
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Australia
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
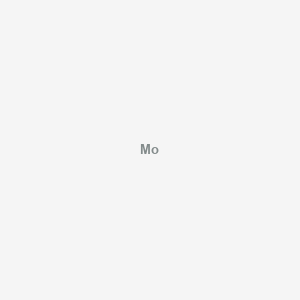

1. Molybdenum 98
2. Molybdenum-98
1. 7439-98-7
2. Mo
3. Molybdenum(6+)
4. Molybdenum Atom
5. Molybdenum(6+) Ion
6. Molybdenum, Metallic
7. Molybdenum(vi) Ion
8. Molybdenum, Elemental
9. Molybdenum(5+)
10. Molybdenum Metallicum
11. Molybdenum, Ion (mo(6+))
12. Molybdenum (vi) Ion
13. Mo6+
14. Molybdenum, Ion (mo5+)
15. Molybdenum Wire
16. Mo(6+)
17. Mfcd00003465
18. 81ah48963u
19. Molybdenum Powder
20. Mchvl
21. 6mo
22. Metco 63
23. Tsm1
24. Molibdeno
25. Molybdenum Wire, Platinum Coated, 0.457mm (0.018in) Dia
26. Amperit 106.2
27. Molybdenum Metal
28. Amperit 105.054
29. Molybdenum Gauze, 10 Mesh Woven From 0.38mm (0.015mm) Dia Wire
30. Molybdenum Gauze, 20 Mesh Woven From 0.305mm (0.012in) Dia Wire
31. Molybdenum Gauze, 30 Mesh Woven From 0.204mm (0.008in) Dia Wire
32. Molybdenum Gauze, 50 Mesh Woven From 0.0509mm (0.002in) Dia Wire
33. Molybdenum Gauze, 100 Mesh Woven From 0.0636mm (0.0025in) Dia Wire
34. Molybdenum Gauze, 165 Mesh Woven From 0.0509mm (0.002in) Dia Wire, Twilled Weave
35. 4mo
36. Hsdb 5032
37. Einecs 231-107-2
38. Molybdenum Rod
39. Molybdenum, Insoluble Compounds
40. Molybdenum Sheet
41. Nitinol Rod
42. Molybdenum Pellet
43. Molybdenum, Crucible, Cylindrical, Length 1 Pcs, Outside Diameter 47mm
44. Molybdenum, Foil, Thickness 0.015 Mm, Size 200 X 200 Mm, Purity 99.9%
45. Molybdenum, Foil, Thickness 0.020 Mm, Size 200 X 200 Mm, Purity 99.9%
46. Molybdenum, Foil, Thickness 0.025 Mm, Size 200 X 200 Mm, Purity 99.9%
47. Molybdenum, Foil, Thickness 0.03 Mm, Size 200 X 200 Mm, Purity 99.9%
48. Molybdenum, Foil, Thickness 0.03 Mm, Size 200 X 500 Mm, Purity 99.9%
49. Unii-81ah48963u
50. Molybdenum Element
51. Molybdenum Hydride
52. Molybdenum Pellets
53. Molybdenum(vi)
54. Molybdenum Nanorods
55. Molybdenum(1+)
56. Molybdenum(5 )
57. Molybdenum Compounds
58. Molybdenum Trihydride
59. Molybdenum Nanopowder
60. Molybdenum(5+) Ion
61. Molybdenum(v) Cation
62. Molybdenum, Compounds
63. Molybdenum Ion(6+)
64. Molybdenum Pentahydride
65. Molybdenum(vi) Cation
66. Molybdenum Foil, 3n5
67. Molybdenum Nanoparticles
68. Molybdenum, Metal And Insoluble Compounds
69. Molybdenum And Compounds
70. Molybdenum [mi]
71. Molybdenum [hsdb]
72. Molybdenum Nano Dispersion
73. Molybdenum [vandf]
74. 42mo
75. Ec 231-107-2
76. Molybdenum [mart.]
77. Molybdenum [who-dd]
78. Ultra Thin Molybdenum Nanofoil
79. Dtxsid1024207
80. Chebi:28685
81. Chebi:30511
82. Chebi:49446
83. Hsdb 7540
84. Mo(5+)
85. Molybdenum, 99.99%
86. Dtxsid00872441
87. Dtxsid40872440
88. Dtxsid901317203
89. Nsc622116
90. Nsc622124
91. Akos015902698
92. Db11137
93. Molybdenum Rod, 12.7mm (0.5in) Dia
94. Molybdenum Rod, 6.4mm (0.25in) Dia
95. Molybdenum, Ion (mo6+) (8ci,9ci)
96. Molybdenum Powder, Spherical, -170 Mesh
97. Molybdenum Foil, 1.0mm (0.04in) Thick
98. Molybdenum Foil, 0.1mm (0.004in) Thick
99. Molybdenum Foil, 0.5mm (0.02 In) Thick
100. Ft-0695133
101. Molybdenum Foil, 0.05mm (0.002in) Thick
102. Molybdenum Plate, 2.5mm (0.098in) Thick
103. Q1053
104. Molybdenum (mo), 99.5% (metal Basis) Nano
105. Molybdenum Foil, 0.025mm (0.001in) Thick
106. Molybdenum Foil, 0.127mm (0.005in) Thick
107. Molybdenum Plate, 12.5mm (0.492in) Thick
108. Molybdenum Plate, 4.76mm (0.188in) Thick
109. C00150
110. A838110
111. Lithium Cobalt Oxide (licoo2) Sputtering Targets
112. Molybdenum Wire, 0.025mm (0.001in) Dia, Hard
113. Molybdenum Wire, 0.203mm (0.008in) Dia, Hard
114. Molybdenum Wire, 0.25mm (0.01in) Dia, Annealed
115. Molybdenum Wire, 0.5mm (0.02in) Dia, Annealed
116. Molybdenum Wire, 1.0mm (0.04in) Dia, Annealed
117. Molybdenum Wire, 1.6mm (0.063in) Dia, Annealed
118. Molybdenum Wire, 2.0mm (0.08in) Dia, Annealed
119. Molybdenum Foil, 0.25mm (0.01in) Thick, Annealed
120. Molybdenum Wire, 1.14mm (0.045in) Dia, Annealed
121. Molybdenum, Rod, 50mm, Diameter 5.0mm, 99.98%
122. Molybdenum Wire, 0.013mm (0.00052in) Dia, Hard
123. Molybdenum Wire, 0.127mm (0.005in) Dia, Annealed
124. Molybdenum Wire, 0.375mm (0.015in) Dia, Annealed
125. Molybdenum Wire, 1.58mm (0.0625in) Dia, Annealed
126. Molybdenum, Pellets, 100g, Max. Size 10mm, 99.9%
127. Molybdenum, Pellets, 200g, Max. Size 10mm, 99.9%
128. Molybdenum, Pellets, 500g, Max. Size 10mm, 99.9%
129. Molybdenum, Pellets, 50g, Max. Size 10mm, 99.9%
130. Molybdenum, Rod, 100mm, Diameter 5.0mm, 99.98%
131. Molybdenum, Rod, 150mm, Diameter 5.0mm, 99.98%
132. Q27104656
133. Q27113829
134. M087500000
135. Molybdenum Wire, Gold Plated, 0.025mm (0.001in) Dia
136. Molybdenum, Foil, 1m Coil, Thickness 0.008mm, 99.9%
137. Molybdenum, Foil, 1m Coil, Thickness 0.01mm, 99.9%
138. Molybdenum, Foil, 4mm Disks, Thickness 0.01mm, 99.9%
139. Molybdenum, Foil, 6mm Disks, Thickness 0.01mm, 99.9%
140. Molybdenum, Foil, 8mm Disks, Thickness 0.01mm, 99.9%
141. Molybdenum Boat, Thickness (mm), 0.05, Length (mm), 75
142. Molybdenum Rod, 5mm (0.2in) Dia, 99.97% (metals Basis)
143. Molybdenum Standard For Aas, Ready-to-use, In Nitric Acid
144. Molybdenum, Foil, 0.2m Coil, Thickness 0.008mm, 99.9%
145. Molybdenum, Foil, 0.2m Coil, Thickness 0.01mm, 99.9%
146. Molybdenum, Foil, 0.5m Coil, Thickness 0.008mm, 99.9%
147. Molybdenum, Foil, 0.5m Coil, Thickness 0.01mm, 99.9%
148. Molybdenum, Foil, 10mm Disks, Thickness 0.005mm, 99.9%
149. Molybdenum, Foil, 10mm Disks, Thickness 0.006mm, 99.9%
150. Molybdenum, Foil, 10mm Disks, Thickness 0.007mm, 99.9%
151. Molybdenum, Foil, 10mm Disks, Thickness 0.008mm, 99.9%
152. Molybdenum, Foil, 10mm Disks, Thickness 0.009mm, 99.9%
153. Molybdenum, Foil, 10mm Disks, Thickness 0.01mm, 99.9%
154. Molybdenum, Foil, 15mm Disks, Thickness 0.005mm, 99.9%
155. Molybdenum, Foil, 15mm Disks, Thickness 0.006mm, 99.9%
156. Molybdenum, Foil, 15mm Disks, Thickness 0.007mm, 99.9%
157. Molybdenum, Foil, 15mm Disks, Thickness 0.008mm, 99.9%
158. Molybdenum, Foil, 15mm Disks, Thickness 0.009mm, 99.9%
159. Molybdenum, Foil, 15mm Disks, Thickness 0.01mm, 99.9%
160. Molybdenum, Foil, 25mm Disks, Thickness 0.008mm, 99.9%
161. Molybdenum, Foil, 25mm Disks, Thickness 0.01mm, 99.9%
162. Molybdenum, Foil, 4mm Disks, Thickness 0.005mm, 99.9%
163. Molybdenum, Foil, 4mm Disks, Thickness 0.006mm, 99.9%
164. Molybdenum, Foil, 4mm Disks, Thickness 0.007mm, 99.9%
165. Molybdenum, Foil, 4mm Disks, Thickness 0.008mm, 99.9%
166. Molybdenum, Foil, 4mm Disks, Thickness 0.009mm, 99.9%
167. Molybdenum, Foil, 4mm Disks, Thickness 0.0125mm, 99.9%
168. Molybdenum, Foil, 50mm Disks, Thickness 0.008mm, 99.9%
169. Molybdenum, Foil, 50mm Disks, Thickness 0.01mm, 99.9%
170. Molybdenum, Foil, 6mm Disks, Thickness 0.005mm, 99.9%
171. Molybdenum, Foil, 6mm Disks, Thickness 0.006mm, 99.9%
172. Molybdenum, Foil, 6mm Disks, Thickness 0.007mm, 99.9%
173. Molybdenum, Foil, 6mm Disks, Thickness 0.008mm, 99.9%
174. Molybdenum, Foil, 6mm Disks, Thickness 0.009mm, 99.9%
175. Molybdenum, Foil, 6mm Disks, Thickness 0.0125mm, 99.9%
176. Molybdenum, Foil, 8mm Disks, Thickness 0.005mm, 99.9%
177. Molybdenum, Foil, 8mm Disks, Thickness 0.006mm, 99.9%
178. Molybdenum, Foil, 8mm Disks, Thickness 0.007mm, 99.9%
179. Molybdenum, Foil, 8mm Disks, Thickness 0.008mm, 99.9%
180. Molybdenum, Foil, 8mm Disks, Thickness 0.009mm, 99.9%
181. Molybdenum, Foil, 8mm Disks, Thickness 0.0125mm, 99.9%
182. Molybdenum, Nut, Size M3, Length 10 Pcs, Style Hexagonal
183. Molybdenum, Nut, Size M4, Length 25 Pcs, Style Hexagonal
184. Molybdenum, Nut, Size M4, Length 50 Pcs, Style Hexagonal
185. Molybdenum, Nut, Size M5, Length 25 Pcs, Style Hexagonal
186. Molybdenum, Nut, Size M5, Length 50 Pcs, Style Hexagonal
187. Molybdenum, Nut, Size M6, Length 25 Pcs, Style Hexagonal
188. Molybdenum, Nut, Size M6, Length 50 Pcs, Style Hexagonal
189. Molybdenum, Powder, <150 Mum, 99.9% Trace Metals Basis
190. Molybdenum, Powder, <150 Mum, 99.99% Trace Metals Basis
191. Molybdenum, Powder, 1-5 Mum, >=99.9% Trace Metals Basis
192. Molybdenum, Powder, 10 Mum, >=99.95% Trace Metals Basis
193. Molybdenum, Aas Standard Solution, Specpure?, Mo 1000?g/ml
194. Molybdenum, Foil, 0.2m Coil, Thickness 0.0125mm, 99.9%
195. Molybdenum, Foil, 0.5m Coil, Thickness 0.0125mm, 99.9%
196. Molybdenum, Foil, 10mm Disks, Thickness 0.0125mm, 99.9%
197. Molybdenum, Foil, 15mm Disks, Thickness 0.0125mm, 99.9%
198. Molybdenum, Foil, 1m Coil, Thickness 0.1mm, Annealed, 99.9%
199. Molybdenum, Foil, 25mm Disks, Thickness 0.0125mm, 99.9%
200. Molybdenum, Foil, 50mm Disks, Thickness 0.0125mm, 99.9%
201. Molybdenum, Foil, 50x50mm, Thickness 10mm, Annealed, 99.9%
202. Molybdenum, Oil Based Standard Solution, Specpure, Mo 5000g/g
203. Molybdenum, Plasma Standard Solution, Specpure?, Mo 10?g/ml
204. Molybdenum, Wire Reel, 10m, Diameter 0.01mm, Hard, 99.8+%
205. Molybdenum, Wire Reel, 1m, Diameter 0.013mm, Hard, 99.8+%
206. Molybdenum, Wire Reel, 1m, Diameter 0.5mm, Annealed, 99.95%
207. Molybdenum, Wire Reel, 1m, Diameter 0.7mm, Annealed, 99.95%
208. Molybdenum, Wire Reel, 1m, Diameter 0.8mm, Annealed, 99.95%
209. Molybdenum, Wire Reel, 1m, Diameter 1.0mm, Annealed, 99.95%
210. Molybdenum, Wire Reel, 20m, Diameter 0.01mm, Hard, 99.8+%
211. Molybdenum, Wire Reel, 50m, Diameter 0.03mm, Hard, 99.95%
212. Molybdenum, Wire Reel, 5m, Diameter 0.013mm, Hard, 99.8+%
213. Molybdenum, Wire Reel, 5m, Diameter 0.025mm, Hard, 99.95%
214. Molybdenum, Wire Reel, 5m, Diameter 0.03mm, Hard, 99.95%
215. Molybdenum, Wire Reel, 5m, Diameter 0.5mm, Annealed, 99.95%
216. Molybdenum, Wire Reel, 5m, Diameter 0.7mm, Annealed, 99.95%
217. Molybdenum, Wire Reel, 5m, Diameter 0.8mm, Annealed, 99.95%
218. Molybdenum, Wire Reel, 5m, Diameter 1.0mm, Annealed, 99.95%
219. Molybdenum, Wire, Diam. 1.0 Mm, 99.95% Trace Metals Basis
220. Molybdenum, Wire, Straight, 1000mm, Diameter 0.5mm, 99.95%
221. Molybdenum, Wire, Straight, 1000mm, Diameter 1.0mm, 99.95%
222. Molybdenum, Wire, Straight, 500mm, Diameter 1.0mm, 99.95%
223. Molybdenum, Wire, Straight, 600mm, Diameter 0.5mm, 99.95%
224. Molybdenum Gauze, 40 Mesh Woven From 0.13mm (0.005in) Dia Wire
225. Molybdenum Gauze, 40 Mesh Woven From 0.13mm (0.005mm) Dia Wire
226. Molybdenum Slug, 6.35mm (0.25in) Dia X 12.7mm (0.50in) Length
227. Molybdenum Slug, 6.35mm (0.25in) Dia X 6.35mm (0.25in) Length
228. Molybdenum, Foil, 0.5m Coil, Thickness 0.015mm, Annealed, 99.9%
229. Molybdenum, Foil, 0.5m Coil, Thickness 0.01mm, Annealed, 99.9%
230. Molybdenum, Foil, 0.5m Coil, Thickness 0.020mm, Annealed, 99.9%
231. Molybdenum, Foil, 0.5m Coil, Thickness 0.025mm, Annealed, 99.9%
232. Molybdenum, Foil, 0.5m Coil, Thickness 0.05mm, Annealed, 99.9%
233. Molybdenum, Foil, 0.5m Coil, Thickness 0.1mm, Annealed, 99.9%
234. Molybdenum, Foil, 100x100mm, Thickness 0.15mm, Annealed, 99.9%
235. Molybdenum, Foil, 100x100mm, Thickness 0.1mm, Annealed, 99.9%
236. Molybdenum, Foil, 100x100mm, Thickness 0.20mm, Annealed, 99.9%
237. Molybdenum, Foil, 100x100mm, Thickness 0.25mm, Annealed, 99.9%
238. Molybdenum, Foil, 100x100mm, Thickness 0.30mm, Annealed, 99.9%
239. Molybdenum, Foil, 100x100mm, Thickness 0.5mm, Annealed, 99.9%
240. Molybdenum, Foil, 100x100mm, Thickness 0.75mm, Annealed, 99.9%
241. Molybdenum, Foil, 100x100mm, Thickness 1.0mm, Annealed, 99.9%
242. Molybdenum, Foil, 100x100mm, Thickness 1.0mm, Annealed, 99.98%
243. Molybdenum, Foil, 100x100mm, Thickness 1.5mm, Annealed, 99.9%
244. Molybdenum, Foil, 100x100mm, Thickness 2.0mm, Annealed, 99.9%
245. Molybdenum, Foil, 100x100mm, Thickness 3.0mm, Annealed, 99.9%
246. Molybdenum, Foil, 10mm Disks, Thickness 0.015mm, Annealed, 99.9%
247. Molybdenum, Foil, 10mm Disks, Thickness 0.01mm, Annealed, 99.9%
248. Molybdenum, Foil, 10mm Disks, Thickness 0.020mm, Annealed, 99.9%
249. Molybdenum, Foil, 10mm Disks, Thickness 0.025mm, Annealed, 99.9%
250. Molybdenum, Foil, 10mm Disks, Thickness 0.03mm, Annealed, 99.9%
251. Molybdenum, Foil, 10mm Disks, Thickness 0.05mm, Annealed, 99.9%
252. Molybdenum, Foil, 10mm Disks, Thickness 0.075mm, Annealed, 99.9%
253. Molybdenum, Foil, 10mm Disks, Thickness 0.125mm, Annealed, 99.9%
254. Molybdenum, Foil, 10mm Disks, Thickness 0.15mm, Annealed, 99.9%
255. Molybdenum, Foil, 10mm Disks, Thickness 0.1mm, Annealed, 99.9%
256. Molybdenum, Foil, 10mm Disks, Thickness 0.20mm, Annealed, 99.9%
257. Molybdenum, Foil, 10mm Disks, Thickness 0.25mm, Annealed, 99.9%
258. Molybdenum, Foil, 10mm Disks, Thickness 0.30mm, Annealed, 99.9%
259. Molybdenum, Foil, 10mm Disks, Thickness 0.5mm, Annealed, 99.9%
260. Molybdenum, Foil, 10mm Disks, Thickness 0.75mm, Annealed, 99.9%
261. Molybdenum, Foil, 150x150mm, Thickness 0.15mm, Annealed, 99.9%
262. Molybdenum, Foil, 150x150mm, Thickness 0.1mm, Annealed, 99.9%
263. Molybdenum, Foil, 150x150mm, Thickness 0.20mm, Annealed, 99.9%
264. Molybdenum, Foil, 150x150mm, Thickness 0.25mm, Annealed, 99.9%
265. Molybdenum, Foil, 150x150mm, Thickness 0.30mm, Annealed, 99.9%
266. Molybdenum, Foil, 150x150mm, Thickness 0.5mm, Annealed, 99.9%
267. Molybdenum, Foil, 150x150mm, Thickness 1.0mm, Annealed, 99.9%
268. Molybdenum, Foil, 150x150mm, Thickness 1.0mm, Annealed, 99.98%
269. Molybdenum, Foil, 150x150mm, Thickness 2.0mm, Annealed, 99.9%
270. Molybdenum, Foil, 15mm Disks, Thickness 0.015mm, Annealed, 99.9%
271. Molybdenum, Foil, 15mm Disks, Thickness 0.01mm, Annealed, 99.9%
272. Molybdenum, Foil, 15mm Disks, Thickness 0.020mm, Annealed, 99.9%
273. Molybdenum, Foil, 15mm Disks, Thickness 0.025mm, Annealed, 99.9%
274. Molybdenum, Foil, 15mm Disks, Thickness 0.03mm, Annealed, 99.9%
275. Molybdenum, Foil, 15mm Disks, Thickness 0.05mm, Annealed, 99.9%
276. Molybdenum, Foil, 15mm Disks, Thickness 0.075mm, Annealed, 99.9%
277. Molybdenum, Foil, 15mm Disks, Thickness 0.125mm, Annealed, 99.9%
278. Molybdenum, Foil, 15mm Disks, Thickness 0.15mm, Annealed, 99.9%
279. Molybdenum, Foil, 15mm Disks, Thickness 0.1mm, Annealed, 99.9%
280. Molybdenum, Foil, 15mm Disks, Thickness 0.20mm, Annealed, 99.9%
281. Molybdenum, Foil, 15mm Disks, Thickness 0.25mm, Annealed, 99.9%
282. Molybdenum, Foil, 15mm Disks, Thickness 0.30mm, Annealed, 99.9%
283. Molybdenum, Foil, 15mm Disks, Thickness 0.5mm, Annealed, 99.9%
284. Molybdenum, Foil, 15mm Disks, Thickness 0.75mm, Annealed, 99.9%
285. Molybdenum, Foil, 1m Coil, Thickness 0.015mm, Annealed, 99.9%
286. Molybdenum, Foil, 1m Coil, Thickness 0.015mm, As Rolled, 99.9%
287. Molybdenum, Foil, 1m Coil, Thickness 0.01mm, Annealed, 99.9%
288. Molybdenum, Foil, 1m Coil, Thickness 0.020mm, Annealed, 99.9%
289. Molybdenum, Foil, 1m Coil, Thickness 0.025mm, Annealed, 99.9%
290. Molybdenum, Foil, 200x200mm, Thickness 0.75mm, Annealed, 99.9%
291. Molybdenum, Foil, 250x300mm, Thickness 0.1mm, Annealed, 99.9%
292. Molybdenum, Foil, 25mm Disks, Thickness 0.015mm, Annealed, 99.9%
293. Molybdenum, Foil, 25mm Disks, Thickness 0.01mm, Annealed, 99.9%
294. Molybdenum, Foil, 25mm Disks, Thickness 0.020mm, Annealed, 99.9%
295. Molybdenum, Foil, 25mm Disks, Thickness 0.025mm, Annealed, 99.9%
296. Molybdenum, Foil, 25mm Disks, Thickness 0.03mm, Annealed, 99.9%
297. Molybdenum, Foil, 25mm Disks, Thickness 0.05mm, Annealed, 99.9%
298. Molybdenum, Foil, 25mm Disks, Thickness 0.075mm, Annealed, 99.9%
299. Molybdenum, Foil, 25mm Disks, Thickness 0.125mm, Annealed, 99.9%
300. Molybdenum, Foil, 25mm Disks, Thickness 0.15mm, Annealed, 99.9%
301. Molybdenum, Foil, 25mm Disks, Thickness 0.1mm, Annealed, 99.9%
302. Molybdenum, Foil, 25mm Disks, Thickness 0.20mm, Annealed, 99.9%
303. Molybdenum, Foil, 25mm Disks, Thickness 0.25mm, Annealed, 99.9%
304. Molybdenum, Foil, 25mm Disks, Thickness 0.30mm, Annealed, 99.9%
305. Molybdenum, Foil, 25mm Disks, Thickness 0.5mm, Annealed, 99.9%
306. Molybdenum, Foil, 25mm Disks, Thickness 0.75mm, Annealed, 99.9%
307. Molybdenum, Foil, 25x25mm, Thickness 0.125mm, Annealed, 99.9%
308. Molybdenum, Foil, 25x25mm, Thickness 0.15mm, Annealed, 99.9%
309. Molybdenum, Foil, 25x25mm, Thickness 0.1mm, Annealed, 99.9%
310. Molybdenum, Foil, 25x25mm, Thickness 0.20mm, Annealed, 99.9%
311. Molybdenum, Foil, 25x25mm, Thickness 0.25mm, Annealed, 99.9%
312. Molybdenum, Foil, 25x25mm, Thickness 0.30mm, Annealed, 99.9%
313. Molybdenum, Foil, 25x25mm, Thickness 0.5mm, Annealed, 99.9%
314. Molybdenum, Foil, 25x25mm, Thickness 0.75mm, Annealed, 99.9%
315. Molybdenum, Foil, 25x25mm, Thickness 1.0mm, Annealed, 99.9%
316. Molybdenum, Foil, 300x300mm, Thickness 0.15mm, Annealed, 99.9%
317. Molybdenum, Foil, 300x300mm, Thickness 0.20mm, Annealed, 99.9%
318. Molybdenum, Foil, 300x300mm, Thickness 0.25mm, Annealed, 99.9%
319. Molybdenum, Foil, 300x300mm, Thickness 0.30mm, Annealed, 99.9%
320. Molybdenum, Foil, 300x300mm, Thickness 0.5mm, Annealed, 99.9%
321. Molybdenum, Foil, 34mm Disks, Thickness 10mm, Annealed, 99.9%
322. Molybdenum, Foil, 4mm Disks, Thickness 0.015mm, Annealed, 99.9%
323. Molybdenum, Foil, 4mm Disks, Thickness 0.015mm, As Rolled, 99.9%
324. Molybdenum, Foil, 4mm Disks, Thickness 0.01mm, Annealed, 99.9%
325. Molybdenum, Foil, 4mm Disks, Thickness 0.020mm, Annealed, 99.9%
326. Molybdenum, Foil, 4mm Disks, Thickness 0.025mm, Annealed, 99.9%
327. Molybdenum, Foil, 4mm Disks, Thickness 0.03mm, Annealed, 99.9%
328. Molybdenum, Foil, 4mm Disks, Thickness 0.05mm, Annealed, 99.9%
329. Molybdenum, Foil, 4mm Disks, Thickness 0.075mm, Annealed, 99.9%
330. Molybdenum, Foil, 4mm Disks, Thickness 0.125mm, Annealed, 99.9%
331. Molybdenum, Foil, 4mm Disks, Thickness 0.15mm, Annealed, 99.9%
332. Molybdenum, Foil, 4mm Disks, Thickness 0.1mm, Annealed, 99.9%
333. Molybdenum, Foil, 4mm Disks, Thickness 0.20mm, Annealed, 99.9%
334. Molybdenum, Foil, 4mm Disks, Thickness 0.25mm, Annealed, 99.9%
335. Molybdenum, Foil, 4mm Disks, Thickness 0.30mm, Annealed, 99.9%
336. Molybdenum, Foil, 4mm Disks, Thickness 0.5mm, Annealed, 99.9%
337. Molybdenum, Foil, 4mm Disks, Thickness 0.75mm, Annealed, 99.9%
338. Molybdenum, Foil, 50mm Disks, Thickness 0.015mm, Annealed, 99.9%
339. Molybdenum, Foil, 50mm Disks, Thickness 0.01mm, Annealed, 99.9%
340. Molybdenum, Foil, 50mm Disks, Thickness 0.020mm, Annealed, 99.9%
341. Molybdenum, Foil, 50mm Disks, Thickness 0.025mm, Annealed, 99.9%
342. Molybdenum, Foil, 50mm Disks, Thickness 0.03mm, Annealed, 99.9%
343. Molybdenum, Foil, 50mm Disks, Thickness 0.05mm, Annealed, 99.9%
344. Molybdenum, Foil, 50mm Disks, Thickness 0.075mm, Annealed, 99.9%
345. Molybdenum, Foil, 50mm Disks, Thickness 0.125mm, Annealed, 99.9%
346. Molybdenum, Foil, 50mm Disks, Thickness 0.15mm, Annealed, 99.9%
347. Molybdenum, Foil, 50mm Disks, Thickness 0.1mm, Annealed, 99.9%
348. Molybdenum, Foil, 50mm Disks, Thickness 0.20mm, Annealed, 99.9%
349. Molybdenum, Foil, 50mm Disks, Thickness 0.25mm, Annealed, 99.9%
350. Molybdenum, Foil, 50mm Disks, Thickness 0.30mm, Annealed, 99.9%
351. Molybdenum, Foil, 50mm Disks, Thickness 0.5mm, Annealed, 99.9%
352. Molybdenum, Foil, 50mm Disks, Thickness 0.75mm, Annealed, 99.9%
353. Molybdenum, Foil, 50x50mm, Thickness 0.125mm, Annealed, 99.9%
354. Molybdenum, Foil, 50x50mm, Thickness 0.15mm, Annealed, 99.9%
355. Molybdenum, Foil, 50x50mm, Thickness 0.1mm, Annealed, 99.9%
356. Molybdenum, Foil, 50x50mm, Thickness 0.20mm, Annealed, 99.9%
357. Molybdenum, Foil, 50x50mm, Thickness 0.25mm, Annealed, 99.9%
358. Molybdenum, Foil, 50x50mm, Thickness 0.30mm, Annealed, 99.9%
359. Molybdenum, Foil, 50x50mm, Thickness 0.5mm, Annealed, 99.9%
360. Molybdenum, Foil, 50x50mm, Thickness 0.75mm, Annealed, 99.9%
361. Molybdenum, Foil, 50x50mm, Thickness 1.0mm, Annealed, 99.9%
362. Molybdenum, Foil, 50x50mm, Thickness 1.0mm, Annealed, 99.98%
363. Molybdenum, Foil, 50x50mm, Thickness 1.5mm, Annealed, 99.9%
364. Molybdenum, Foil, 50x50mm, Thickness 2.0mm, Annealed, 99.9%
365. Molybdenum, Foil, 6mm Disks, Thickness 0.015mm, Annealed, 99.9%
366. Molybdenum, Foil, 6mm Disks, Thickness 0.015mm, As Rolled, 99.9%
367. Molybdenum, Foil, 6mm Disks, Thickness 0.01mm, Annealed, 99.9%
368. Molybdenum, Foil, 6mm Disks, Thickness 0.020mm, Annealed, 99.9%
369. Molybdenum, Foil, 6mm Disks, Thickness 0.025mm, Annealed, 99.9%
370. Molybdenum, Foil, 6mm Disks, Thickness 0.03mm, Annealed, 99.9%
371. Molybdenum, Foil, 6mm Disks, Thickness 0.05mm, Annealed, 99.9%
372. Molybdenum, Foil, 6mm Disks, Thickness 0.075mm, Annealed, 99.9%
373. Molybdenum, Foil, 6mm Disks, Thickness 0.125mm, Annealed, 99.9%
374. Molybdenum, Foil, 6mm Disks, Thickness 0.15mm, Annealed, 99.9%
375. Molybdenum, Foil, 6mm Disks, Thickness 0.1mm, Annealed, 99.9%
376. Molybdenum, Foil, 6mm Disks, Thickness 0.20mm, Annealed, 99.9%
377. Molybdenum, Foil, 6mm Disks, Thickness 0.25mm, Annealed, 99.9%
378. Molybdenum, Foil, 6mm Disks, Thickness 0.30mm, Annealed, 99.9%
379. Molybdenum, Foil, 6mm Disks, Thickness 0.5mm, Annealed, 99.9%
380. Molybdenum, Foil, 6mm Disks, Thickness 0.75mm, Annealed, 99.9%
381. Molybdenum, Foil, 8mm Disks, Thickness 0.015mm, Annealed, 99.9%
382. Molybdenum, Foil, 8mm Disks, Thickness 0.015mm, As Rolled, 99.9%
383. Molybdenum, Foil, 8mm Disks, Thickness 0.01mm, Annealed, 99.9%
384. Molybdenum, Foil, 8mm Disks, Thickness 0.020mm, Annealed, 99.9%
385. Molybdenum, Foil, 8mm Disks, Thickness 0.025mm, Annealed, 99.9%
386. Molybdenum, Foil, 8mm Disks, Thickness 0.03mm, Annealed, 99.9%
387. Molybdenum, Foil, 8mm Disks, Thickness 0.05mm, Annealed, 99.9%
388. Molybdenum, Foil, 8mm Disks, Thickness 0.075mm, Annealed, 99.9%
389. Molybdenum, Foil, 8mm Disks, Thickness 0.125mm, Annealed, 99.9%
390. Molybdenum, Foil, 8mm Disks, Thickness 0.15mm, Annealed, 99.9%
391. Molybdenum, Foil, 8mm Disks, Thickness 0.1mm, Annealed, 99.9%
392. Molybdenum, Foil, 8mm Disks, Thickness 0.20mm, Annealed, 99.9%
393. Molybdenum, Foil, 8mm Disks, Thickness 0.25mm, Annealed, 99.9%
394. Molybdenum, Foil, 8mm Disks, Thickness 0.30mm, Annealed, 99.9%
395. Molybdenum, Foil, 8mm Disks, Thickness 0.5mm, Annealed, 99.9%
396. Molybdenum, Foil, 8mm Disks, Thickness 0.75mm, Annealed, 99.9%
397. Molybdenum, Foil, Thickness 0.025 Mm, >=99.9% Trace Metals Basis
398. Molybdenum, Foil, Thickness 0.05 Mm, >=99.9% Trace Metals Basis
399. Molybdenum, Foil, Thickness 0.1 Mm, >=99.9% Trace Metals Basis
400. Molybdenum, Foil, Thickness 1.0 Mm, >=99.9% Trace Metals Basis
401. Molybdenum, Rod, 1000mm, Diameter 12mm, Centerless Ground, 99.9%
402. Molybdenum, Rod, 100mm, Diameter 1.5mm, Centerless Ground, 99.9%
403. Molybdenum, Rod, 100mm, Diameter 12mm, Centerless Ground, 99.9%
404. Molybdenum, Rod, 100mm, Diameter 2.0mm, Centerless Ground, 99.9%
405. Molybdenum, Rod, 100mm, Diameter 25mm, Centerless Ground, 99.9%
406. Molybdenum, Rod, 100mm, Diameter 3.0mm, Centerless Ground, 99.9%
407. Molybdenum, Rod, 100mm, Diameter 30mm, Centerless Ground, 99.9%
408. Molybdenum, Rod, 100mm, Diameter 4.0mm, Centerless Ground, 99.9%
409. Molybdenum, Rod, 100mm, Diameter 5.0mm, Centerless Ground, 99.9%
410. Molybdenum, Rod, 100mm, Diameter 6.0mm, Centerless Ground, 99.9%
411. Molybdenum, Rod, 100mm, Diameter 8.0mm, Centerless Ground, 99.9%
412. Molybdenum, Rod, 15.0 Mm Diameter, Length 100 Mm, Purity 99.9%
413. Molybdenum, Rod, 200mm, Diameter 1.5mm, Centerless Ground, 99.9%
414. Molybdenum, Rod, 200mm, Diameter 12mm, Centerless Ground, 99.9%
415. Molybdenum, Rod, 200mm, Diameter 2.0mm, Centerless Ground, 99.9%
416. Molybdenum, Rod, 200mm, Diameter 25mm, Centerless Ground, 99.9%
417. Molybdenum, Rod, 200mm, Diameter 3.0mm, Centerless Ground, 99.9%
418. Molybdenum, Rod, 200mm, Diameter 4.0mm, Centerless Ground, 99.9%
419. Molybdenum, Rod, 200mm, Diameter 5.0mm, Centerless Ground, 99.9%
420. Molybdenum, Rod, 200mm, Diameter 6.0mm, Centerless Ground, 99.9%
421. Molybdenum, Rod, 200mm, Diameter 8.0mm, Centerless Ground, 99.9%
422. Molybdenum, Rod, 25mm, Diameter 25mm, Centerless Ground, 99.9%
423. Molybdenum, Rod, 300mm, Diameter 6.0mm, Centerless Ground, 99.9%
424. Molybdenum, Rod, 500mm, Diameter 1.5mm, Centerless Ground, 99.9%
425. Molybdenum, Rod, 500mm, Diameter 12mm, Centerless Ground, 99.9%
426. Molybdenum, Rod, 500mm, Diameter 2.0mm, Centerless Ground, 99.9%
427. Molybdenum, Rod, 500mm, Diameter 3.0mm, Centerless Ground, 99.9%
428. Molybdenum, Rod, 500mm, Diameter 4.0mm, Centerless Ground, 99.9%
429. Molybdenum, Rod, 500mm, Diameter 5.0mm, Centerless Ground, 99.9%
430. Molybdenum, Rod, 500mm, Diameter 6.0mm, Centerless Ground, 99.9%
431. Molybdenum, Rod, 500mm, Diameter 8.0mm, Centerless Ground, 99.9%
432. Molybdenum, Rod, 50mm, Diameter 15.0mm, Centerless Ground, 99.9%
433. Molybdenum, Rod, 50mm, Diameter 20.0mm, Centerless Ground, 99.9%
434. Molybdenum, Rod, 50mm, Diameter 25mm, Centerless Ground, 99.9%
435. Molybdenum, Rod, 50mm, Diameter 30mm, Centerless Ground, 99.9%
436. Molybdenum, Wire Reel, 0.5m, Diameter 1.25mm, Annealed, 99.95%
437. Molybdenum, Wire Reel, 1000m, Diameter 0.025mm, Hard, 99.95%
438. Molybdenum, Wire Reel, 1000m, Diameter 0.03mm, Hard, 99.95%
439. Molybdenum, Wire Reel, 1000m, Diameter 0.05mm, Annealed, 99.95%
440. Molybdenum, Wire Reel, 100m, Diameter 0.013mm, Hard, 99.8+%
441. Molybdenum, Wire Reel, 100m, Diameter 0.025mm, Hard, 99.95%
442. Molybdenum, Wire Reel, 100m, Diameter 0.03mm, Hard, 99.95%
443. Molybdenum, Wire Reel, 100m, Diameter 0.125mm, Annealed, 99.95%
444. Molybdenum, Wire Reel, 100m, Diameter 0.25mm, Annealed, 99.95%
445. Molybdenum, Wire Reel, 100m, Diameter 0.2mm, As Drawn, 99.95%
446. Molybdenum, Wire Reel, 100m, Diameter 0.30mm, Annealed, 99.95%
447. Molybdenum, Wire Reel, 100m, Diameter 0.5mm, Annealed, 99.95%
448. Molybdenum, Wire Reel, 100m, Diameter 0.75mm, Annealed, 99.95%
449. Molybdenum, Wire Reel, 100m, Diameter 0.7mm, Annealed, 99.95%
450. Molybdenum, Wire Reel, 100m, Diameter 0.8mm, Annealed, 99.95%
451. Molybdenum, Wire Reel, 100m, Diameter 1.0mm, Annealed, 99.95%
452. Molybdenum, Wire Reel, 100m, Diameter 1.25mm, Annealed, 99.95%
453. Molybdenum, Wire Reel, 10m, Diameter 0.013mm, Hard, 99.8+%
454. Molybdenum, Wire Reel, 10m, Diameter 0.05mm, Annealed, 99.95%
455. Molybdenum, Wire Reel, 10m, Diameter 0.25mm, Annealed, 99.95%
456. Molybdenum, Wire Reel, 10m, Diameter 0.2mm, As Drawn, 99.95%
457. Molybdenum, Wire Reel, 10m, Diameter 0.30mm, Annealed, 99.95%
458. Molybdenum, Wire Reel, 10m, Diameter 0.5mm, Annealed, 99.95%
459. Molybdenum, Wire Reel, 10m, Diameter 0.75mm, Annealed, 99.95%
460. Molybdenum, Wire Reel, 10m, Diameter 0.7mm, Annealed, 99.95%
461. Molybdenum, Wire Reel, 10m, Diameter 0.8mm, Annealed, 99.95%
462. Molybdenum, Wire Reel, 10m, Diameter 1.0mm, Annealed, 99.95%
463. Molybdenum, Wire Reel, 1m, Diameter 0.75mm, Annealed, 99.95%
464. Molybdenum, Wire Reel, 1m, Diameter 1.25mm, Annealed, 99.95%
465. Molybdenum, Wire Reel, 2000m, Diameter 0.025mm, Hard, 99.95%
466. Molybdenum, Wire Reel, 2000m, Diameter 0.03mm, Hard, 99.95%
467. Molybdenum, Wire Reel, 2000m, Diameter 0.05mm, Annealed, 99.95%
468. Molybdenum, Wire Reel, 200m, Diameter 0.25mm, Annealed, 99.95%
469. Molybdenum, Wire Reel, 200m, Diameter 0.2mm, As Drawn, 99.95%
470. Molybdenum, Wire Reel, 200m, Diameter 0.30mm, Annealed, 99.95%
471. Molybdenum, Wire Reel, 200m, Diameter 0.5mm, Annealed, 99.95%
472. Molybdenum, Wire Reel, 20m, Diameter 0.013mm, Hard, 99.8+%
473. Molybdenum, Wire Reel, 250m, Diameter 0.05mm, Annealed, 99.95%
474. Molybdenum, Wire Reel, 25m, Diameter 0.125mm, Annealed, 99.95%
475. Molybdenum, Wire Reel, 25m, Diameter 1.0mm, Annealed, 99.95%
476. Molybdenum, Wire Reel, 25m, Diameter 1.25mm, Annealed, 99.95%
477. Molybdenum, Wire Reel, 2m, Diameter 0.25mm, Annealed, 99.95%
478. Molybdenum, Wire Reel, 2m, Diameter 0.2mm, As Drawn, 99.95%
479. Molybdenum, Wire Reel, 2m, Diameter 0.30mm, Annealed, 99.95%
480. Molybdenum, Wire Reel, 500m, Diameter 0.025mm, Hard, 99.95%
481. Molybdenum, Wire Reel, 500m, Diameter 0.03mm, Hard, 99.95%
482. Molybdenum, Wire Reel, 500m, Diameter 0.05mm, Annealed, 99.95%
483. Molybdenum, Wire Reel, 500m, Diameter 0.125mm, Annealed, 99.95%
484. Molybdenum, Wire Reel, 50m, Diameter 0.013mm, Hard, 99.8+%
485. Molybdenum, Wire Reel, 50m, Diameter 0.025mm, Hard, 99.95%
486. Molybdenum, Wire Reel, 50m, Diameter 0.035mm, As Drawn, 99.95%
487. Molybdenum, Wire Reel, 50m, Diameter 0.05mm, Annealed, 99.95%
488. Molybdenum, Wire Reel, 50m, Diameter 0.125mm, Annealed, 99.95%
489. Molybdenum, Wire Reel, 50m, Diameter 0.25mm, Annealed, 99.95%
490. Molybdenum, Wire Reel, 50m, Diameter 0.2mm, As Drawn, 99.95%
491. Molybdenum, Wire Reel, 50m, Diameter 0.30mm, Annealed, 99.95%
492. Molybdenum, Wire Reel, 50m, Diameter 0.5mm, Annealed, 99.95%
493. Molybdenum, Wire Reel, 50m, Diameter 0.75mm, Annealed, 99.95%
494. Molybdenum, Wire Reel, 50m, Diameter 0.7mm, Annealed, 99.95%
495. Molybdenum, Wire Reel, 50m, Diameter 0.8mm, Annealed, 99.95%
496. Molybdenum, Wire Reel, 50m, Diameter 1.0mm, Annealed, 99.95%
497. Molybdenum, Wire Reel, 50m, Diameter 1.25mm, Annealed, 99.95%
498. Molybdenum, Wire Reel, 5m, Diameter 0.035mm, As Drawn, 99.95%
499. Molybdenum, Wire Reel, 5m, Diameter 0.05mm, Gold Plated, 99.95%
500. Molybdenum, Wire Reel, 5m, Diameter 0.125mm, Annealed, 99.95%
501. Molybdenum, Wire Reel, 5m, Diameter 0.75mm, Annealed, 99.95%
502. Molybdenum, Wire Reel, 5m, Diameter 1.25mm, Annealed, 99.95%
503. Molybdenum, Wire, 0.075 Mm Diameter, Length 10 M, Purity 99.95%
504. Molybdenum, Wire, 0.075 Mm Diameter, Length 50 M, Purity 99.95%
505. Molybdenum, Wire, 0.635 Mm Diameter, Length 1 M, Purity 99.95%
506. Molybdenum, Wire, Straight, 1000mm, Diameter 0.25mm, 99.95%
507. Molybdenum Gauze, 35 Mesh Woven From 0.0509mm (0.002in) Dia Wire
508. Molybdenum Slug, 3.175mm (0.125in) Dia X 3.175mm (0.125in) Length
509. Molybdenum Slug, 3.175mm (0.125in) Dia X 6.35mm (0.25in) Length
510. Molybdenum, Bolt, Size M3 X 30 Mm, Length 10 Pcs, Head Style Hexagonal
511. Molybdenum, Bolt, Size M4 X 30 Mm, Length 25 Pcs, Head Style Hexagonal
512. Molybdenum, Bolt, Size M4 X 30 Mm, Length 50 Pcs, Head Style Hexagonal
513. Molybdenum, Bolt, Size M5 X 30 Mm, Length 25 Pcs, Head Style Hexagonal
514. Molybdenum, Bolt, Size M5 X 30 Mm, Length 50 Pcs, Head Style Hexagonal
515. Molybdenum, Bolt, Size M6 X 30 Mm, Length 25 Pcs, Head Style Hexagonal
516. Molybdenum, Bolt, Size M6 X 30 Mm, Length 50 Pcs, Head Style Hexagonal
517. Molybdenum, Crucible, Cylindrical, Length 1 Pcs, Outside Diameter 33mm
518. Molybdenum, Crucible, Cylindrical, Length 1 Pcs, Outside Diameter 59mm
519. Molybdenum, Crucible, Cylindrical, Outside Diameter 47mm, Length 1 Pcs
520. Molybdenum, Foil, 0.5m Coil, Thickness 0.015mm, As Rolled, 99.9%
521. Molybdenum, Foil, 100x100mm, Thickness 0.125mm, Annealed, 99.9%
522. Molybdenum, Foil, 10mm Disks, Thickness 0.015mm, As Rolled, 99.9%
523. Molybdenum, Foil, 150x150mm, Thickness 0.125mm, Annealed, 99.9%
524. Molybdenum, Foil, 15mm Disks, Thickness 0.015mm, As Rolled, 99.9%
525. Molybdenum, Foil, 25mm Disks, Thickness 0.015mm, As Rolled, 99.9%
526. Molybdenum, Foil, 300x300mm, Thickness 0.125mm, Annealed, 99.9%
527. Molybdenum, Foil, 50mm Disks, Thickness 0.015mm, As Rolled, 99.9%
528. Molybdenum, Foil, Light Tested, 100x100mm, Thickness 0.008mm, 99.9%
529. Molybdenum, Foil, Light Tested, 100x100mm, Thickness 0.0125mm, 99.9%
530. Molybdenum, Foil, Light Tested, 100x100mm, Thickness 0.01mm, 99.9%
531. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.004mm, 99.9%
532. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.005mm, 99.9%
533. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.007mm, 99.9%
534. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.008mm, 99.9%
535. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.009mm, 99.9%
536. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.0125mm, 99.9%
537. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.01mm, 99.9%
538. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.006mm, 99.9%
539. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.007mm, 99.9%
540. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.008mm, 99.9%
541. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.009mm, 99.9%
542. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.0125mm, 99.9%
543. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.01mm, 99.9%
544. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.004mm, 99.9%
545. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.005mm, 99.9%
546. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.006mm, 99.9%
547. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.007mm, 99.9%
548. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.008mm, 99.9%
549. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.009mm, 99.9%
550. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.01mm, 99.9%
551. Molybdenum, Foil, Not Light Tested, 150x150mm, Thickness 0.008mm, 99.9%
552. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.001mm, 99.9%
553. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.0025mm, 99.9%
554. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.002mm, 99.9%
555. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.003mm, 99.9%
556. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.004mm, 99.9%
557. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.005mm, 99.9%
558. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.006mm, 99.9%
559. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.008mm, 99.9%
560. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.0125mm, 99.9%
561. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.01mm, 99.9%
562. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.001mm, 99.9%
563. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.0025mm, 99.9%
564. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.002mm, 99.9%
565. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.003mm, 99.9%
566. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.004mm, 99.9%
567. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.005mm, 99.9%
568. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.006mm, 99.9%
569. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.007mm, 99.9%
570. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.008mm, 99.9%
571. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.009mm, 99.9%
572. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.0125mm, 99.9%
573. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.01mm, 99.9%
574. Molybdenum, Foil, Thickness 0.01 Mm, Size 150 X 150 Mm, Purity 99.9%
575. Molybdenum, Foil, Thickness 0.015 Mm, Size 100 X 100 Mm, Purity 99.9%
576. Molybdenum, Foil, Thickness 0.015 Mm, Size 25 X 25 Mm, Purity 99.9%
577. Molybdenum, Foil, Thickness 0.015 Mm, Size 50 X 50 Mm, Purity 99.9%
578. Molybdenum, Foil, Thickness 0.020 Mm, Size 100 X 100 Mm, Purity 99.9%
579. Molybdenum, Foil, Thickness 0.020 Mm, Size 25 X 25 Mm, Purity 99.9%
580. Molybdenum, Foil, Thickness 0.020 Mm, Size 50 X 50 Mm, Purity 99.9%
581. Molybdenum, Foil, Thickness 0.025 Mm, Purity 99.9%, Size 25 X 25 Mm
582. Molybdenum, Foil, Thickness 0.025 Mm, Size 100 X 100 Mm, Purity 99.9%
583. Molybdenum, Foil, Thickness 0.025 Mm, Size 50 X 50 Mm, Purity 99.9%
584. Molybdenum, Foil, Thickness 0.03 Mm, Size 100 X 100 Mm, Purity 99.9%
585. Molybdenum, Foil, Thickness 0.03 Mm, Size 25 X 25 Mm, Purity 99.9%
586. Molybdenum, Foil, Thickness 0.03 Mm, Size 50 X 50 Mm, Purity 99.9%
587. Molybdenum, Foil, Thickness 2.0 Mm, Size 20 X 20 Mm, Purity 99.9%
588. Molybdenum, Rod, 1000mm, Diameter 1.25mm, Centerless Ground, 99.9%
589. Molybdenum, Rod, 1000mm, Diameter 1.5mm, Centerless Ground, 99.9%
590. Molybdenum, Rod, 1000mm, Diameter 10.0mm, Centerless Ground, 99.9%
591. Molybdenum, Rod, 1000mm, Diameter 2.0mm, Centerless Ground, 99.9%
592. Molybdenum, Rod, 1000mm, Diameter 3.0mm, Centerless Ground, 99.9%
593. Molybdenum, Rod, 1000mm, Diameter 4.0mm, Centerless Ground, 99.9%
594. Molybdenum, Rod, 1000mm, Diameter 5.0mm, Centerless Ground, 99.9%
595. Molybdenum, Rod, 1000mm, Diameter 6.0mm, Centerless Ground, 99.9%
596. Molybdenum, Rod, 1000mm, Diameter 8.0mm, Centerless Ground, 99.9%
597. Molybdenum, Rod, 100mm, Diameter 1.25mm, Centerless Ground, 99.9%
598. Molybdenum, Rod, 100mm, Diameter 10.0mm, Centerless Ground, 99.9%
599. Molybdenum, Rod, 100mm, Diameter 15.0mm, Centerless Ground, 99.9%
600. Molybdenum, Rod, 100mm, Diameter 20.0mm, Centerless Ground, 99.9%
601. Molybdenum, Rod, 200mm, Diameter 1.25mm, Centerless Ground, 99.9%
602. Molybdenum, Rod, 200mm, Diameter 10.0mm, Centerless Ground, 99.9%
603. Molybdenum, Rod, 200mm, Diameter 15.0mm, Centerless Ground, 99.9%
604. Molybdenum, Rod, 200mm, Diameter 20.0mm, Centerless Ground, 99.9%
605. Molybdenum, Rod, 500mm, Diameter 1.25mm, Centerless Ground, 99.9%
606. Molybdenum, Rod, 500mm, Diameter 10.0mm, Centerless Ground, 99.9%
607. Molybdenum, Rod, 500mm, Diameter 15.0mm, Centerless Ground, 99.9%
608. Molybdenum, Wire Reel, 1000m, Diameter 0.035mm, As Drawn, 99.95%
609. Molybdenum, Wire Reel, 1000m, Diameter 0.125mm, Annealed, 99.95%
610. Molybdenum, Wire Reel, 100m, Diameter 0.025mm, Gold Plated, 99.95%
611. Molybdenum, Wire Reel, 100m, Diameter 0.035mm, As Drawn, 99.95%
612. Molybdenum, Wire Reel, 100m, Diameter 0.05mm, Gold Plated, 99.95%
613. Molybdenum, Wire Reel, 10m, Diameter 0.015mm, Gold Plated, 99.95%
614. Molybdenum, Wire Reel, 10m, Diameter 0.025mm, Gold Plated, 99.95%
615. Molybdenum, Wire Reel, 10m, Diameter 0.05mm, Gold Plated, 99.95%
616. Molybdenum, Wire Reel, 2000m, Diameter 0.035mm, As Drawn, 99.95%
617. Molybdenum, Wire Reel, 200m, Diameter 0.025mm, Gold Plated, 99.95%
618. Molybdenum, Wire Reel, 200m, Diameter 0.05mm, Gold Plated, 99.95%
619. Molybdenum, Wire Reel, 20m, Diameter 0.015mm, Gold Plated, 99.95%
620. Molybdenum, Wire Reel, 20m, Diameter 0.025mm, Gold Plated, 99.95%
621. Molybdenum, Wire Reel, 20m, Diameter 0.05mm, Gold Plated, 99.95%
622. Molybdenum, Wire Reel, 500m, Diameter 0.035mm, As Drawn, 99.95%
623. Molybdenum, Wire Reel, 50m, Diameter 0.015mm, Gold Plated, 99.95%
624. Molybdenum, Wire Reel, 50m, Diameter 0.025mm, Gold Plated, 99.95%
625. Molybdenum, Wire Reel, 50m, Diameter 0.05mm, Gold Plated, 99.95%
626. Molybdenum, Wire Reel, 5m, Diameter 0.015mm, Gold Plated, 99.95%
627. Molybdenum, Wire Reel, 5m, Diameter 0.025mm, Gold Plated, 99.95%
628. Molybdenum, Wire, 0.075 Mm Diameter, Length 1000 M, Purity 99.95%
629. Molybdenum, Wire, 0.075 Mm Diameter, Length 2000 M, Purity 99.95%
630. Molybdenum, Wire, 0.075 Mm Diameter, Length 250 M, Purity 99.95%
631. Molybdenum, Wire, 0.075 Mm Diameter, Length 500 M, Purity 99.95%
632. Molybdenum Slug, 6.35mm (0.25in) Dia X 12.7mm (0.50in) Length, 99.95% (metals Basis)
633. Molybdenum Slug, 6.35mm (0.25in) Dia X 6.35mm (0.25in) Length, 99.95% (metals Basis)
634. Molybdenum Sputtering Target, 50.8mm (2.0in) Dia X 3.18mm (0.125in) Thick
635. Molybdenum Sputtering Target, 50.8mm (2.0in) Dia X 6.35mm (0.250in) Thick
636. Molybdenum Sputtering Target, 76.2mm (3.0in) Dia X 3.18mm (0.125in) Thick
637. Molybdenum Sputtering Target, 76.2mm (3.0in) Dia X 6.35mm (0.250in) Thick
638. Molybdenum Wire, Gold Plated, 0.025mm (0.001in) Dia, 99.95% (metals Basis)
639. Molybdenum, Foil, 0.5m Coil, Thickness 0.04mm, Coil Width 0.7mm, Annealed, 99.9%
640. Molybdenum, Foil, 100x100mm, Thickness 0.25mm, Annealed, Polished On Both Sides, 99.9%
641. Molybdenum, Foil, 100x100mm, Thickness 0.5mm, Polished On Both Sides, 99.9%
642. Molybdenum, Foil, 10m Coil, Thickness 0.04mm, Coil Width 0.7mm, Annealed, 99.9%
643. Molybdenum, Foil, 1m Coil, Thickness 0.04mm, Coil Width 0.7mm, Annealed, 99.9%
644. Molybdenum, Foil, 20m Coil, Thickness 0.04mm, Coil Width 0.7mm, Annealed, 99.9%
645. Molybdenum, Foil, 2m Coil, Thickness 0.04mm, Coil Width 0.7mm, Annealed, 99.9%
646. Molybdenum, Foil, 50x50mm, Thickness 0.25mm, Annealed, Polished On Both Sides, 99.9%
647. Molybdenum, Foil, 50x50mm, Thickness 0.5mm, Polished On Both Sides, 99.9%
648. Molybdenum, Foil, 5m Coil, Thickness 0.04mm, Coil Width 0.7mm, Annealed, 99.9%
649. Molybdenum, Foil, Light Tested, 100x100mm, Thickness 0.01mm, Annealed, 99.9%
650. Molybdenum, Foil, Light Tested, 100x100mm, Thickness 0.025mm, Annealed, 99.9%
651. Molybdenum, Foil, Light Tested, 100x100mm, Thickness 0.05mm, Annealed, 99.9%
652. Molybdenum, Foil, Light Tested, 100x100mm, Thickness 0.075mm, Annealed, 99.9%
653. Molybdenum, Foil, Light Tested, 130x130mm, Thickness 0.015mm, As Rolled, 99.9%
654. Molybdenum, Foil, Light Tested, 150x150mm, Thickness 0.05mm, Annealed, 99.9%
655. Molybdenum, Foil, Light Tested, 150x150mm, Thickness 0.075mm, Annealed, 99.9%
656. Molybdenum, Foil, Light Tested, 200x200mm, Thickness 0.025mm, Annealed, 99.9%
657. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.01mm, Annealed, 99.9%
658. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.020mm, Annealed, 99.9%
659. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.05mm, Annealed, 99.9%
660. Molybdenum, Foil, Light Tested, 25x25mm, Thickness 0.075mm, Annealed, 99.9%
661. Molybdenum, Foil, Light Tested, 29x100mm, Thickness 0.0712mm, Annealed, 99.9%
662. Molybdenum, Foil, Light Tested, 29x200mm, Thickness 0.0712mm, Annealed, 99.9%
663. Molybdenum, Foil, Light Tested, 29x500mm, Thickness 0.0712mm, Annealed, 99.9%
664. Molybdenum, Foil, Light Tested, 29x600mm, Thickness 0.0712mm, Annealed, 99.9%
665. Molybdenum, Foil, Light Tested, 300x300mm, Thickness 0.05mm, Annealed, 99.9%
666. Molybdenum, Foil, Light Tested, 300x300mm, Thickness 0.075mm, Annealed, 99.9%
667. Molybdenum, Foil, Light Tested, 30x30mm, Thickness 0.015mm, As Rolled, 99.9%
668. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.01mm, Annealed, 99.9%
669. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.020mm, Annealed, 99.9%
670. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.05mm, Annealed, 99.9%
671. Molybdenum, Foil, Light Tested, 50x50mm, Thickness 0.075mm, Annealed, 99.9%
672. Molybdenum, Foil, Light Tested, 60x60mm, Thickness 0.015mm, As Rolled, 99.9%
673. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.0125mm, 99.9%
674. Molybdenum, Foil, Not Light Tested, 100x100mm, Thickness 0.01mm, Annealed, 99.9%
675. Molybdenum, Foil, Not Light Tested, 150x150mm, Thickness 0.0125mm, 99.9%
676. Molybdenum, Foil, Not Light Tested, 150x150mm, Thickness 0.05mm, Annealed, 99.9%
677. Molybdenum, Foil, Not Light Tested, 250x300mm, Thickness 0.01mm, Annealed, 99.9%
678. Molybdenum, Foil, Not Light Tested, 25x25mm, Thickness 0.01mm, Annealed, 99.9%
679. Molybdenum, Foil, Not Light Tested, 300x300mm, Thickness 0.05mm, Annealed, 99.9%
680. Molybdenum, Foil, Not Light Tested, 50x50mm, Thickness 0.01mm, Annealed, 99.9%
681. Molybdenum, Foil, Thickness 0.0125 Mm, Size 267 X 300 Mm, Purity 99.9%
682. Molybdenum, Nanopowder, <100 Nm Particle Size (tem), 99.8% Trace Metals Basis
683. Molybdenum, Plasma Standard Solution, Specpure?, Mo 10,000?g/ml, (nh4)2moo4 In 1% Nh4oh
684. Molybdenum, Plasma Standard Solution, Specpure?, Mo 10,000?g/ml, Mo In 5% Hno3/tr. Hf
685. Molybdenum, Plasma Standard Solution, Specpure?, Mo 1000?g/ml, (nh4)2moo4 In 1% Nh4oh
686. Molybdenum, Plasma Standard Solution, Specpure?, Mo 1000?g/ml, Mo In 5% Hno3/tr. Hf
687. Molybdenum, Powder, 350 Max. Part. Size (micron), Weight 1000 G, Purity 99.9+%
688. Molybdenum, Powder, 350 Max. Part. Size (micron), Weight 200 G, Purity 99.9+%
689. Molybdenum, Powder, 350 Max. Part. Size (micron), Weight 50 G, Purity 99.9+%
690. Molybdenum, Powder, 350 Max. Part. Size (micron), Weight 500 G, Purity 99.9+%
691. Molybdenum, Powder, Max. Particle Size 350 Micron, Weight 100 G, Purity 99.9+%
692. Molybdenum, Powder, Mean Particle Size (fsss) <5 Micron, Weight 100 G, Purity 99.9+%
693. Molybdenum, Powder, Mean Particle Size (fsss) <5 Micron, Weight 1000 G, Purity 99.9+%
694. Molybdenum, Powder, Mean Particle Size (fsss) <5 Micron, Weight 200 G, Purity 99.9+%
695. Molybdenum, Powder, Mean Particle Size (fsss) <5 Micron, Weight 50 G, Purity 99.9+%
696. Molybdenum, Powder, Mean Particle Size (fsss) <5 Micron, Weight 500 G, Purity 99.9+%
697. Molybdenum, Tube, Outside Diameter 2.0 Mm, Length 100 Mm, Wall Thickness 0.4 Mm
698. Molybdenum, Tube, Outside Diameter 2.0 Mm, Length 347 Mm, Wall Thickness 0.4 Mm
699. Molybdenum, Tube, Outside Diameter 5.0 Mm, Length 50 Mm, Wall Thickness 0.5 Mm
700. Molybdenum, Washer, Size M3, Length 10 Pcs, Outside Diameter 6.64 - 7.00mm
701. Molybdenum, Washer, Size M4, Length 25 Pcs, Outside Diameter 8.64 - 9.00mm
702. Molybdenum, Washer, Size M4, Length 50 Pcs, Outside Diameter 8.64 - 9.00mm
703. Molybdenum, Washer, Size M5, Length 25 Pcs, Outside Diameter 9.64 - 10.00mm
704. Molybdenum, Washer, Size M5, Length 50 Pcs, Outside Diameter 9.64 - 10.00mm
705. Molybdenum, Washer, Size M6, Length 25 Pcs, Outside Diameter 11.57 - 12.00mm
706. Molybdenum, Washer, Size M6, Length 50 Pcs, Outside Diameter 11.57 - 12.00mm
707. 16727-12-1
708. Mo5
709. Molybdenum Atomic Spectroscopy Standard Concentrate 1.00 G Mo, 1.00 G/l, For 1l Standard Solution, Analytical Standard
710. Molybdenum Single Crystal Disc, 10mm (0.39in) Dia, 1-3mm (0.04-0.1in) Thick, (100) Orientation
711. Molybdenum Single Crystal Disc, 10mm (0.39in) Dia, 1-3mm (0.04-0.1in) Thick, (110) Orientation
712. Molybdenum Single Crystal Disc, 10mm (0.39in) Dia, 1-3mm (0.04-0.1in) Thick, (111) Orientation
713. Molybdenum Single Crystal, 11-12mm (0.43-0.47in) Dia, 100mm (3.9in) Long, (100) Orientation
714. Molybdenum Single Crystal, 11-12mm (0.43-0.47in) Dia, 100mm (3.9in) Long, (110) Orientation, Zone Refined
715. Molybdenum Single Crystal, 11-12mm (0.43-0.47in) Dia, 100mm (3.9in) Long, (111) Orientation, Zone Refined
716. Molybdenum Single Crystal, 11-12mm (0.43-0.47in) Dia, 100mm (3.9in) Long, Random Orientation, Zone Refined
717. Molybdenum Slug, 3.175mm (0.125in) Dia X 3.175mm (0.125in) Length, 99.95% (metals Basis)
718. Molybdenum Slug, 3.175mm (0.125in) Dia X 6.35mm (0.25in) Length, 99.95% (metals Basis)
719. Molybdenum Sputtering Target, 50.8mm (2.0in) Dia X 3.18mm (0.125in) Thick, 99.95% (metals Basis)
720. Molybdenum Sputtering Target, 50.8mm (2.0in) Dia X 6.35mm (0.250in) Thick, 99.95% (metals Basis)
721. Molybdenum Sputtering Target, 76.2mm (3.0in) Dia X 3.18mm (0.125in) Thick, 99.95% (metals Basis)
722. Molybdenum Sputtering Target, 76.2mm (3.0in) Dia X 6.35mm (0.250in) Thick, 99.95% (metals Basis)
723. Molybdenum, Insulated Wire, 10m, Conductor Diameter 0.125mm, Insulation Thickness 0.012mm, Polyimide Insulation, 99.95%
724. Molybdenum, Insulated Wire, 20m, Conductor Diameter 0.125mm, Insulation Thickness 0.012mm, Polyimide Insulation, 99.95%
725. Molybdenum, Insulated Wire, 50m, Conductor Diameter 0.125mm, Insulation Thickness 0.012mm, Polyimide Insulation, 99.95%
726. Molybdenum, Insulated Wire, 5m, Conductor Diameter 0.125mm, Insulation Thickness 0.012mm, Polyimide Insulation, 99.95%
727. Molybdenum, Mesh, 100x100mm, Nominal Aperture 0.44mm, Wire Diameter 0.07mm, 50 Wires/inch, Open Area 67%, Plain Weave Mesh
728. Molybdenum, Mesh, 100x100mm, Nominal Aperture 0.8mm, Wire Diameter 0.18mm, 26 Wires/inch, Open Area 67%, Plain Weave Mesh
729. Molybdenum, Mesh, 150x150mm, Nominal Aperture 0.44mm, Wire Diameter 0.07mm, 50 Wires/inch, Open Area 67%, Plain Weave Mesh
730. Molybdenum, Mesh, 150x150mm, Nominal Aperture 0.8mm, Wire Diameter 0.18mm, 26 Wires/inch, Open Area 67%, Plain Weave Mesh
731. Molybdenum, Mesh, 50x50mm, Nominal Aperture 0.44mm, Wire Diameter 0.07mm, 50 Wires/inch, Open Area 67%, Plain Weave Mesh
732. Molybdenum, Mesh, 50x50mm, Nominal Aperture 0.8mm, Wire Diameter 0.18mm, 26 Wires/inch, Open Area 67%, Plain Weave Mesh
733. Molybdenum, Microfoil, Disks, 10mm, Thinness 0.1mum, Specific Density 92.4mug/cm2, Permanent Mylar 3.5mum Support, 99.9%
734. Molybdenum, Microfoil, Disks, 10mm, Thinness 1.0mum, Specific Density 1022mug/cm2, Permanent Mylar 3.5mum Support, 99.9%
735. Molybdenum, Microfoil, Disks, 25mm, Thinness 0.1mum, Specific Density 92.4mug/cm2, Permanent Mylar 3.5mum Support, 99.9%
736. Molybdenum, Microfoil, Disks, 25mm, Thinness 1.0mum, Specific Density 1022mug/cm2, Permanent Mylar 3.5mum Support, 99.9%
737. Molybdenum, Microfoil, Disks, 50mm, Thinness 1.0mum, Specific Density 1022mug/cm2, Permanent Mylar 3.5mum Support, 99.9%
738. Molybdenum, Tube, 1000mm, Outside Diameter 1.0mm, Inside Diameter 0.7mm, Wall Thickness 0.15mm, 99.9%
739. Molybdenum, Tube, 1000mm, Outside Diameter 1.5mm, Inside Diameter 0.95mm, Wall Thickness 0.275mm, 99.9%
740. Molybdenum, Tube, 1000mm, Outside Diameter 2.0mm, Inside Diameter 1.3mm, Wall Thickness 0.35mm, 99.9%
741. Molybdenum, Tube, 1000mm, Outside Diameter 2.0mm, Inside Diameter 1.4mm, Wall Thickness 0.3mm, 99.9%
742. Molybdenum, Tube, 1000mm, Outside Diameter 3.15mm, Inside Diameter 2.39mm, Wall Thickness 0.38mm, 99.9%
743. Molybdenum, Tube, 1000mm, Outside Diameter 6.0mm, Inside Diameter 5.6mm, Wall Thickness 0.20mm, 99.9%
744. Molybdenum, Tube, 100mm, Outside Diameter 0.51mm, Inside Diameter 0.37mm, Wall Thickness 0.07mm, 99.9%
745. Molybdenum, Tube, 100mm, Outside Diameter 0.5mm, Inside Diameter 0.38mm, Wall Thickness 0.06mm, 99.9%
746. Molybdenum, Tube, 100mm, Outside Diameter 1.0mm, Inside Diameter 0.7mm, Wall Thickness 0.15mm, 99.9%
747. Molybdenum, Tube, 100mm, Outside Diameter 1.5mm, Inside Diameter 0.95mm, Wall Thickness 0.275mm, 99.9%
748. Molybdenum, Tube, 100mm, Outside Diameter 2.0mm, Inside Diameter 1.3mm, Wall Thickness 0.35mm, 99.9%
749. Molybdenum, Tube, 100mm, Outside Diameter 2.0mm, Inside Diameter 1.4mm, Wall Thickness 0.3mm, 99.9%
750. Molybdenum, Tube, 100mm, Outside Diameter 3.15mm, Inside Diameter 2.39mm, Wall Thickness 0.38mm, 99.9%
751. Molybdenum, Tube, 100mm, Outside Diameter 5.0mm, Inside Diameter 4mm, Wall Thickness 0.5mm, 99.9%
752. Molybdenum, Tube, 100mm, Outside Diameter 6.0mm, Inside Diameter 4.4mm, Wall Thickness 0.8mm, 99.9%
753. Molybdenum, Tube, 100mm, Outside Diameter 6.0mm, Inside Diameter 5.6mm, Wall Thickness 0.20mm, 99.9%
754. Molybdenum, Tube, 100mm, Outside Diameter 6.4mm, Inside Diameter 4.6mm, Wall Thickness 0.9mm, 99.9%
755. Molybdenum, Tube, 200mm, Outside Diameter 0.51mm, Inside Diameter 0.37mm, Wall Thickness 0.07mm, 99.9%
756. Molybdenum, Tube, 200mm, Outside Diameter 0.5mm, Inside Diameter 0.38mm, Wall Thickness 0.06mm, 99.9%
757. Molybdenum, Tube, 200mm, Outside Diameter 1.0mm, Inside Diameter 0.7mm, Wall Thickness 0.15mm, 99.9%
758. Molybdenum, Tube, 200mm, Outside Diameter 1.5mm, Inside Diameter 0.95mm, Wall Thickness 0.275mm, 99.9%
759. Molybdenum, Tube, 200mm, Outside Diameter 2.0mm, Inside Diameter 1.3mm, Wall Thickness 0.35mm, 99.9%
760. Molybdenum, Tube, 200mm, Outside Diameter 2.0mm, Inside Diameter 1.4mm, Wall Thickness 0.3mm, 99.9%
761. Molybdenum, Tube, 200mm, Outside Diameter 3.15mm, Inside Diameter 2.39mm, Wall Thickness 0.38mm, 99.9%
762. Molybdenum, Tube, 200mm, Outside Diameter 5.0mm, Inside Diameter 4mm, Wall Thickness 0.5mm, 99.9%
763. Molybdenum, Tube, 200mm, Outside Diameter 6.0mm, Inside Diameter 4.4mm, Wall Thickness 0.8mm, 99.9%
764. Molybdenum, Tube, 200mm, Outside Diameter 6.0mm, Inside Diameter 5.6mm, Wall Thickness 0.20mm, 99.9%
765. Molybdenum, Tube, 200mm, Outside Diameter 6.4mm, Inside Diameter 4.6mm, Wall Thickness 0.9mm, 99.9%
766. Molybdenum, Tube, 300mm, Outside Diameter 0.5mm, Inside Diameter 0.38mm, Wall Thickness 0.06mm, 99.9%
767. Molybdenum, Tube, 500mm, Outside Diameter 1.0mm, Inside Diameter 0.7mm, Wall Thickness 0.15mm, 99.9%
768. Molybdenum, Tube, 500mm, Outside Diameter 1.5mm, Inside Diameter 0.95mm, Wall Thickness 0.275mm, 99.9%
769. Molybdenum, Tube, 500mm, Outside Diameter 2.0mm, Inside Diameter 1.3mm, Wall Thickness 0.35mm, 99.9%
770. Molybdenum, Tube, 500mm, Outside Diameter 2.0mm, Inside Diameter 1.4mm, Wall Thickness 0.3mm, 99.9%
771. Molybdenum, Tube, 500mm, Outside Diameter 3.15mm, Inside Diameter 2.39mm, Wall Thickness 0.38mm, 99.9%
772. Molybdenum, Tube, 500mm, Outside Diameter 5.0mm, Inside Diameter 4mm, Wall Thickness 0.5mm, 99.9%
773. Molybdenum, Tube, 500mm, Outside Diameter 6.0mm, Inside Diameter 4.4mm, Wall Thickness 0.8mm, 99.9%
774. Molybdenum, Tube, 500mm, Outside Diameter 6.0mm, Inside Diameter 5.6mm, Wall Thickness 0.20mm, 99.9%
775. Molybdenum, Tube, 500mm, Outside Diameter 6.4mm, Inside Diameter 4.6mm, Wall Thickness 0.9mm, 99.9%
776. Molybdenum, Tube, 50mm, Outside Diameter 3.15mm, Inside Diameter 2.39mm, Wall Thickness 0.38mm, 99.9%
777. Molybdenum, Tube, 50mm, Outside Diameter 6.0mm, Inside Diameter 4.4mm, Wall Thickness 0.8mm, 99.9%
778. Molybdenum, Tube, 50mm, Outside Diameter 6.4mm, Inside Diameter 4.6mm, Wall Thickness 0.9mm, 99.9%
| Molecular Weight | 95.95 g/mol |
|---|---|
| Molecular Formula | Mo |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 0 |
| Exact Mass | 97.905404 g/mol |
| Monoisotopic Mass | 97.905404 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 1 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Trace Elements
A group of chemical elements that are needed in minute quantities for the proper growth, development, and physiology of an organism. (From McGraw-Hill Dictionary of Scientific and Technical Terms, 4th ed) (See all compounds classified as Trace Elements.)
The chronic inhalation of molybdenum dusts (57 mg molybdenum/cu m during 120 days, 4 hr/day) in rats showed a high accumulation of molybdenum in lung, spleen, and heart and a decrease of DNA and RNA levels in liver, kidneys, and spleen. /Molybdenum dusts/
Seiler, H.G., H. Sigel and A. Sigel (eds.). Handbook on the Toxicity of Inorganic Compounds. New York, NY: Marcel Dekker, Inc. 1988., p. 440
Plasma clearance in humans: 4-70 minutes (fast component) and 3-30 hours (slow component); [Nordberg, p. 734]
Nordberg - Nordberg GF, Fowler BA, Nordberg M (eds). Handbook on the Toxicology of Metals, 4th Ed. Boston: Elsevier, 2015., p. 734
ABOUT THIS PAGE
35
PharmaCompass offers a list of Molybdenum API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Molybdenum manufacturer or Molybdenum supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Molybdenum manufacturer or Molybdenum supplier.
A Molybdenum manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Molybdenum, including repackagers and relabelers. The FDA regulates Molybdenum manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Molybdenum API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Molybdenum supplier is an individual or a company that provides Molybdenum active pharmaceutical ingredient (API) or Molybdenum finished formulations upon request. The Molybdenum suppliers may include Molybdenum API manufacturers, exporters, distributors and traders.
Molybdenum Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Molybdenum GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Molybdenum GMP manufacturer or Molybdenum GMP API supplier for your needs.
A Molybdenum CoA (Certificate of Analysis) is a formal document that attests to Molybdenum's compliance with Molybdenum specifications and serves as a tool for batch-level quality control.
Molybdenum CoA mostly includes findings from lab analyses of a specific batch. For each Molybdenum CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Molybdenum may be tested according to a variety of international standards, such as European Pharmacopoeia (Molybdenum EP), Molybdenum JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Molybdenum USP).

