
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
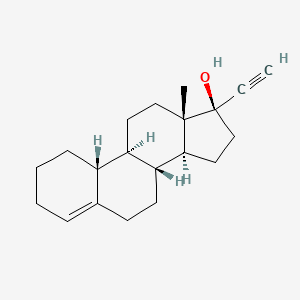

1. 19-norpregn-4-en-20-yn-17-ol, (17alpha)-
2. Ethinylestrenol
3. Exluton
4. Linesterol
5. Linestrenol
6. Lynestrenol, (9 Beta, 10 Alpha,17 Alpha)-isomer
7. Lynoestrenol
1. 52-76-6
2. Linestrenol
3. Lynoestrenol
4. Ethinylestrenol
5. Ethynylestrenol
6. Orgametril
7. Orgametrol
8. Exluton
9. Endometril
10. Exlution
11. Exlutona
12. Orgametil
13. 3-desoxynorlutin
14. Nsc-37725
15. Org 485-50
16. Ind 1006
17. 17.alpha.-ethynylestrenol
18. N2z8alg4u5
19. Exluten
20. Chebi:31790
21. 19-nor-17.alpha.-pregn-4-en-20-yn-17-ol
22. 19-norpregn-4-en-20-yn-17-ol, (17a)-
23. Nsc37725
24. 19-norpregn-4-en-20-yn-17-ol, (17.alpha.)-
25. (8r,9s,10r,13s,14s,17r)-17-ethynyl-13-methyl-2,3,6,7,8,9,10,11,12,14,15,16-dodecahydro-1h-cyclopenta[a]phenanthren-17-ol
26. 19-norpregn-4-en-20-yn-17alpha-ol
27. Ethynloestrenol
28. Estr-4-en-17.beta.-ol, 17-ethynyl-
29. Lynenol
30. Lynstranol
31. Ethinyloestranol
32. Ethinyl Oestrenol
33. .delta.4-17.alpha.-ethinylestren-17.beta.-ol
34. Linestrenolo [dcit]
35. Linestrenolo
36. Lynestrenolum
37. 17alpha-ethynylestrenol
38. 17-alpha-ethynylestrenol
39. 17-alpha-ethynyloestrenol
40. Linestrenol [inn-spanish]
41. Lynestrenolum [inn-latin]
42. Ccris 9093
43. 19 Norpregn-4-en-20-yn-17-ol, (17alpha)-
44. Einecs 200-151-4
45. Lynoestrenol [progestins]
46. Nsc 37725
47. 19-norpregn-4-en-20-yn-17-ol, (17alpha)-
48. Estr-4-en-17beta-ol, 17-ethynyl-
49. Unii-n2z8alg4u5
50. 17-alpha-ethynylestr-4-en-17-beta-ol
51. Delta4-17alpha-ethinylestren-17beta-ol
52. 17-alpha-ethynyloestr-4-en-17-beta-ol
53. 17-alpha-ethinyl-17-beta-hydroxyestr-4-ene
54. 19-nor-17alpha-pregn-4-en-20-yn-17-ol
55. 17-alpha-ethinyl-17-beta-hydroxyoestr-4-ene
56. Hsdb 7899
57. 17-alpha-ethynil-delta-4-estrene-17-beta-ol
58. (17-alpha)-19-norpregn-4-en-20-yn-17-ol
59. Delta(sup 4)-17-alpha-ethinylestren-17-beta-ol
60. Delta(sup 4)-17-alpha-ethinyloestren-17-beta-ol
61. Lynestrenol [usan:inn:ban:jan]
62. Prestwick_533
63. Lynestrenol [mi]
64. Prestwick0_000095
65. Prestwick1_000095
66. Prestwick2_000095
67. Prestwick3_000095
68. Lynestrenol [inn]
69. Lynestrenol [jan]
70. Lynestrenol (jan/usan)
71. Lynestrenol [usan]
72. 19-nor-17-alpha-pregn-4-en-20-yn-17-ol
73. Lynestrenol [mart.]
74. Schembl37816
75. Bspbio_000050
76. Lynestrenol [usp-rs]
77. Lynestrenol [who-dd]
78. Spbio_001989
79. 17-ethynylestr-4-en-17-ol
80. Bpbio1_000056
81. Chembl2107431
82. Dtxsid4021478
83. Hms1568c12
84. Hms2095c12
85. Hms3712c12
86. Lynestrenol [ep Monograph]
87. Bcp04136
88. Nsc73879
89. Zinc3875355
90. Lmst02030127
91. Mfcd00051135
92. Nsc-73879
93. S4883
94. Akos024462708
95. 19-nor-4-pregnen-20-yn-17beta-ol
96. Ccg-220095
97. Db12474
98. Ncgc00179676-01
99. L0246
100. 19-nor-17-alpha-pregn-4-en-20-yn-17-l
101. C75475
102. D01580
103. 052l766
104. A829241
105. Q2071071
106. 19 Norpregn-4-en-20-yn-17-ol, (17.alpha.)-
107. (8r,9s,10r,13s,14s,17s)-17-ethynyl-13-methyl-2,3,6,7,8,9,10,11,12,14,15,16-dodecahydro-1h-cyclopenta[a]phenanthren-17-ol;lynestrenol
108. 17-ethynyl-13-methyl-2,3,6,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1h-cyclopeta[a]phenanthren-17-ol
| Molecular Weight | 284.4 g/mol |
|---|---|
| Molecular Formula | C20H28O |
| XLogP3 | 4.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Exact Mass | 284.214015512 g/mol |
| Monoisotopic Mass | 284.214015512 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 523 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Contraceptives, Oral, Synthetic
Oral contraceptives which owe their effectiveness to synthetic preparations. (See all compounds classified as Contraceptives, Oral, Synthetic.)
Contraceptives, Oral, Hormonal
Oral contraceptives which owe their effectiveness to hormonal preparations. (See all compounds classified as Contraceptives, Oral, Hormonal.)
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03A - Hormonal contraceptives for systemic use
G03AC - Progestogens
G03AC02 - Lynestrenol
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03D - Progestogens
G03DC - Estren derivatives
G03DC03 - Lynestrenol
The tissue distribution of 4-14C-lynestrenol (17 alpha-ethynyl-oestr-4-en-17 beta-ol) following oral administration to pregnant rats was studied by whole body autoradiography and liquid scintillation counting. Pregnant females were sacrificed on days 10, 12, 14 and 19 of gestation, in each case 5 hours after oral administration of 43 uCi 4-14C-lynestrenol per animal. The isotopelabelled compound was distributed throughout most tissues, including the fetuses. The highest concentrations were found in the liver, while there was lower activity in the fatty tissues and the activity in the fetuses was comparable with that in the brain. The placental transfer was verified by the results of liquid scintillation counting. The concentration of labelled substance in the fetuses increased with the duration of pregnancy.
PMID:7361560 Sannes E, Nafstad I; Acta Pharmacol Toxicol (Copenh) 46 (1): 47-50 (1980)
... The evaluation of plasma norethindrone levels in subjects receiving lynestrenol has not yet been reported. After the administration of norethindrone, the peak level of norethindrone in the plasma was obtained within 2 hours, and the peak concentration in the plasma was about 3.5 ng/mL/mg norethindrone. During a period of 2-6 hours after the administration of norethindrone, the half-life of norethindrone in the plasma was approximately 1.8 hours, and during the period of 6-24 hours, half-life was variable. On the other hand, after lynestrenol administration, the peak level of norethindrone in the plasma was obtained within 4 hours, and the peak concentration of norethindrone was about 1.9 ng/mL/mg lynestrenol. During a period of 4-12 hours, the half-life of norethindrone was about 2.5 hours. The peak of norethindrone level after lynestrenol administration was lower and appeared later than that after administration of the same dose of norethindrone. Norethindrone in plasma in subjects receiving lynestrenol could be measured for a longer period than in those receiving the same dose of norethindrone. These results suggest that lynestrenol is stored in fat tissue and is slowly metabolized to norethindrone.
PMID:7319102 Yasuda J et al: Nippon Naibunpi Gakkai Zasshi 57 (8): 1159-66 (1981)
The peak concentration, peak time, the area under the serum concentration time curve (AUC) and half-life of serum norethisterone (NET) after a single application of lynestrenol (LYN) to female volunteers demonstrated that 0.7 mg NET is bioequivalent to 1 mg LYN which is rapidly converted to NET. There was a decrease of the peak values and an increase of half-life of NET during the periovulatory and luteal phase which was, however, not significant due to the great individual differences. The shift of the peak time to longer intervals and the increase of half-life of NET after ingestion of higher LYN doses indicate a certain limitation of the metabolic capacity of the liver. ...
PMID:7172676 Kuhl H et al; Contraception 26 (3): 303-15 (1982)
... Eleven normal males were administer either 1 capsule containing 2.5 mg lynestrenol and 0.05 mg ethynylestradiol or tablets containing the same dose of steroids. Blood samples were taken just before and 1,2,4,8, and 24 hours after the drugs were administered. 8 days later the subjects were given the opposite formulations and blood samples were again obtained at the same time intervals. Radioimmunoassay was used to assess levels of free and conjugated ethynylestradiol and norethisterone in the serum samples. Biovailability assessments were made by calculating the areas under the time-concentration curves. Results demonstrated that the conversion of lynestrenol to norethisterone in vivo was complete. Unconjugated ethynylestradiol mean values, 2 and 4 hours after administration of the drugs, were higher for capsules than for tablets. For blood samples taken at other time periods the mean values of unconjugated ethynylestradiol were similar for both capsules and tablets. For conjugated ethynylestradiol the mean values and the areas under the curve, for blood samples obtained at each time interval, showed no significant difference for tablets and capsules.
PMID:7387763 Shrimanker K et al; Arzneimittelforschung 30 (3): 500-2 (1980)
167 women were treated with 0.5 mg lynestrenol/day for a period of 8-32 weeks immediately postpartum. The lactation periods of 67 of the women were observed during the stay in hospital (maximum 7 days) in comparison with a control group (n = 20) of untreated women in the puerperium. There was no quantitative difference in milk production between the lynestrenol-treated women and the control group. ...
PMID:408604 Mettler L et al; MMW Munch Med Wochenschr 119 (25): 853-6 (1977)
The concentration of norethindrone in plasma samples from subjects receiving norethindrone (norethisterone) and lynestrenol orally was measured by high pressure liquid chromatography (HPLC). Norethindrone is a synthetic gestagen widely used in contraceptive formulations, and lynestrenol is also a synthetic gestagen which is metabolized to norethindrone in humans.
PMID:7319102 Yasuda J et al: Nippon Naibunpi Gakkai Zasshi 57 (8): 1159-66 (1981)
Although /little/ is known about the transformation of lynestrenol and norethynodrel, it appears that lynestrenol first undergoes hydroxylation at carbon 3 and then oxidation of the hydroxyl group to form norethisterone.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V91 146 (2007)
There is limited information on the metabolism of levonorgestrel, norethindrone and structurally related contraceptive steroids. Both levonorgestrel and norethindrone undergo extensive reduction of the alpha, beta-unsaturated ketone in ring A. Levonorgestrel also undergoes hydroxylation at carbons 2 and 16. The metabolites of both compounds circulate predominantly as sulfates. In urine, levonorgestrel metabolites are found primarily in the glucuronide form, whereas norethindrone metabolites are present in approximately equal amounts as sulfates and glucuronides. Of the progestogens structurally related to norethindrone, norethindrone acetate, ethynodiol diacetate, norethindrone enanthate, and perhaps lynestrenol, undergo rapid hydrolysis and are converted to the parent compound and its metabolites. There is no convincing evidence that norethynodrel is converted to norethindrone. Of the progestogens structurally related to levonorgestrel, it appears that neither desogestrel nor gestodene are transformed to the parent compound. However, there is evidence that norgestimate can be, at least partly, converted to levonorgestrel. ...
PMID:2143719 Stanczyk FZ, Roy S; Contraception 42 (1): 67-96 (1990)
Lynestrenol, a synthetic steroid progesterone-like substance, has been found to enhance the stimulation of normal human lymphocytes. It increases the phytohemagglutinin response and the mixed-lymphocyte cultures. This augmentation can, in some cases, be higher than 100% of the phytohemagglutinin or mixed lymphocyte reaction baseline. There is no significant effect on the pokeweed mitogen stimulation. It is concluded that lynestrenol can modulate, "in vitro", some T cell functions.
PMID:856311 Wybran J et al; Biomedicine 27 (1): 16-9 (1977)
In 10 women taking 0.5 mg lynestrenol for oral contraception endometrium biopsies were carried out in several consecutive cycles between the 21st and 25th day of each cycle. In addition pregnandiol excretion in the urine was measured in one of the last three treatment cycles always on the 10th and 21st day of cycle. During treatment of a characteristic, monomorphic microscopical endometrium picture was observed with hardly distinct endometrium glands and only slight decidualike transformation of cellular elements of the stroma. From these histological pictures it is concluded that oral application of 0,5 mg lynestrenol produces endometrium changes characterized by insufficient transformation so that nidation of a blastocyte becomes impossible.
PMID:1246215 Gerber E, Koch P; Med Klin 71 (2): 51-5 (1976)
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
70
PharmaCompass offers a list of Lynestrenol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Lynestrenol manufacturer or Lynestrenol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Lynestrenol manufacturer or Lynestrenol supplier.
A Lynestrenol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Lynestrenol, including repackagers and relabelers. The FDA regulates Lynestrenol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Lynestrenol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Lynestrenol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Lynestrenol supplier is an individual or a company that provides Lynestrenol active pharmaceutical ingredient (API) or Lynestrenol finished formulations upon request. The Lynestrenol suppliers may include Lynestrenol API manufacturers, exporters, distributors and traders.
click here to find a list of Lynestrenol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Lynestrenol written confirmation (Lynestrenol WC) is an official document issued by a regulatory agency to a Lynestrenol manufacturer, verifying that the manufacturing facility of a Lynestrenol active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Lynestrenol APIs or Lynestrenol finished pharmaceutical products to another nation, regulatory agencies frequently require a Lynestrenol WC (written confirmation) as part of the regulatory process.
click here to find a list of Lynestrenol suppliers with Written Confirmation (WC) on PharmaCompass.
Lynestrenol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Lynestrenol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Lynestrenol GMP manufacturer or Lynestrenol GMP API supplier for your needs.
A Lynestrenol CoA (Certificate of Analysis) is a formal document that attests to Lynestrenol's compliance with Lynestrenol specifications and serves as a tool for batch-level quality control.
Lynestrenol CoA mostly includes findings from lab analyses of a specific batch. For each Lynestrenol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Lynestrenol may be tested according to a variety of international standards, such as European Pharmacopoeia (Lynestrenol EP), Lynestrenol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Lynestrenol USP).

