
Synopsis
Synopsis
0
KDMF
0
VMF
0
Australia
0
South Africa
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
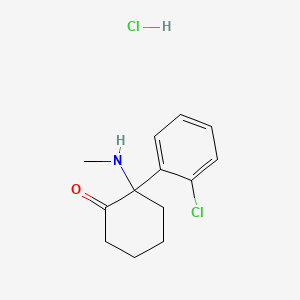

1. 2-(2-chlorophenyl)-2-(methylamino)cyclohexanone
2. Calipsol
3. Calypsol
4. Ci 581
5. Ci-581
6. Ci581
7. Kalipsol
8. Ketalar
9. Ketamine
10. Ketanest
11. Ketaset
1. 1867-66-9
2. Vetalar
3. Ketaset
4. Ketalar
5. Ketamine Hcl
6. Ketanest
7. Ketolar
8. 2-(2-chlorophenyl)-2-(methylamino)cyclohexanone Hydrochloride
9. Cl 369
10. Cn-52,372-2
11. (+-)-ketamine Hydrochloride
12. Ci-581
13. Ketamine Hydrochloride Ciii
14. Ketamine (as Hydrochloride)
15. O18yuo0i83
16. Calipsol
17. Kalipsol
18. Ketavet
19. (+-)-2-(o-chlorophenyl)-2-(methylamino)cyclohexanone Hydrochloride
20. Cl-369
21. Dea No. 7285
22. Cn-523722
23. 2-(2-chlorophenyl)-2-(methylamino)cyclohexan-1-one;hydrochloride
24. Cn-52372-2
25. Ketamine Chloride
26. Ketavet 100
27. Ketavet (veterinary)
28. Ci 581
29. Chebi:650657
30. Einecs 217-484-6
31. Unii-o18yuo0i83
32. Ketalar (tn)
33. Ketamini Hydrochloridum
34. Ketamine Hydrochloride [usan:usp:jan]
35. Cyclohexanone, 2-(2-chlorophenyl)-2-(methylamino)-, Hydrochloride
36. Cyclohexanone, 2-(2-chlorophenyl)-2-(methylamino)-, Hydrochloride, (+-)-
37. Ec 217-484-6
38. Chembl1714
39. Schembl26999
40. Dtxsid4040137
41. Ketamine Hydrochloride [mi]
42. 2-(2-chlorophenyl)-2-(methylamino)cyclohexan-1-one Hydrochloride
43. Bcp25943
44. Ketamine Hydrochloride (jp17/usp)
45. Ketamine Hydrochloride [jan]
46. (+/-)-2-(o-chlorophenyl)-2-(methylamino)cyclohexanone Hydrochloride
47. Ketamine Hydrochloride [usan]
48. Nsc116131
49. (+/-)-ketamine Hydrochloride, Solid
50. Ketamine Hydrochloride [vandf]
51. Akos005287313
52. Cyclohexanone, 2-(o-chlorophenyl)-2-(methylamino)-, Hydrochloride, (+-)-
53. Ketamine Hydrochloride [mart.]
54. Ab02117
55. Ketamine Hydrochloride [who-dd]
56. Ketamine Hydrochloride [who-ip]
57. Nsc-116131
58. Ketamine Hydrochloride [green Book]
59. Db-044614
60. Db-048467
61. Ketamine Hydrochloride [ep Impurity]
62. Ketamine Hydrochloride [orange Book]
63. Ketamine Hydrochloride [ep Monograph]
64. Ketamine Hydrochloride Ciii [usp-rs]
65. Ketamine Hydrochloride [usp Monograph]
66. Ketamini Hydrochloridum [who-ip Latin]
67. C07843
68. D00711
69. 867k669
70. A813079
71. Q-201266
72. Q27105184
73. Ketamine Hydrochloride 1.0 Mg/ml In Methanol (as Free Base)
74. 2-(2-chlorophenyl)-2-(methylamino)-1-cyclohexanone Hydrochloride
75. Ketamine Hydrochloride, European Pharmacopoeia (ep) Reference Standard
76. Cyclohexanone, 2-(2-chlorophenyl)-2-(methylamino)-, Hydrochloride (1:1)
77. Cyclohexanone, 2-(o-chlorophenyl)-2-(methylamino)-, Hydrochloride, (+/-)-
78. Ketamine (+-)-2-(o-chlorophenyl)-2-(methylamino)cyclohexanonehydrochloride
79. Ketamine Hydrochloride Solution, 1.0 Mg/ml In Methanol (as Free Base), Ampule Of 1 Ml, Certified Reference Material
| Molecular Weight | 274.18 g/mol |
|---|---|
| Molecular Formula | C13H17Cl2NO |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 2 |
| Exact Mass | 273.0687196 g/mol |
| Monoisotopic Mass | 273.0687196 g/mol |
| Topological Polar Surface Area | 29.1 Ų |
| Heavy Atom Count | 17 |
| Formal Charge | 0 |
| Complexity | 269 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 4 | |
|---|---|
| Drug Name | Ketalar |
| Drug Label | Ketalar is a nonbarbiturate anesthetic chemically designated dl 2-(0-chlorophenyl)-2-(methylamino) cyclohexanone hydrochloride. It is formulated as a slightly acid (pH 3.5-5.5) sterile solution for intravenous or intramuscular injection in concentrat... |
| Active Ingredient | Ketamine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 10mg base/ml; eq 50mg base/ml; eq 100mg base/ml |
| Market Status | Prescription |
| Company | Par Sterile Products |
| 2 of 4 | |
|---|---|
| Drug Name | Ketamine hydrochloride |
| PubMed Health | Ketamine (Injection) |
| Drug Label | Ketamine hydrochloride is a nonbarbiturate anesthetic chemically designated dl 2-(0-chlorophenyl)-2-(methylamino) cyclohexanone hydrochloride. It is formulated as a slightly acid (pH 3.5-5.5) sterile solution for intravenous or intramuscular injectio... |
| Active Ingredient | Ketamine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 10mg base/ml; eq 50mg base/ml; eq 100mg base/ml |
| Market Status | Prescription |
| Company | Hospira; Mylan Institutional; Eurohlth Intl |
| 3 of 4 | |
|---|---|
| Drug Name | Ketalar |
| Drug Label | Ketalar is a nonbarbiturate anesthetic chemically designated dl 2-(0-chlorophenyl)-2-(methylamino) cyclohexanone hydrochloride. It is formulated as a slightly acid (pH 3.5-5.5) sterile solution for intravenous or intramuscular injection in concentrat... |
| Active Ingredient | Ketamine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 10mg base/ml; eq 50mg base/ml; eq 100mg base/ml |
| Market Status | Prescription |
| Company | Par Sterile Products |
| 4 of 4 | |
|---|---|
| Drug Name | Ketamine hydrochloride |
| PubMed Health | Ketamine (Injection) |
| Drug Label | Ketamine hydrochloride is a nonbarbiturate anesthetic chemically designated dl 2-(0-chlorophenyl)-2-(methylamino) cyclohexanone hydrochloride. It is formulated as a slightly acid (pH 3.5-5.5) sterile solution for intravenous or intramuscular injectio... |
| Active Ingredient | Ketamine hydrochloride |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | eq 10mg base/ml; eq 50mg base/ml; eq 100mg base/ml |
| Market Status | Prescription |
| Company | Hospira; Mylan Institutional; Eurohlth Intl |
Anesthetics, Dissociative
Intravenous anesthetics that induce a state of sedation, immobility, amnesia, and marked analgesia. Subjects may experience a strong feeling of dissociation from the environment. The condition produced is similar to NEUROLEPTANALGESIA, but is brought about by the administration of a single drug. (From Gilman et al., Goodman and Gilman's The Pharmacological Basis of Therapeutics, 8th ed) (See all compounds classified as Anesthetics, Dissociative.)
Analgesics
Compounds capable of relieving pain without the loss of CONSCIOUSNESS. (See all compounds classified as Analgesics.)
Excitatory Amino Acid Antagonists
Drugs that bind to but do not activate excitatory amino acid receptors, thereby blocking the actions of agonists. (See all compounds classified as Excitatory Amino Acid Antagonists.)
 Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2019-11-05
Pay. Date : 2019-10-28
DMF Number : 7686
Submission : 1988-09-22
Status : Active
Type : II

Certificate Number : CEP 2005-281 - Rev 02
Issue Date : 2024-03-11
Type : Chemical
Substance Number : 1020
Status : Valid

Registration Number : 218MF10980
Registrant's Address : Raiffeisenstrasse 4, D-77933 Lahr Germany
Initial Date of Registration : 2006-12-01
Latest Date of Registration :

NDC Package Code : 54382-128
Start Marketing Date : 1989-08-02
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT FOR HUMAN PRESCRIPTION COMPOUNDING

| Available Reg Filing : ASMF, CA |

 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.

NDC Package Code : 49169-1041
Start Marketing Date : 2024-01-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 54382-114
Start Marketing Date : 1989-02-08
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37014
Submission : 2022-03-29
Status : Active
Type : II

Certificate Number : CEP 2022-157 - Rev 00
Issue Date : 2024-03-04
Type : Chemical
Substance Number : 1020
Status : Valid

Date of Issue : 2024-02-12
Valid Till : 2027-02-11
Written Confirmation Number : WC-0407
Address of the Firm :

NDC Package Code : 70600-035
Start Marketing Date : 2022-03-22
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : ASMF, BR |

 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.

GDUFA
DMF Review : Reviewed
Rev. Date : 2022-12-22
Pay. Date : 2022-12-20
DMF Number : 27921
Submission : 2014-01-15
Status : Active
Type : II

Certificate Number : CEP 2016-194 - Rev 01
Issue Date : 2024-05-07
Type : Chemical
Substance Number : 1020
Status : Valid

Date of Issue : 2025-09-02
Valid Till : 2028-07-02
Written Confirmation Number : WC-0218
Address of the Firm :

NDC Package Code : 61281-8000
Start Marketing Date : 2013-07-08
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : ASMF |

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22256
Submission : 2008-11-28
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
GDUFA
DMF Review : Complete
Rev. Date : 2019-11-05
Pay. Date : 2019-10-28
DMF Number : 7686
Submission : 1988-09-22
Status : Active
Type : II
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
GDUFA
DMF Review : Complete
Rev. Date : 2022-12-22
Pay. Date : 2022-12-20
DMF Number : 27921
Submission : 2014-01-15
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37014
Submission : 2022-03-29
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2016-04-06
Pay. Date : 2015-04-16
DMF Number : 29094
Submission : 2015-04-14
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 19082
Submission : 2005-12-16
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5462
Submission : 1984-07-17
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2021-06-09
Pay. Date : 2021-04-29
DMF Number : 35385
Submission : 2020-12-30
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22256
Submission : 2008-11-28
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
NDC Package Code : 54382-128
Start Marketing Date : 1989-08-02
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT FOR HUMAN P...
NDC Package Code : 49169-1041
Start Marketing Date : 2024-01-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 54382-114
Start Marketing Date : 1989-02-08
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
NDC Package Code : 61281-8000
Start Marketing Date : 2013-07-08
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 70600-035
Start Marketing Date : 2022-03-22
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
NDC Package Code : 10920-597
Start Marketing Date : 2013-12-09
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

NDC Package Code : 62991-1087
Start Marketing Date : 2009-06-11
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT

NDC Package Code : 38779-1754
Start Marketing Date : 2015-03-16
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT FOR HUMAN P...

NDC Package Code : 51927-0120
Start Marketing Date : 2021-06-07
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT FOR HUMAN P...

NDC Package Code : 82393-116
Start Marketing Date : 2021-02-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
Seqens is an integrated global leader in pharmaceutical solutions & specialty ingredients, & custom-made solutions to our customers.
About the Company : Founded in 2003, Seqens has evolved into a global leader in pharmaceutical solutions and specialty ingredients. The company supports customers in the development, scale-up, and man...
 LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
LGM Pharma accelerates & optimizes the new product pathway from early development through commercialization.
About the Company : LGM Pharma is a global leader in sourcing APIs, including hard-to-find drug substances, for pharmaceutical and biotech industries. LGM also operates as a full-service drug product ...
About the Company : Virupaksha Organics, founded in 2003, is a leading manufacturer of APIs and intermediates. Its FDA-audited, ISO-certified facilities in Kazipally and Pashamylaram produce high-qual...
 Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
Supriya Lifesciences is a cGMP-compliant API manufacturing organization with a leadership position across key & niche products.
About the Company : Supriya Lifescience Limited, established in 1987 and headquartered in Mumbai, is a globally recognized, technology-driven manufacturer of APIs, CDMO, and formulations. Its faciliti...
 Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
Tenatra connects Indian manufacturers with global buyers through active partners in Germany, Switzerland, Belgium, Spain & Turkey.
About the Company : Tenatra International was established as a proprietorship firm in 1999. It got off to a very good start, supporting clients in the United States, Mexico and Europe. As business opp...
About the Company : Maithri Drugs Pvt. Ltd. is a global supplier of Active Pharmaceutical Ingredients (APIs), serving pharmaceutical companies in 60+ countries. Its API portfolio spans antivirals, ant...
About the Company : Arevipharma GmbH is a modern manufacturer of active pharmaceutical ingredients and intermediates with more than 140 years of expertise. Our customers are generic and research-based...

About the Company : BIOTECHNICA DWC LLC has carved a niche for itself in providing value added compliance, regulatory qualification, project management and GDP guidance services to pharma companies al...

About the Company : Globe Quimica S.A. is a major Brazilian API producer, GMP certificated by ANVISA, manufactures more than 20 different API's such as Antiretrovirals, Anxiolytic, Antidepressant, Ant...

About the Company : SB Pharma GmbH, with its headquarters in Cologne, is a leading company that has set itself the goal of offering pharmaceuticals, medical products and medical devices as well as a b...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : ANESKETIN
Dosage Form : SOLUTION
Dosage Strength : 100MG/ML
Packaging :
Approval Date :
Application Number : 2534576
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETAMINE HYDROCHLORIDE INJECTION, USP
Dosage Form : SOLUTION
Dosage Strength : 10MG/ML
Packaging :
Approval Date :
Application Number : 2550431
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETAMINE HYDROCHLORIDE INJECTION, USP
Dosage Form : SOLUTION
Dosage Strength : 50MG/ML
Packaging :
Approval Date :
Application Number : 2550458
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETAMINE HYDROCHLORIDE INJECTION USP
Dosage Form : SOLUTION
Dosage Strength : 10MG/ML
Packaging : 20 ML
Approval Date :
Application Number : 2246795
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETAMINE HYDROCHLORIDE INJECTION USP
Dosage Form : SOLUTION
Dosage Strength : 50MG/ML
Packaging : 10ML
Approval Date :
Application Number : 2246796
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETALAR
Dosage Form : SOLUTION
Dosage Strength : 10MG/ML
Packaging :
Approval Date :
Application Number : 224391
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETALAR
Dosage Form : SOLUTION
Dosage Strength : 50MG/ML
Packaging :
Approval Date :
Application Number : 224405
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETAMINE INJECTION, BP
Dosage Form : SOLUTION
Dosage Strength : 50MG/ML
Packaging :
Approval Date :
Application Number : 2535645
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : NARKETAN
Dosage Form : SOLUTION
Dosage Strength : 100MG/ML
Packaging : 10ML/50ML
Approval Date :
Application Number : 2374994
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada
KETAMINE (KETAMINE HYDROCHLORIDE)
Brand Name : KETASET
Dosage Form : SOLUTION
Dosage Strength : 100MG/ML
Packaging : 10ML/50ML
Approval Date :
Application Number : 2173239
Regulatory Info : NARCOTICS (CDSA I)
Registration Country : Canada

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Dosage Form : Injectable / Parenteral
Grade : Parenteral, Oral, Topical
Dosage Form : Emulsion
Grade : Parenteral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Global Sales Information

Dosage Form : Ketamine 100Mg 2Ml 5 Units Paren...
Dosage Strength : 5 vials 2ml 50 mg/ml
Price Per Pack (Euro) : 24.58
Published in :
Country : Italy
RX/OTC/DISCN : Class H

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Reply
13 Apr 2024
Reply
04 Jan 2024
Reply
08 Dec 2023
Reply
13 Nov 2020
Reply
21 Aug 2019
Reply
18 Jul 2019
Reply
13 Jun 2019
Reply
13 Oct 2018
Reply
09 Oct 2018
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
94
PharmaCompass offers a list of Ketamine Hydrochloride API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ketamine Hydrochloride manufacturer or Ketamine Hydrochloride supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ketamine Hydrochloride manufacturer or Ketamine Hydrochloride supplier.
A Ketamine Hydrochloride manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ketamine Hydrochloride, including repackagers and relabelers. The FDA regulates Ketamine Hydrochloride manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ketamine Hydrochloride API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ketamine Hydrochloride manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Ketamine Hydrochloride supplier is an individual or a company that provides Ketamine Hydrochloride active pharmaceutical ingredient (API) or Ketamine Hydrochloride finished formulations upon request. The Ketamine Hydrochloride suppliers may include Ketamine Hydrochloride API manufacturers, exporters, distributors and traders.
click here to find a list of Ketamine Hydrochloride suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Ketamine Hydrochloride DMF (Drug Master File) is a document detailing the whole manufacturing process of Ketamine Hydrochloride active pharmaceutical ingredient (API) in detail. Different forms of Ketamine Hydrochloride DMFs exist exist since differing nations have different regulations, such as Ketamine Hydrochloride USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ketamine Hydrochloride DMF submitted to regulatory agencies in the US is known as a USDMF. Ketamine Hydrochloride USDMF includes data on Ketamine Hydrochloride's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ketamine Hydrochloride USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ketamine Hydrochloride suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Ketamine Hydrochloride Drug Master File in Japan (Ketamine Hydrochloride JDMF) empowers Ketamine Hydrochloride API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Ketamine Hydrochloride JDMF during the approval evaluation for pharmaceutical products. At the time of Ketamine Hydrochloride JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Ketamine Hydrochloride suppliers with JDMF on PharmaCompass.
A Ketamine Hydrochloride CEP of the European Pharmacopoeia monograph is often referred to as a Ketamine Hydrochloride Certificate of Suitability (COS). The purpose of a Ketamine Hydrochloride CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Ketamine Hydrochloride EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Ketamine Hydrochloride to their clients by showing that a Ketamine Hydrochloride CEP has been issued for it. The manufacturer submits a Ketamine Hydrochloride CEP (COS) as part of the market authorization procedure, and it takes on the role of a Ketamine Hydrochloride CEP holder for the record. Additionally, the data presented in the Ketamine Hydrochloride CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Ketamine Hydrochloride DMF.
A Ketamine Hydrochloride CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Ketamine Hydrochloride CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Ketamine Hydrochloride suppliers with CEP (COS) on PharmaCompass.
A Ketamine Hydrochloride written confirmation (Ketamine Hydrochloride WC) is an official document issued by a regulatory agency to a Ketamine Hydrochloride manufacturer, verifying that the manufacturing facility of a Ketamine Hydrochloride active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Ketamine Hydrochloride APIs or Ketamine Hydrochloride finished pharmaceutical products to another nation, regulatory agencies frequently require a Ketamine Hydrochloride WC (written confirmation) as part of the regulatory process.
click here to find a list of Ketamine Hydrochloride suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Ketamine Hydrochloride as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Ketamine Hydrochloride API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Ketamine Hydrochloride as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Ketamine Hydrochloride and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Ketamine Hydrochloride NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Ketamine Hydrochloride suppliers with NDC on PharmaCompass.
Ketamine Hydrochloride Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ketamine Hydrochloride GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Ketamine Hydrochloride GMP manufacturer or Ketamine Hydrochloride GMP API supplier for your needs.
A Ketamine Hydrochloride CoA (Certificate of Analysis) is a formal document that attests to Ketamine Hydrochloride's compliance with Ketamine Hydrochloride specifications and serves as a tool for batch-level quality control.
Ketamine Hydrochloride CoA mostly includes findings from lab analyses of a specific batch. For each Ketamine Hydrochloride CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ketamine Hydrochloride may be tested according to a variety of international standards, such as European Pharmacopoeia (Ketamine Hydrochloride EP), Ketamine Hydrochloride JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ketamine Hydrochloride USP).

