. 1-[4-(2-methoxyphenyl)-1-piperazinyl]-3-(1-naphthalenyloxy)-2-propanol
111. 1-[4-(2-methoxyphenyl)piperazino]-3-(1-naphthoxy)propan-2-ol;hydrochloride
112. Bm-15275; Kt-611; Bm 15275; Kt 611; Bm15275; Kt611
113. (+/-)-4-(o-methoxyphenyl)-.alpha.-((1-naphthyloxy)methyl)-1-piperazineethanol
114. 1-[4-(2-methoxyphenyl)-1-piperazinyl]-3-(1-naphthalenyloxy)-2-propanol;hydrochloride
115. 1-[4-(2-methoxyphenyl)piperazin-1-yl]-3-naphthalen-1-yloxy-propan-2-ol;hydrochloride
116. 1-[4-(2-methoxyphenyl)piperazin-1-yl]-3-naphthalen-1-yloxypropan-2-ol;hydrochloride
| Molecular Weight | 392.5 g/mol |
|---|---|
| Molecular Formula | C24H28N2O3 |
| XLogP3 | 4.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Exact Mass | 392.20999276 g/mol |
| Monoisotopic Mass | 392.20999276 g/mol |
| Topological Polar Surface Area | 45.2 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 483 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
Adrenergic alpha-Antagonists
Drugs that bind to but do not activate alpha-adrenergic receptors thereby blocking the actions of endogenous or exogenous adrenergic agonists. Adrenergic alpha-antagonists are used in the treatment of hypertension, vasospasm, peripheral vascular disease, shock, and pheochromocytoma. (See all compounds classified as Adrenergic alpha-Antagonists.)
Calcium Channel Blockers
A class of drugs that act by selective inhibition of calcium influx through cellular membranes. (See all compounds classified as Calcium Channel Blockers.)
Platelet Aggregation Inhibitors
Drugs or agents which antagonize or impair any mechanism leading to blood platelet aggregation, whether during the phases of activation and shape change or following the dense-granule release reaction and stimulation of the prostaglandin-thromboxane system. (See all compounds classified as Platelet Aggregation Inhibitors.)
ANALYTICAL

ABOUT THIS PAGE
80
PharmaCompass offers a list of Naftopidil API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Naftopidil manufacturer or Naftopidil supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Naftopidil manufacturer or Naftopidil supplier.
A Naftopidil manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Naftopidil, including repackagers and relabelers. The FDA regulates Naftopidil manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Naftopidil API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Naftopidil manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Naftopidil supplier is an individual or a company that provides Naftopidil active pharmaceutical ingredient (API) or Naftopidil finished formulations upon request. The Naftopidil suppliers may include Naftopidil API manufacturers, exporters, distributors and traders.
click here to find a list of Naftopidil suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Naftopidil DMF (Drug Master File) is a document detailing the whole manufacturing process of Naftopidil active pharmaceutical ingredient (API) in detail. Different forms of Naftopidil DMFs exist exist since differing nations have different regulations, such as Naftopidil USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Naftopidil DMF submitted to regulatory agencies in the US is known as a USDMF. Naftopidil USDMF includes data on Naftopidil's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Naftopidil USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Naftopidil suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Naftopidil Drug Master File in Japan (Naftopidil JDMF) empowers Naftopidil API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Naftopidil JDMF during the approval evaluation for pharmaceutical products. At the time of Naftopidil JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Naftopidil suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Naftopidil Drug Master File in Korea (Naftopidil KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Naftopidil. The MFDS reviews the Naftopidil KDMF as part of the drug registration process and uses the information provided in the Naftopidil KDMF to evaluate the safety and efficacy of the drug.
After submitting a Naftopidil KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Naftopidil API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Naftopidil suppliers with KDMF on PharmaCompass.
A Naftopidil written confirmation (Naftopidil WC) is an official document issued by a regulatory agency to a Naftopidil manufacturer, verifying that the manufacturing facility of a Naftopidil active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Naftopidil APIs or Naftopidil finished pharmaceutical products to another nation, regulatory agencies frequently require a Naftopidil WC (written confirmation) as part of the regulatory process.
click here to find a list of Naftopidil suppliers with Written Confirmation (WC) on PharmaCompass.
Naftopidil Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Naftopidil GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Naftopidil GMP manufacturer or Naftopidil GMP API supplier for your needs.
A Naftopidil CoA (Certificate of Analysis) is a formal document that attests to Naftopidil's compliance with Naftopidil specifications and serves as a tool for batch-level quality control.
Naftopidil CoA mostly includes findings from lab analyses of a specific batch. For each Naftopidil CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Naftopidil may be tested according to a variety of international standards, such as European Pharmacopoeia (Naftopidil EP), Naftopidil JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Naftopidil USP).

