
Synopsis
0
USDMF
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
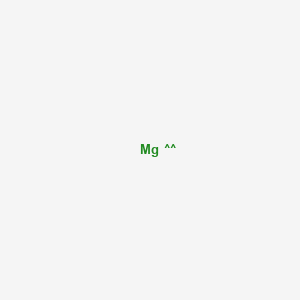
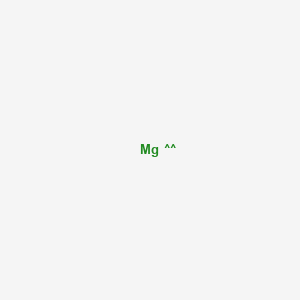
1. 7439-95-4
2. Mg
3. Magnesio
4. Magnesium Powder
5. Magnesium Turnings
6. Magnesium Metallicum
7. Mfcd00085308
8. Magnesium Sheet
9. I38zp9992a
10. Magnesium Powdered
11. Magnesio [italian]
12. Magnesium Metal
13. Rieke's Active Magnesium
14. Magnesium Ribbon
15. Magnesium Compounds
16. Hsdb 654
17. Einecs 231-104-6
18. Un1418
19. Un1869
20. Un2950
21. 12mg
22. Magnesium Atom
23. Magnesium Rod
24. Unii-i38zp9992a
25. Magnesium, Foil, Thickness 0.0125 Mm, Size 25 X 25 Mm, Purity 99.9%
26. Magnesium, Foil, Thickness 0.0125 Mm, Size 50 X 50 Mm, Purity 99.9%
27. Magnesium, Foil, Thickness 0.015 Mm, Size 100 X 100 Mm, Purity 99.9%
28. Magnesium, Foil, Thickness 0.015 Mm, Size 25 X 25 Mm, Purity 99.9%
29. Magnesium, Foil, Thickness 0.015 Mm, Size 50 X 50 Mm, Purity 99.9%
30. Magnesium, Foil, Thickness 0.02 Mm, Size 100 X 100 Mm, Purity 99.9%
31. Magnesium, Foil, Thickness 0.02 Mm, Size 25 X 25 Mm, Purity 99.9%
32. Magnesium, Foil, Thickness 0.02 Mm, Size 50 X 50 Mm, Purity 99.9%
33. Magnesium, Foil, Thickness 0.025 Mm, Size 100 X 100 Mm, Purity 99.9%
34. Magnesium, Foil, Thickness 0.025 Mm, Size 50 X 50 Mm, Purity 99.9%
35. Magnesium, Foil, Thickness 1.0 Mm, Size 100 X 100 Mm, Purity 99.9%
36. Magnesium, Foil, Thickness 1.0 Mm, Size 25 X 25 Mm, Purity 99.9%
37. Magnesium, Foil, Thickness 1.0 Mm, Size 50 X 50 Mm, Purity 99.9%
38. Magnesium(0)
39. Magnesium, Turnings
40. Magnesium (0)
41. Magnesium, Elemental
42. Magnesium (turnings)
43. Magnesium, 99.5%
44. Magnesium [mi]
45. Magnesium [hsdb]
46. Magnesium [vandf]
47. Magnesium Powder 50 Mesh
48. Ec 231-104-6
49. Magnesium, >=99.8%
50. Magnesium [who-dd]
51. Magnesium Powder 325 Mesh
52. Magnesium Turnings, -4 Mesh
53. Magnesium, Powder, >=99%
54. Magnesium, Plate, 99.95%
55. Magnesium Powder, -325 Mesh
56. Magnesium, Puriss., 98.0%
57. Magnesium, Puriss., 99.5%
58. Magnesium Powder [inci]
59. Magnesium Turnings For Grignards
60. Chembl2146125
61. Dtxsid0049658
62. Chebi:25107
63. Hsdb 7065
64. Magnesium Granules, -12+50 Mesh
65. Magnesium Metallicum [hpus]
66. Magnesium Powder, -20+100 Mesh
67. Magnesium Powder,-100+200 Mesh
68. Magnesium Powder, -100+200 Mesh
69. Mfcd00011101
70. Un2010
71. Akos015902697
72. Akos016017174
73. Akos028110047
74. Magnesium, Grit, >=99.0% (kt)
75. Db14513
76. Magnesium, Lr, Turnings, >=99.5%
77. Magnesium Hydride, Hydrogen-storage Grade
78. Magnesium, Ribbon, >=99.5% Mg Basis
79. Magnesium, Saj First Grade, >=98.0%
80. Magnesium, Saj First Grade, >=99.0%
81. Bp-30179
82. Bp-31186
83. Magnesium, Hydrogen Activated, >=99.5%
84. Magnesium, Turnings, Reagent Grade, 98%
85. Q660
86. Rieke Magnesium 2.5g In 100 Ml Of Thf
87. Ft-0628088
88. M3358
89. Magnesium, Ingot, >=99% Trace Metals Basis
90. Magnesium, Ribbon, =99% Trace Metals Basis
91. Magnesium, Powder Or Magnesium Alloys, Powder
92. Magnesium Foil, 1.0mm (0.04in) Thick, Hard
93. Magnesium Foil, 0.25mm (0.01in) Thick, Hard
94. Magnesium, 20-230 Mesh, Reagent Grade, 98%
95. Magnesium Foil, 0.05mm (0.002in) Thick, Hard
96. 2.5 Grams Of Rieke Magnesium In 100 Ml Of Thf
97. 25 Grams Of Rieke(r) Magnesium In 1000ml Of Thf
98. 5.0 Grams Of Rieke(r) Magnesium In 200ml Of Thf
99. Magnesium Foil, 0.125mm (0.005in) Thick, Hard
100. Magnesium Hydride [un2010] [dangerous When Wet]
101. Magnesium Matrix Modifier, For Graphite Furnace-aas
102. 2.5 Grams Of Rieke(r) Magnesium In 100 Ml Of Thf
103. Magnesium Turnings, 1cm (0.4in) & Down, Puratronic?
104. Magnesium, Nist(r) Srm(r) 980, Isotopic Standard
105. M000000010
106. Magnesium, Reagentplus(r), Powder, -50 Mesh, >=99%
107. Magnesium Granules, Coated Particle Size Not <149 Microns
108. Magnesium Rod, 3.3cm (1.3in) Dia X 30cm (12in) Long
109. Magnesium Rod, 7.9mm (0.31in) Dia X 25mm (1in) Length
110. Magnesium, Chips 6-35 Mesh, 99.98% Trace Metals Basis
111. Magnesium, Chips, 6-35 Mesh, 99.98% Trace Metals Basis
112. Magnesium, Rod, Diam. 6 Mm, >=99.9% Trace Metals Basis
113. Magnesium, In A Sure/seal(tm) Bottle, Turnings, 37.5 Mmol
114. Magnesium, Purum, For Grignard Reactions, >=99.5%, Turnings
115. Magnesium, Reagentplus(r), Ribbon, >=99% Trace Metals Basis
116. Magnesium, Turnings, ~1/8 In., 99.95% Trace Metals Basis
117. Magnesium, Wire Reel, 1m, Diameter 0.4mm, As Drawn, 99.9+%
118. Magnesium, Wire Reel, 1m, Diameter 0.5mm, As Drawn, 99.9+%
119. Magnesium, Wire Reel, 1m, Diameter 1.0mm, As Drawn, 99.9+%
120. Magnesium, Wire Reel, 1m, Diameter 1.1mm, As Drawn, 99.9+%
121. Magnesium, Wire Reel, 2m, Diameter 0.4mm, As Drawn, 99.9+%
122. Magnesium, Wire Reel, 2m, Diameter 0.5mm, As Drawn, 99.9+%
123. Magnesium, Wire Reel, 2m, Diameter 1.0mm, As Drawn, 99.9+%
124. Magnesium, Wire Reel, 2m, Diameter 1.1mm, As Drawn, 99.9+%
125. Magnesium, Wire, 127 Mum Diameter, 99.9% Trace Metals Basis
126. Magnesium Slug, 6.35mm (0.25in) Dia X 12.7mm (0.50in) Length
127. Magnesium Slug, 6.35mm (0.25in) Dia X 6.35mm (0.25in) Length
128. Magnesium, Foil, Diameter 8 Mm, Thickness 0.025 Mm, Purity 99.9%
129. Magnesium, Foil, Diameter 8 Mm, Thickness 0.075 Mm, Purity 99.9%
130. Magnesium, Foil, Diameter 8 Mm, Thickness 0.25 Mm, Purity 99.9%
131. Magnesium, Foil, Thickness 0.002 Mm, 4 Mm Diameter, Purity 99.9%
132. Magnesium, Foil, Thickness 0.002 Mm, 6 Mm Diameter, Purity 99.9%
133. Magnesium, Foil, Thickness 0.002 Mm, Diameter 8 Mm, Purity 99.9%
134. Magnesium, Foil, Thickness 0.003 Mm, 4 Mm Diameter, Purity 99.9%
135. Magnesium, Foil, Thickness 0.003 Mm, 6 Mm Diameter, Purity 99.9%
136. Magnesium, Foil, Thickness 0.003 Mm, 8 Mm Diameter, Purity 99.9%
137. Magnesium, Foil, Thickness 0.005 Mm, 4 Mm Diameter, Purity 99.9%
138. Magnesium, Foil, Thickness 0.005 Mm, 6 Mm Diameter, Purity 99.9%
139. Magnesium, Foil, Thickness 0.005 Mm, 8 Mm Diameter, Purity 99.9%
140. Magnesium, Foil, Thickness 0.01 Mm, 15 Mm Diameter, Purity 99.9%
141. Magnesium, Foil, Thickness 0.01 Mm, 4 Mm Diameter, Purity 99.9%
142. Magnesium, Foil, Thickness 0.01 Mm, 8 Mm Diameter, Purity 99.9%
143. Magnesium, Foil, Thickness 0.01 Mm, Diameter 10 Mm, Purity 99.9%
144. Magnesium, Foil, Thickness 0.01 Mm, Diameter 6 Mm, Purity 99.9%
145. Magnesium, Foil, Thickness 0.015 Mm, 6 Mm Diameter, Purity 99.9%
146. Magnesium, Foil, Thickness 0.015 Mm, Diameter 4 Mm, Purity 99.9%
147. Magnesium, Foil, Thickness 0.015 Mm, Diameter 8 Mm, Purity 99.9%
148. Magnesium, Foil, Thickness 0.02 Mm, 15 Mm Diameter, Purity 99.9%
149. Magnesium, Foil, Thickness 0.02 Mm, 4 Mm Diameter, Purity 99.9%
150. Magnesium, Foil, Thickness 0.02 Mm, 6 Mm Diameter, Purity 99.9%
151. Magnesium, Foil, Thickness 0.02 Mm, 8 Mm Diameter, Purity 99.9%
152. Magnesium, Foil, Thickness 0.02 Mm, Diameter 25 Mm, Purity 99.9%
153. Magnesium, Foil, Thickness 0.02 Mm, Purity 99.9%, Diameter 10 Mm
154. Magnesium, Foil, Thickness 0.025 Mm, 4 Mm Diameter, Purity 99.9%
155. Magnesium, Foil, Thickness 0.025 Mm, Diameter 6 Mm, Purity 99.9%
156. Magnesium, Foil, Thickness 0.05 Mm, 10 Mm Diameter, Purity 99.9%
157. Magnesium, Foil, Thickness 0.05 Mm, 15 Mm Diameter, Purity 99.9%
158. Magnesium, Foil, Thickness 0.05 Mm, 4 Mm Diameter, Purity 99.9%
159. Magnesium, Foil, Thickness 0.05 Mm, 8 Mm Diameter, Purity 99.9%
160. Magnesium, Foil, Thickness 0.05 Mm, Diameter 25 Mm, Purity 99.9%
161. Magnesium, Foil, Thickness 0.05 Mm, Diameter 6 Mm, Purity 99.9%
162. Magnesium, Foil, Thickness 0.075 Mm, 4 Mm Diameter, Purity 99.9%
163. Magnesium, Foil, Thickness 0.075 Mm, 6 Mm Diameter, Purity 99.9%
164. Magnesium, Foil, Thickness 0.1 Mm, 10 Mm Diameter, Purity 99.9%
165. Magnesium, Foil, Thickness 0.1 Mm, 15 Mm Diameter, Purity 99.9%
166. Magnesium, Foil, Thickness 0.1 Mm, 25 Mm Diameter, Purity 99.9%
167. Magnesium, Foil, Thickness 0.1 Mm, 4 Mm Diameter, Purity 99.9%
168. Magnesium, Foil, Thickness 0.1 Mm, 6 Mm Diameter, Purity 99.9%
169. Magnesium, Foil, Thickness 0.1 Mm, 8 Mm Diameter, Purity 99.9%
170. Magnesium, Foil, Thickness 0.125 Mm, 4 Mm Diameter, Purity 99.9%
171. Magnesium, Foil, Thickness 0.125 Mm, 6 Mm Diameter, Purity 99.9%
172. Magnesium, Foil, Thickness 0.125 Mm, 8 Mm Diameter, Purity 99.9%
173. Magnesium, Foil, Thickness 0.15 Mm, 10 Mm Diameter, Purity 99.9%
174. Magnesium, Foil, Thickness 0.15 Mm, 4 Mm Diameter, Purity 99.9%
175. Magnesium, Foil, Thickness 0.15 Mm, 8 Mm Diameter, Purity 99.9%
176. Magnesium, Foil, Thickness 0.15 Mm, Diameter 15 Mm, Purity 99.9%
177. Magnesium, Foil, Thickness 0.15 Mm, Diameter 25 Mm, Purity 99.9%
178. Magnesium, Foil, Thickness 0.15 Mm, Diameter 6 Mm, Purity 99.9%
179. Magnesium, Foil, Thickness 0.25 Mm, 10 Mm Diameter, Purity 99.9%
180. Magnesium, Foil, Thickness 0.25 Mm, 4 Mm Diameter, Purity 99.9%
181. Magnesium, Foil, Thickness 0.25 Mm, 6 Mm Diameter, Purity 99.9%
182. Magnesium, Foil, Thickness 0.25 Mm, Diameter 15 Mm, Purity 99.9%
183. Magnesium, Foil, Thickness 0.25 Mm, Diameter 25 Mm, Purity 99.9%
184. Magnesium, Foil, Thickness 0.5 Mm, 10 Mm Diameter, Purity 99.9%
185. Magnesium, Foil, Thickness 0.5 Mm, 15 Mm Diameter, Purity 99.9%
186. Magnesium, Foil, Thickness 0.5 Mm, 4 Mm Diameter, Purity 99.9%
187. Magnesium, Foil, Thickness 0.5 Mm, 6 Mm Diameter, Purity 99.9%
188. Magnesium, Foil, Thickness 0.5 Mm, Diameter 25 Mm, Purity 99.9%
189. Magnesium, Foil, Thickness 0.5 Mm, Diameter 8 Mm, Purity 99.9%
190. Magnesium, Foil, Thickness 1.0 Mm, 10 Mm Diameter, Purity 99.9%
191. Magnesium, Foil, Thickness 1.0 Mm, 15 Mm Diameter, Purity 99.9%
192. Magnesium, Foil, Thickness 1.0 Mm, 25 Mm Diameter, Purity 99.9%
193. Magnesium, Foil, Thickness 1.0 Mm, 6 Mm Diameter, Purity 99.9%
194. Magnesium, Foil, Thickness 1.0 Mm, 8 Mm Diameter, Purity 99.9%
195. Magnesium, Foil, Thickness 1.0 Mm, Diameter 4 Mm, Purity 99.9%
196. Magnesium, Foil, Thickness 1.5 Mm, 15 Mm Diameter, Purity 99.9%
197. Magnesium, Foil, Thickness 1.5 Mm, 25 Mm Diameter, Purity 99.9%
198. Magnesium, Foil, Thickness 1.5 Mm, 4 Mm Diameter, Purity 99.9%
199. Magnesium, Foil, Thickness 1.5 Mm, 6 Mm Diameter, Purity 99.9%
200. Magnesium, Foil, Thickness 1.5 Mm, 8 Mm Diameter, Purity 99.9%
201. Magnesium, Foil, Thickness 1.5 Mm, Diameter 10 Mm, Purity 99.9%
202. Magnesium, Rod, 1.6 Mm Diameter, Length 1000 Mm, Purity 99.9%
203. Magnesium, Rod, 1.6 Mm Diameter, Length 200 Mm, Purity 99.9%
204. Magnesium, Rod, 1.6 Mm Diameter, Length 500 Mm, Purity 99.9%
205. Magnesium, Rod, 1.6 Mm Diameter, Purity 99.9%, Length 100 Mm
206. Magnesium, Rod, 10 Mm Diameter, Length 100 Mm, Purity 99.97%
207. Magnesium, Rod, 10 Mm Diameter, Length 200 Mm, Purity 99.97%
208. Magnesium, Rod, 10 Mm Diameter, Length 50 Mm, Purity 99.97%
209. Magnesium, Rod, 12.7 Mm Diameter, Length 100 Mm, Purity 99.9%
210. Magnesium, Rod, 12.7 Mm Diameter, Length 500 Mm, Purity 99.9%
211. Magnesium, Rod, 12.7 Mm Diameter, Purity 99.9%, Length 200 Mm
212. Magnesium, Rod, 15 Mm Diameter, Length 50 Mm, Purity 99.97%
213. Magnesium, Rod, 25.4 Mm Diameter, Length 100 Mm, Purity 99.9%
214. Magnesium, Rod, 25.4 Mm Diameter, Length 1000 Mm, Purity 99.9%
215. Magnesium, Rod, 25.4 Mm Diameter, Length 200 Mm, Purity 99.9%
216. Magnesium, Rod, 25.4 Mm Diameter, Length 500 Mm, Purity 99.9%
217. Magnesium, Rod, 3.2 Mm Diameter, Length 100 Mm, Purity 99.9%
218. Magnesium, Rod, 3.2 Mm Diameter, Length 1000 Mm, Purity 99.9%
219. Magnesium, Rod, 3.2 Mm Diameter, Length 200 Mm, Purity 99.9%
220. Magnesium, Rod, 4.8 Mm Diameter, Length 100 Mm, Purity 99.9%
221. Magnesium, Rod, 4.8 Mm Diameter, Length 1000 Mm, Purity 99.9%
222. Magnesium, Rod, 4.8 Mm Diameter, Length 200 Mm, Purity 99.9%
223. Magnesium, Rod, 4.8 Mm Diameter, Length 500 Mm, Purity 99.9%
224. Magnesium, Rod, 50 Mm Diameter, Length 100 Mm, Purity 99.9%
225. Magnesium, Rod, 50 Mm Diameter, Length 200 Mm, Purity 99.9%
226. Magnesium, Rod, 7.9 Mm Diameter, Length 100 Mm, Purity 99.9%
227. Magnesium, Rod, 7.9 Mm Diameter, Length 1000 Mm, Purity 99.9%
228. Magnesium, Rod, 7.9 Mm Diameter, Length 200 Mm, Purity 99.9%
229. Magnesium, Rod, 7.9 Mm Diameter, Length 500 Mm, Purity 99.9%
230. Magnesium, Rod, 76 Mm Diameter, Length 150 Mm, Purity 99.9%
231. Magnesium, Rod, Length 100 Mm, 15 Mm Diameter, Purity 99.97%
232. Magnesium, Rod, Length 1000 Mm, 12.7 Mm Diameter, Purity 99.9%
233. Magnesium, Rod, Length 500 Mm, 3.2 Mm Diameter, Purity 99.9%
234. Magnesium, Rod, Length 500 Mm, 50 Mm Diameter, Purity 99.9%
235. Magnesium, Wire Reel, 0.1m, Diameter 0.125mm, As Drawn, 99.9+%
236. Magnesium, Wire Reel, 0.1m, Diameter 0.25mm, As Drawn, 99.9+%
237. Magnesium, Wire Reel, 0.1m, Diameter 0.4mm, As Drawn, 99.9+%
238. Magnesium, Wire Reel, 0.1m, Diameter 0.5mm, As Drawn, 99.9+%
239. Magnesium, Wire Reel, 0.1m, Diameter 1.0mm, As Drawn, 99.9+%
240. Magnesium, Wire Reel, 0.1m, Diameter 1.1mm, As Drawn, 99.9+%
241. Magnesium, Wire Reel, 0.5m, Diameter 0.125mm, As Drawn, 99.9+%
242. Magnesium, Wire Reel, 0.5m, Diameter 0.25mm, As Drawn, 99.9+%
243. Magnesium, Wire Reel, 0.5m, Diameter 0.4mm, As Drawn, 99.9+%
244. Magnesium, Wire Reel, 0.5m, Diameter 0.5mm, As Drawn, 99.9+%
245. Magnesium, Wire Reel, 0.5m, Diameter 1.0mm, As Drawn, 99.9+%
246. Magnesium, Wire Reel, 0.5m, Diameter 1.1mm, As Drawn, 99.9+%
247. Magnesium, Wire Reel, 1m, Diameter 0.125mm, As Drawn, 99.9+%
248. Magnesium, Wire Reel, 1m, Diameter 0.25mm, As Drawn, 99.9+%
249. Magnesium, Wire Reel, 2m, Diameter 0.125mm, As Drawn, 99.9+%
250. Magnesium, Wire Reel, 2m, Diameter 0.25mm, As Drawn, 99.9+%
251. Aquanal(tm)-plus Magnesium (mg) 100-1500 Mg/l, Refill Pack For 37426
252. Magnesium Cube, 19mm (0.75in) X 19mm (0.75in) X 19mm (0.75in)
253. Magnesium Slug, 3.175mm (0.125in) Dia X 3.175mm (0.125in) Length
254. Magnesium, Foil, Diameter 10 Mm, Thickness 0.003 Mm, Purity 99.9%
255. Magnesium, Foil, Thickness 0.002 Mm, 10 Mm Diameter, Purity 99.9%
256. Magnesium, Foil, Thickness 0.002 Mm, 15 Mm Diameter, Purity 99.9%
257. Magnesium, Foil, Thickness 0.002 Mm, Size 25 X 25 Mm, Purity 99.9%
258. Magnesium, Foil, Thickness 0.002 Mm, Size 50 X 50 Mm, Purity 99.9%
259. Magnesium, Foil, Thickness 0.003 Mm, 15 Mm Diameter, Purity 99.9%
260. Magnesium, Foil, Thickness 0.003 Mm, Size 25 X 25 Mm, Purity 99.9%
261. Magnesium, Foil, Thickness 0.003 Mm, Size 50 X 50 Mm, Purity 99.9%
262. Magnesium, Foil, Thickness 0.005 Mm, Diameter 10 Mm, Purity 99.9%
263. Magnesium, Foil, Thickness 0.005 Mm, Diameter 15 Mm, Purity 99.9%
264. Magnesium, Foil, Thickness 0.005 Mm, Size 25 X 25 Mm, Purity 99.9%
265. Magnesium, Foil, Thickness 0.005 Mm, Size 50 X 50 Mm, Purity 99.9%
266. Magnesium, Foil, Thickness 0.01 Mm, Size 100 X 100 Mm, Purity 99.9%
267. Magnesium, Foil, Thickness 0.01 Mm, Size 25 X 25 Mm, Purity 99.9%
268. Magnesium, Foil, Thickness 0.01 Mm, Size 50 X 50 Mm, Purity 99.9%
269. Magnesium, Foil, Thickness 0.0125 Mm, 25 Mm Diameter, Purity 99.9%
270. Magnesium, Foil, Thickness 0.0125 Mm, 8 Mm Diameter, Purity 99.9%
271. Magnesium, Foil, Thickness 0.0125 Mm, Diameter 10 Mm, Purity 99.9%
272. Magnesium, Foil, Thickness 0.0125 Mm, Diameter 15 Mm, Purity 99.9%
273. Magnesium, Foil, Thickness 0.0125 Mm, Diameter 4 Mm, Purity 99.9%
274. Magnesium, Foil, Thickness 0.0125 Mm, Diameter 6 Mm, Purity 99.9%
275. Magnesium, Foil, Thickness 0.0125 Mm, Size 100 X 100 Mm, Purity 99.9%
276. Magnesium, Foil, Thickness 0.015 Mm, 10 Mm Diameter, Purity 99.9%
277. Magnesium, Foil, Thickness 0.015 Mm, 15 Mm Diameter, Purity 99.9%
278. Magnesium, Foil, Thickness 0.015 Mm, Diameter 25 Mm, Purity 99.9%
279. Magnesium, Foil, Thickness 0.025 Mm, 10 Mm Diameter, Purity 99.9%
280. Magnesium, Foil, Thickness 0.025 Mm, 25 Mm Diameter, Purity 99.9%
281. Magnesium, Foil, Thickness 0.025 Mm, Diameter 15 Mm, Purity 99.9%
282. Magnesium, Foil, Thickness 0.025 Mm, Size 25 X 25 Mm, Purity 99.9%
283. Magnesium, Foil, Thickness 0.05 Mm, Size 100 X 100 Mm, Purity 99.9%
284. Magnesium, Foil, Thickness 0.05 Mm, Size 25 X 25 Mm, Purity 99.9%
285. Magnesium, Foil, Thickness 0.05 Mm, Size 50 X 50 Mm, Purity 99.9%
286. Magnesium, Foil, Thickness 0.075 Mm, 10 Mm Diameter, Purity 99.9%
287. Magnesium, Foil, Thickness 0.075 Mm, 15 Mm Diameter, Purity 99.9%
288. Magnesium, Foil, Thickness 0.075 Mm, Diameter 25 Mm, Purity 99.9%
289. Magnesium, Foil, Thickness 0.075 Mm, Size 100 X 100 Mm, Purity 99.9%
290. Magnesium, Foil, Thickness 0.075 Mm, Size 25 X 25 Mm, Purity 99.9%
291. Magnesium, Foil, Thickness 0.075 Mm, Size 50 X 50 Mm, Purity 99.9%
292. Magnesium, Foil, Thickness 0.1 Mm, Size 100 X 100 Mm, Purity 99.9%
293. Magnesium, Foil, Thickness 0.1 Mm, Size 25 X 25 Mm, Purity 99.9%
294. Magnesium, Foil, Thickness 0.1 Mm, Size 50 X 50 Mm, Purity 99.9%
295. Magnesium, Foil, Thickness 0.125 Mm, 10 Mm Diameter, Purity 99.9%
296. Magnesium, Foil, Thickness 0.125 Mm, 15 Mm Diameter, Purity 99.9%
297. Magnesium, Foil, Thickness 0.125 Mm, 25 Mm Diameter, Purity 99.9%
298. Magnesium, Foil, Thickness 0.125 Mm, Size 100 X 100 Mm, Purity 99.9%
299. Magnesium, Foil, Thickness 0.125 Mm, Size 25 X 25 Mm, Purity 99.9%
300. Magnesium, Foil, Thickness 0.125 Mm, Size 50 X 50 Mm, Purity 99.9%
301. Magnesium, Foil, Thickness 0.15 Mm, Size 100 X 100 Mm, Purity 99.9%
302. Magnesium, Foil, Thickness 0.15 Mm, Size 25 X 25 Mm, Purity 99.9%
303. Magnesium, Foil, Thickness 0.15 Mm, Size 50 X 50 Mm, Purity 99.9%
304. Magnesium, Foil, Thickness 0.25 Mm, Size 100 X 100 Mm, Purity 99.9%
305. Magnesium, Foil, Thickness 0.25 Mm, Size 150 X 150 Mm, Purity 99.9%
306. Magnesium, Foil, Thickness 0.25 Mm, Size 25 X 25 Mm, Purity 99.9%
307. Magnesium, Foil, Thickness 0.25 Mm, Size 50 X 50 Mm, Purity 99.9%
308. Magnesium, Foil, Thickness 0.5 Mm, Size 100 X 100 Mm, Purity 99.9%
309. Magnesium, Foil, Thickness 0.5 Mm, Size 150 X 150 Mm, Purity 99.9%
310. Magnesium, Foil, Thickness 0.5 Mm, Size 25 X 25 Mm, Purity 99.9%
311. Magnesium, Foil, Thickness 0.5 Mm, Size 50 X 50 Mm, Purity 99.9%
312. Magnesium, Foil, Thickness 1.0 Mm, Size 150 X 150 Mm, Purity 99.9%
313. Magnesium, Foil, Thickness 1.5 Mm, Size 100 X 100 Mm, Purity 99.9%
314. Magnesium, Foil, Thickness 1.5 Mm, Size 25 X 25 Mm, Purity 99.9%
315. Magnesium, Foil, Thickness 1.5 Mm, Size 50 X 50 Mm, Purity 99.9%
316. Magnesium, Foil, Thickness 2.0 Mm, Size 100 X 100 Mm, Purity 99.9%
317. Magnesium, Foil, Thickness 2.0 Mm, Size 25 X 25 Mm, Purity 99.9%
318. Magnesium, Foil, Thickness 2.0 Mm, Size 50 X 50 Mm, Purity 99.9%
319. Magnesium, Foil, Thickness 3.0 Mm, Size 100 X 100 Mm, Purity 99.9%
320. Magnesium, Foil, Thickness 3.0 Mm, Size 50 X 50 Mm, Purity 99.9%
321. Magnesium, Foil, Thickness 6.0 Mm, Size 100 X 100 Mm, Purity 99.9%
322. Magnesium, Foil, Thickness 6.0 Mm, Size 50 X 50 Mm, Purity 99.9%
323. Magnesium, Reagentplus(r), Powder, -325 Mesh, 99.5% Trace Metals Basis
324. Magnesium Granules, Coated Particle Size Not <149 Microns [un2950] [dangerous When Wet]
325. Magnesium Or Magnesium Alloys With >50% Magnesium In Pellets, Turnings Or Ribbons
326. Magnesium Preparation, Highly Reactive Rieke(r)metal, Suspension, 2.5 G In Thf
327. Magnesium Ribbon, Approximate Dimensions, 3.0mm (0.12in) Wide, 0.15mm (0.0059in) Thick
328. Magnesium Slug, 3.175mm (0.125in) Dia X 6.35mm (0.25in) Length, Annealed
329. Magnesium Slug, 6.35mm (0.25in) Dia X 12.7mm (0.50in) Length, 99.95% (metals Basis)
330. Magnesium Slug, 6.35mm (0.25in) Dia X 6.35mm (0.25in) Length, 99.95% (metals Basis)
331. Magnesium Sputtering Target, 50.8mm (2.0in) Dia X 6.35mm (0.250in) Thick
332. Magnesium Sputtering Target, 76.2mm (3.0in) Dia X 3.18mm (0.125in) Thick
333. Magnesium Sputtering Target, 76.2mm (3.0in) Dia X 6.35mm (0.250in) Thick
334. Magnesium, Dendritic Pieces, Purified By Distillation, 99.99% Trace Metals Basis
335. Magnesium, Dendritic Pieces, Purified By Distillation, 99.998% Trace Metals Basis
336. Magnesium, In A Sure/seal(tm) Bottle, Turnings, Anhydrous Tetrahydrofuran 37.5 Mmol
337. Magnesium, Powder, 250 Max. Part. Size (micron), Weight 1000 G, Purity 99.8+%
338. Magnesium, Powder, 50 Max. Part. Size (micron), Purity 99.8%, Weight 1000 G
339. Magnesium, Powder, 50 Max. Part. Size (micron), Weight 100 G, Purity 99.8%
340. Magnesium, Powder, 50 Max. Part. Size (micron), Weight 200 G, Purity 99.8%
341. Magnesium, Powder, Max. Particle Size 250 Micron, Weight 100 G, Purity 99.8+%
342. Magnesium, Powder, Max. Particle Size 250 Micron, Weight 200 G, Purity 99.8+%
343. Magnesium, Powder, Max. Particle Size 250 Micron, Weight 500 G, Purity 99.8+%
344. Magnesium, Powder, Max. Particle Size 50 Micron, Weight 500 G, Purity 99.8%
345. Aquanal(tm)-plus Magnesium (mg) 100-1500 Mg/l, Check Solution For 37426 (calibrated Standard (mg = 0.3 Mg/ml))
346. Aquanal(tm)-plus Magnesium (mg) 100-1500 Mg/l, Test Set With Color Comparator And Check Solution
347. Magnesium Or Magnesium Alloys With >50% Magnesium In Pellets, Turnings Or Ribbons [un1869] [flammable Solid]
348. Magnesium Slug, 3.175mm (0.125in) Dia X 6.35mm (0.25in) Length, Annealed, 99.95% (metals Basis)
349. Magnesium, Microfoil, 25x25mm, Thinness 1.0mum, Specific Density 174mug/cm2, 6 Micron Aluminum Permanent Support, 99.9%
350. Magnesium, Microfoil, 50x50mm, Thinness 1.0mum, Specific Density 174mug/cm2, 6 Micron Aluminum Permanent Support, 99.9%
351. Magnesium, Microleaf, 25x25mm, Thinness 1.0mum, Specific Density 174mug/cm2, Removable Support, 99.9%
352. Magnesium, Microleaf, 50x50mm, Thinness 1.0mum, Specific Density 174mug/cm2, Removable Support, 99.9%
353. Magnesium, Powder Or Magnesium Alloys, Powder [un1418] [dangerous When Wet, Spontaneously Combustible]
| Molecular Weight | 24.305 g/mol |
|---|---|
| Molecular Formula | Mg |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 0 |
| Exact Mass | 23.9850417 g/mol |
| Monoisotopic Mass | 23.9850417 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 1 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Healthy levels of magnesium can be achieved through a well balanced diet, but if food sources are insufficient, magnesium supplements can be used to prevent and treat magnesium deficiencies. In medicine, various magnesium salts may be used in laxative and antacid products. For example, [magnesium citrate] is available over-the-counter and may be used to manage occasional constipation. [Magnesium sulfate] may be used on its own or with total parenteral nutrition to treat hypomagnesemia. [Magnesium sulfate] is also indicated to prevent seizures in pregnant women with pre-eclampsia, and to manage seizures associated with eclampsia.
Magnesium is important for many biochemical processes and is therefore quite common in humans. The majority of magnesium is stored in the bones (>50%), while the remainder is stored in muscle, soft tissue, red blood cells and serum. This is functionally important since the bones behave as a magnesium exchange reservoir and help maintain healthy levels of magnesium. Magnesium plays an important role in the regulation of several bodily processes including blood pressure, insulin metabolism, muscular contraction, vasomotor tone, cardiac excitability, nerve transmission and neuromuscular conduction. Disruptions in homeostatic levels of magnesium (often times hypomagnesemia) can impact the nervous system, muscles, or can lead to cardiac abnormalities.
Absorption
Approximately 24-76% of ingested magnesium is absorbed in the gastrointestinal tract, primarily via passive paracellular absorption in the small intestine.
Route of Elimination
The majority of magnesium is excreted renally.
Volume of Distribution
According to a pharmacokinetic review, the volume of distribution of magnesium sulphate when used to manage patients with pre-eclampsia and eclampsia ranged from 13.65 to 49.00 L.
Magnesium does not appear to be metabolized.
Magnesiums biologic half-life is reported to be approximately 1000 hours or 42 days.
Magnesium is a cofactor for at least 300 enzymes and is important for several functions in the body with some key processes identified below. Enzymes that rely on magnesium to operate help produce energy through oxidative phosphorylation, glycolysis and ATP metabolism. They are also involved in nerve function, muscle contraction, blood glucose control, hormone receptor binding, protein synthesis, cardiac excitability, blood pressure control, gating of calcium channels and transmembrane ion flux. The mitochondrial intracellular space is rich in magnesium, since it is required to produce the active form of ATP (adenosine triphosphate) from ADP (adenosine diphosphate) and inorganic phosphate, and behaves as a counter ion for the energy rich molecule. Additionally, magnesium is essential for ATP metabolism.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
80
PharmaCompass offers a list of Magnesium API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Magnesium manufacturer or Magnesium supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Magnesium manufacturer or Magnesium supplier.
A Magnesium manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Magnesium, including repackagers and relabelers. The FDA regulates Magnesium manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Magnesium API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Magnesium manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Magnesium supplier is an individual or a company that provides Magnesium active pharmaceutical ingredient (API) or Magnesium finished formulations upon request. The Magnesium suppliers may include Magnesium API manufacturers, exporters, distributors and traders.
click here to find a list of Magnesium suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Magnesium CEP of the European Pharmacopoeia monograph is often referred to as a Magnesium Certificate of Suitability (COS). The purpose of a Magnesium CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Magnesium EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Magnesium to their clients by showing that a Magnesium CEP has been issued for it. The manufacturer submits a Magnesium CEP (COS) as part of the market authorization procedure, and it takes on the role of a Magnesium CEP holder for the record. Additionally, the data presented in the Magnesium CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Magnesium DMF.
A Magnesium CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Magnesium CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Magnesium suppliers with CEP (COS) on PharmaCompass.
A Magnesium written confirmation (Magnesium WC) is an official document issued by a regulatory agency to a Magnesium manufacturer, verifying that the manufacturing facility of a Magnesium active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Magnesium APIs or Magnesium finished pharmaceutical products to another nation, regulatory agencies frequently require a Magnesium WC (written confirmation) as part of the regulatory process.
click here to find a list of Magnesium suppliers with Written Confirmation (WC) on PharmaCompass.
Magnesium Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Magnesium GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Magnesium GMP manufacturer or Magnesium GMP API supplier for your needs.
A Magnesium CoA (Certificate of Analysis) is a formal document that attests to Magnesium's compliance with Magnesium specifications and serves as a tool for batch-level quality control.
Magnesium CoA mostly includes findings from lab analyses of a specific batch. For each Magnesium CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Magnesium may be tested according to a variety of international standards, such as European Pharmacopoeia (Magnesium EP), Magnesium JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Magnesium USP).

