
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe
0
Canada
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
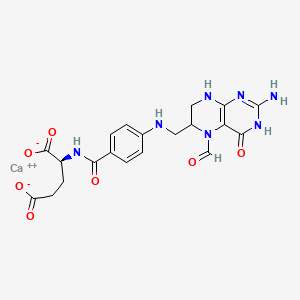

1. Citrovorum Factor
2. Calcium Folinate
3. Calcium Leucovorin
1. Calcium Leucovorin
2. Folidan
3. Rescuvolin
4. Calcii Folinas
5. Leucovorin Calcium Preservative Free
6. Lederfolat
7. Folinate Calcium
8. Anhydrous Calcium Folinate
9. Rpr1r4c0p4
10. Folaren
11. Nsc-3590
12. Leucovorin Calcium [usp]
13. Calcium N-(p-((((6rs)-2-amino-5-formyl-5,6,7,8-tetrahydro-4-hydroxy-6-pteridinyl)methyl)amino)benzoyl)-l-glutamate (1:1)
14. Lederfolin
15. Rescufolin
16. Folacal
17. Foliben
18. Leucosar
19. Tonofolin
20. Calc Folinate
21. Leucovorin Calcium (usp)
22. Citrovorum Factor
23. Nsc3590
24. Calcifolin
25. Adinepar
26. Calfolex
27. Calinat
28. Cehafolin
29. Citofolin
30. Dalisol
31. Disintox
32. Divical
33. Folaxin
34. Folidar
35. Folinac
36. Folinoral
37. Folinvit
38. Foliplus
39. Citrec
40. Ecofol
41. Emovis
42. Folix
43. Uzel
44. Flynoken A
45. Foli-cell
46. Factor, Citrovorum
47. Cromatonbic Folinico
48. Refchem:55463
49. 5-formyl Tetrahydrofolate
50. 216-082-8
51. 846-741-0
52. N-(4-(((2-amino-5-formyl-1,4,5,6,7,8-hexahydro-4-oxo-6-pteridinyl)methyl)amnio)benzoyl)-l-glutamic Acid, Calcium Salt (1:1)
53. N-(p-(((2-amino-5-formyl-5,6,7,8-tetrahydro-4-hydroxy-6-pteridinyl)methyl)amino)benzoyl)glutamic Acid
54. Folinic Acid Calcium Salt Pentahydrate
55. Folinic Acid Calcium Salt
56. Wellcovorin
57. Folinate-sf Calcium
58. Calcium Citrovorum Factor
59. Calcium Folinate [inn]
60. Folinic Acid-sf, Calcium Salt
61. Folinic Acid (calcium)
62. (+)-l-folinic Acid, Calcium Salt
63. Calciumfolinate
64. Leucovorin Calcium Salt
65. L-glutamic Acid, N-[4-[[(2-amino-5-formyl-1,4,5,6,7,8-hexahydro-4-oxo-6-pteridinyl)methyl]amino]benzoyl]-, Calcium Salt (1:1)
66. Lederfoline
67. 80433-71-2
68. Calcium (4-(((2-amino-5-formyl-4-oxo-1,4,5,6,7,8-hexahydropteridin-6-yl)methyl)amino)benzoyl)-l-glutamate
69. L-glutamic Acid, N-(4-(((2-amino-5-formyl-1,4,5,6,7,8-hexahydro-4-oxo-6-pteridinyl)methyl)amino)benzoyl)-, Calcium Salt (1:1)
70. Calcium L-folinate
71. Calcii Folinas [inn-latin]
72. Calcium;(2s)-2-[[4-[(2-amino-5-formyl-4-oxo-3,6,7,8-tetrahydropteridin-6-yl)methylamino]benzoyl]amino]pentanedioate
73. Folinic Acid Calcium Salt Hydrate
74. Calcium 5-formyltetrahydrofolate
75. Folinato Calcico [inn-spanish]
76. Folinate De Calcium [inn-french]
77. Nsc 3590
78. Einecs 216-082-8
79. Unii-rpr1r4c0p4
80. 5-formyl-5,6,7,8-tetrahydrofolic Acid Calcium Salt Hydrate
81. Prestwick_224
82. Calcium Folinate Rs
83. Calcium Folinate Crs
84. Schembl18573
85. Spectrum1500364
86. Calcium Folinate [jan]
87. Orb1310893
88. Calcium Folinate [vandf]
89. Chembl1201138
90. Schembl29360881
91. Chebi:31340
92. Hms500l04
93. Calcium Folinate [mart.]
94. Dtxsid20872448
95. Calcium Folinate [who-dd]
96. Folfox Component Leucovorin Calcium
97. Glxc-06576
98. Hms1570c18
99. Hms1920j05
100. Hms2091p21
101. Hms2097c18
102. Hms3714c18
103. Leucovorin Calcium [vandf]
104. Folinic Acid (calcium) (standard)
105. Leucovorin Calcium [usp-rs]
106. Ccg-38930
107. Ebc-15053
108. Hy-13664r
109. Akos015894859
110. Akos032949578
111. Calcium Folinate [ep Impurity]
112. Ac-7456
113. Ccg-269782
114. Cs-1363
115. Fc61510
116. Leucovorin Calcium [orange Book]
117. Calcium Folinate (leucovorin Calcium) Rs
118. As-13746
119. Folinate Calcium 100 Microg/ml In Water
120. Glutamic Acid, N-(p-(((2-amino-5-formyl-5,6,7,8-tetrahydro-3-hydroxy-6-pteridinyl)methyl)amino)benzoyl)-, Calcium Salt (1:1), L-
121. Hy-13664
122. Leucovorin Calcium [usp Monograph]
123. Folinic Acid (as Calcium Folinate)
124. C2235
125. Ns00127423
126. En300-6735051
127. 5-formyl-5,6,7,8-tetrahydrofolic Acid Calcium Salt
128. Q27077063
129. Z2683233771
130. Calcium (2s)-2-[(4-{[(2-amino-5-formyl-4-oxo-1,4,5,6,7,8-hexahydropteridin-6-yl)methyl]amino}phenyl)formamido]pentanedioate
| Molecular Weight | 511.5 g/mol |
|---|---|
| Molecular Formula | C20H21CaN7O7 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 7 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 221 |
| Heavy Atom Count | 35 |
| Formal Charge | 0 |
| Complexity | 900 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Antidotes
Agents counteracting or neutralizing the action of POISONS.
Vitamin B Complex
A group of water-soluble vitamins, some of which are COENZYMES.
A12AA20
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (2021) DOI:10.1021/acsenvironau.1c00008. List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
Hepatic and intestinal mucosal, the main metabolite being the active 5-methyltetrahydrofolate. Leucovorin is readily converted to another reduced folate, 5,10-methylenetetrahydrofolate, which acts to stabilize the binding of fluorodeoxyridylic acid to thymidylate synthase and thereby enhances the inhibition of this enzyme. Half Life: 6.2 hours
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
12
PharmaCompass offers a list of Leucovorin Calcium API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Leucovorin Calcium manufacturer or Leucovorin Calcium supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Leucovorin Calcium manufacturer or Leucovorin Calcium supplier.
A Leucovorin Calcium manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Leucovorin Calcium, including repackagers and relabelers. The FDA regulates Leucovorin Calcium manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Leucovorin Calcium API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Leucovorin Calcium manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Leucovorin Calcium supplier is an individual or a company that provides Leucovorin Calcium active pharmaceutical ingredient (API) or Leucovorin Calcium finished formulations upon request. The Leucovorin Calcium suppliers may include Leucovorin Calcium API manufacturers, exporters, distributors and traders.
click here to find a list of Leucovorin Calcium suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Leucovorin Calcium as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Leucovorin Calcium API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Leucovorin Calcium as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Leucovorin Calcium and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Leucovorin Calcium NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Leucovorin Calcium suppliers with NDC on PharmaCompass.
Leucovorin Calcium Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Leucovorin Calcium GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Leucovorin Calcium GMP manufacturer or Leucovorin Calcium GMP API supplier for your needs.
A Leucovorin Calcium CoA (Certificate of Analysis) is a formal document that attests to Leucovorin Calcium's compliance with Leucovorin Calcium specifications and serves as a tool for batch-level quality control.
Leucovorin Calcium CoA mostly includes findings from lab analyses of a specific batch. For each Leucovorin Calcium CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Leucovorin Calcium may be tested according to a variety of international standards, such as European Pharmacopoeia (Leucovorin Calcium EP), Leucovorin Calcium JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Leucovorin Calcium USP).

