
Synopsis
Synopsis
0
CEP/COS
0
EU WC
0
NDC API
0
VMF
0
South Africa
DRUG PRODUCT COMPOSITIONS
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
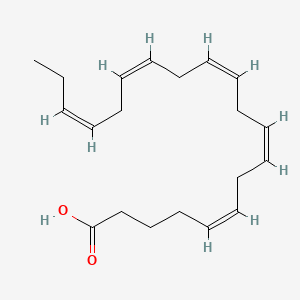

1. 5,8,11,14,17-eicosapentaenoic Acid
2. 5,8,11,14,17-icosapentaenoic Acid
3. Acid, Eicosapentanoic
4. Eicosapentanoic Acid
5. Icosapent
6. Omega 3 Eicosapentaenoic Acid
7. Omega-3-eicosapentaenoic Acid
8. Timnodonic Acid
1. Timnodonic Acid
2. Icosapent
3. 10417-94-4
4. Icosapentaenoic Acid
5. Epa
6. Cis-5,8,11,14,17-eicosapentaenoic Acid
7. (5z,8z,11z,14z,17z)-icosa-5,8,11,14,17-pentaenoic Acid
8. Icosapento
9. Icosapentum
10. 5,8,11,14,17-eicosapentaenoic Acid
11. Eicosapentaenoate
12. 5z,8z,11z,14z,17z-eicosapentaenoic Acid
13. (all-z)-5,8,11,14,17-eicosapentaenoic Acid
14. Icosapent [inn]
15. (5z,8z,11z,14z,17z)-5,8,11,14,17-eicosapentaenoic Acid
16. 5,8,11,14,17-icosapentaenoic Acid
17. Ropufa 70
18. Ccris 3279
19. Incromega E 7010sr
20. Aan7qov9ea
21. (5z,8z,11z,14z,17z)-icosapentaenoic Acid
22. (5z,8z,11z,14z,17z)-eicosapentaenoic Acid
23. Omega-3-carboxylic Acids
24. Epa 45g
25. Chembl460026
26. (5z,8z,11z,14z,17z)-eicosapentaenoate
27. All-cis-5,8,11,14,17-icosapentaenoic Acid
28. Chebi:28364
29. All-cis-5,8,11,14,17-eicosapentaenoic Acid
30. All-cis-icosa-5,8,11,14,17-pentaenoic Acid
31. Icosapent (inn)
32. Cis-delta(5,8,11,14,17)-eicosapentaenoic Acid
33. 5,8,11,14,17-eicosapentaenoic Acid, (all-z)-
34. All-cis-fatty Acid 20:5 Omega-3
35. Eicosapentaenoic Acid (c20:5 N3)
36. Epa;timnodonic Acid
37. Icosapentaenoate
38. (all-z)-delta5,8,11,14,17-eicosapentaenoic Acid
39. C20:5n-3,6,9,12,15
40. Cis-5,8,11,14,17-epa
41. Fa 20:5
42. Ncgc00161344-03
43. C20:5 (n-3)
44. Eicosapentaenoic Acid (20:5 N-3)
45. Eicosa-5z,8z,11z,14z,17z-pentaenoic Acid (20:5, N-3)
46. Miraxion
47. Eicosapentanoic Acid
48. Eye Q
49. Eye-q
50. 5,8,11,14,17-eicosapentaenoic Acid, (5z,8z,11z,14z,17z)-
51. Epa [drug]
52. Unii-aan7qov9ea
53. 1553-41-9
54. Icosapentum [inn-latin]
55. Icosapento [inn-spanish]
56. (5z,8z,11z,14z,17z)-icosapentaenoate
57. Timnodonate
58. 3gwx
59. All Cis-5,8,11,14,17-eicosapentaenoic Acid
60. Mfcd00065716
61. All-cis-icosapentaenoate
62. All-cis-icosapentaenoic Acid
63. Dsstox_cid_21023
64. Dsstox_rid_79612
65. Dsstox_gsid_41023
66. Schembl20469
67. Bspbio_001328
68. Bml3-b01
69. Gtpl3362
70. Dtxsid9041023
71. Eicosapentaenoic Acid [mi]
72. Hms1361c10
73. Hms1791c10
74. Hms1989c10
75. Hms3402c10
76. Hms3649d19
77. Hy-b0660
78. Zinc4474603
79. 5,8,11,14,17-icosapentaenoate
80. Eicosapentaenoic Acid [inci]
81. Tox21_111991
82. 5,8,11,14,17-eicosapentaenoate
83. Bdbm50242349
84. Eicosapentaenoic Acid [vandf]
85. Lmfa01030759
86. S6476
87. Eicosapentaenoic Acid [mart.]
88. Akos027470327
89. Eicosapentaenoic Acid [usp-rs]
90. Eicosapentaenoic Acid [who-dd]
91. Ccg-207957
92. Ccg-208136
93. Db00159
94. Cis-5,8,11,14,17-eicosapentaenoate
95. Idi1_033798
96. Ncgc00161344-01
97. Ncgc00161344-02
98. Ncgc00161344-04
99. Ncgc00161344-07
100. 5z,8z,11z,14z,17z-eicosapentaenoate
101. Ac-31072
102. As-53730
103. Cas-10417-94-4
104. E0441
105. 5,8,11,14,17-eicosapentaenoic Acid (6ci)
106. All Cis-5,8,11,14,17-icosapentaenoic Acid
107. C06428
108. D08061
109. P16966
110. (all-cis)-5,8,11,14,17-eicosapentaenoic Acid
111. Eicosapentaenoic Acid (epa) (c20:5 N3)
112. L001256
113. Q409990
114. Sr-01000946647
115. Fa(20:5(5z,8z,11z,14z,17z))
116. J-001125
117. Sr-01000946647-1
118. Z,z,z,z,z-eicosa-5,8,11,14,17-pentaenoic Acid
119. (z,z,z,z,z)-5,8,11,14,17-eicosapentaenoic Acid
120. Brd-k47192521-001-02-1
121. Cis-5,8,11,14,17-eicosapentaenoic Acid, >=99%
122. 7f8bf016-b146-4f72-a52e-b9298ba3a9ab
123. C20h30o2 (cis-5,8,11,14,17-eicosapentaenoic Acid)
124. Eicosapentaenoic Acid, 5,8,11,14,17-(z,z,z,z,z)-
125. 5,8,11,14,17-eicosapentaenoic Acid, (all-z)- (8ci)
126. Cis-5,8,11,14,17-eicosapentaenoic Acid, >=85%, Liquid
127. Cis-5,8,11,14,17-eicosapentaenoic Acid, Analytical Standard
128. (5z,8z,11 Z,14z,17z)-icosa-5,8,11,14,17-pentaenoic Acid
129. (5z,8z,11z,14z,17z)-eicosa-5,8,11,14,17-pentaenoic Acid
130. Cis, Cis, Cis, Cis, Cis-eicosa-5,8,11,14,17-pentaenoic Acid
131. 5,8,11,14,17-eicosapentaenoic Acid, (5z,8z,11z,14z,17z)- (9ci)
132. Eicosapentaenoic Acid (epa) (c20:5) (constituent Of Krill Oil) [dsc]
133. Cis-5,8,11,14,17-eicosapentaenoic Acid, 500 Mug/ml In Ethanol, Certified Reference Material
| Molecular Weight | 302.5 g/mol |
|---|---|
| Molecular Formula | C20H30O2 |
| XLogP3 | 5.6 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 13 |
| Exact Mass | 302.224580195 g/mol |
| Monoisotopic Mass | 302.224580195 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 398 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 5 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
EPA can be used for lowering elevated triglycerides in those who are hyperglyceridemic. In addition, EPA may play a therapeutic role in patients with cystic fibrosis by reducing disease severity and may play a similar role in type 2 diabetics in slowing the progression of diabetic nephropathy.
FDA Label
OM3-CA is indicated as an adjunct to diet to reduce triglycerides levels in adults patients with severe hypertriglyceridemia (>500 mg/dL). The patients involved in this treatment should be laced with an appropriate lipid-lowering diet. Hypertriglyceridemia is defined as an elevated plasma triglyceride concentration. It is usually correlated to other secondary conditions such as poor diet, alcohol use, obesity, metabolic syndrome and type 2 diabetes.
FDA Label
Treatment of Familial Adenomatous Polyposis
Treatment of dyslipidaemia
Eicosanoids are chemical messengers derived from 20-carbon polyunsaturated fatty acids that play critical roles in immune and inflammatory responses. Both 20-carbon omega-6 fatty acids (arachidonic acid) and 20-carbon omega-3 fatty acids (EPA) can be found in cell membranes. During an inflammatory response, arachidonic acid and EPA are metabolized by enzymes known as cyclooxygenases and lipoxygenases to form eicosanoids. Increasing omega-3 fatty acid intake increases the EPA content of cell membranes and decreases the arachidonic acid content, resulting in higher proportions of eicosanoids derived from EPA. Physiologic responses to arachidonic acid-derived eicosanoids differ from responses to EPA-derived eicosanoids. In general, eicosanoids derived from EPA are less potent inducers of inflammation, blood vessel constriction, and clotting than eicosanoids derived from arachidonic acid.
OM3-CA is very effective in reducing triglyceride levels. After 14 days of treatment, it is possible to observe even a 21% reduction. The reduction of the triglycerides could reach even to 25% in cases with the maximal used concentration of 4 g.
Absorption
When compared to omega-3 -acid ethyl esters, OM3-CA present a 4-fold higher bioavailability. OM3-CA is absorbed directly in the small intestine and the maximal plasma concentration is reached between 4.5-5 hours after initial administration. The absorbed dosage is transferred to the general circulation via the lymphatic system and distributed within tissues throughout the body. The absorption speed and extent is highly promoted by the bile. In preclinical studies performed in dogs, the Cmax, tmax and AUC were reported to be 15.1 mcg/ml, 24 hours and 1210.3 mcg.h/ml, respectively.
Route of Elimination
OM3-CA does not go under renal excretion. After the metabolism, all the dose is excreted as CO2 and water in the form of expelled air and the rest is excreted in feces.
Volume of Distribution
This pharmacokinetic property is not available.
Clearance
The registered clearance rate at steady-state is of 548 ml/h for eicosapentaenoic acid and 518 ml/h for docohexaenoic acid.
OM3-CA is metabolized in the liver following the normal fatty acid oxidation. Once absorbed, they are incorporated into triglycerides, cholesterol esters and phospholipids in tissues. The metabolism is marked by beta-oxidation followed by tricarboxylic acid cycle. It is reported that OM3-CA is an inhibitor of several enzymes such as CYP2C9, CYP2C19 and to a lesser extent to CYP1A2, CYP2E1, CYP3A4. It is thought that the metabolism of OM3-CA is mainly done by CYP3A and CYP4F3B.
Eicosapentaenoic acid has known human metabolites that include Juniperonic acid.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The half-life of OM3-CA depends on the type of component in which for eicosapentaenoic acid it is estimated to be of approximately 4.7-10.8 hours while for docosahexaenoic acid is reported to be of about 7 hours. The half-life of baseline-adjusted at steady-state is of 36 and 46 hours respectively for eicosapentaenoic acid and docosahexaenoic acid.
The anti-inflammatory, antithrombotic and immunomodulatory actions of EPA is probably due to its role in eicosanoid physiology and biochemistry. Most eicosanoids are produced by the metabolism of omega-3 fatty acids, specifically, arachidonic acid. These eicosanoids, leukotriene B4 (LTB4) and thromboxane A2 (TXA2) stimulate leukocyte chemotaxis, platelet aggregation and vasoconstriction. They are thrombogenic and artherogenic. On the other hand, EPA is metabolized to leukotriene B5 (LTB5) and thromboxane A3 (TXA3), which are eicosanoids that promote vasodilation, inhibit platelet aggregation and leukocyte chemotaxis and are anti-artherogenic and anti-thrombotic. The triglyceride-lowering effect of EPA results from inhibition of lipogenesis and stimulation of fatty acid oxidation. Fatty acid oxidation of EPA occurs mainly in the mitochondria. EPA is a substrate for Prostaglandin-endoperoxide synthase 1 and 2. It also appears to affect the function and bind to the Carbohydrate responsive element binding protein (ChREBP) and to a fatty acid receptor (G-coupled receptor) known as GP40.
The reduction of the synthesis of triglycerides in the liver may be caused because the main components of OM3-CA, eicosapentaenoic acid, and docosahexaenoic acid, are poor substrates for the enzymes responsible for the synthesis of triglycerides. These two major components inhibit the esterification of other fatty acids. OM3-CA is also thought to enhance the clearance of triglycerides from the circulating very low-density lipoprotein particles by different potential effects such as inhibition of acyl-CoA:1,2-diacylglycerol acyltransferase, increase in mitochondrial and peroxisomal beta-oxidation in the liver, decrease lipogenesis in the liver and increase lipoprotein lipase activity.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2023-06-07
Pay. Date : 2023-06-05
DMF Number : 27182
Submission : 2013-05-23
Status : Active
Type : II

 Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.

GDUFA
DMF Review : Reviewed
Rev. Date : 2013-04-01
Pay. Date : 2012-11-20
DMF Number : 26271
Submission : 2012-08-01
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27998
Submission : 2014-02-10
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2022-07-25
Pay. Date : 2022-06-10
DMF Number : 36822
Submission : 2022-06-07
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2020-11-02
Pay. Date : 2020-10-20
DMF Number : 23911
Submission : 2010-07-07
Status : Active
Type : II

Registration Number : 223MF10118
Registrant's Address : 4th and 5th Floors, 2 Stockport Exchange, Railway Road, Stockport, SK1 3GG, UK
Initial Date of Registration : 2011-07-25
Latest Date of Registration :



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15062
Submission : 2000-09-29
Status : Active
Type : II

Registration Number : 217MF11111
Registrant's Address : 1-25 Kanda Nishikicho, Chiyoda-ku, Tokyo
Initial Date of Registration : 2005-12-05
Latest Date of Registration :


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22474
Submission : 2009-01-23
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 34019
Submission : 2019-09-16
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 37763
Submission : 2022-12-11
Status : Active
Type : II



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 39040
Submission : 2023-10-25
Status : Active
Type : II


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : Complete
Rev. Date : 2023-06-07
Pay. Date : 2023-06-05
DMF Number : 27182
Submission : 2013-05-23
Status : Active
Type : II
 Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
GDUFA
DMF Review : Complete
Rev. Date : 2013-04-01
Pay. Date : 2012-11-20
DMF Number : 26271
Submission : 2012-08-01
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26946
Submission : 2013-03-04
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27998
Submission : 2014-02-10
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2020-11-02
Pay. Date : 2020-10-20
DMF Number : 23911
Submission : 2010-07-07
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25289
Submission : 2011-09-12
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22077
Submission : 2008-10-09
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-02-20
Pay. Date : 2014-04-09
DMF Number : 28140
Submission : 2014-03-31
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-08-21
Pay. Date : 2013-02-27
DMF Number : 26892
Submission : 2013-02-26
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 22474
Submission : 2009-01-23
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]AK&MN Biofarm Icosapentate (raw material) (for export)
Registration Number : 227MF10010
Registrant's Address : #81, Sanggae-Ro, Nam-Gu, Ulsan, Korea
Initial Date of Registration : 2015-01-05
Latest Date of Registration : 2015-01-05

Registration Number : 223MF10118
Registrant's Address : 4th and 5th Floors, 2 Stockport Exchange, Railway Road, Stockport, SK1 3GG, UK
Initial Date of Registration : 2011-07-25
Latest Date of Registration : 2025-09-11

Registration Number : 217MF10441
Registrant's Address : 363 Tokutomi, Akaiwa City, Okayama Prefecture
Initial Date of Registration : 2005-08-19
Latest Date of Registration : 2020-06-11

Registration Number : 227MF10158
Registrant's Address : 30-5 Dongsunonggongdanji-gil, Naju-si, Jeollanam-do 58275, Korea
Initial Date of Registration : 2015-06-18
Latest Date of Registration : 2016-07-15

Registration Number : 303MF10106
Registrant's Address : No. 37 Section 6, North Chengdu Avenue, Guanghan, Deyang City, Sichuan Province P. R....
Initial Date of Registration : 2021-07-09
Latest Date of Registration : 2023-08-09

Registration Number : 228MF10081
Registrant's Address : Am Kraftwerk 6,66450 Bexbach, Germany
Initial Date of Registration : 2016-04-06
Latest Date of Registration : 2016-04-06

Registration Number : 303MF10048
Registrant's Address : Am Kraftwerk 6,66450 Bexbach, Germany
Initial Date of Registration : 2021-03-17
Latest Date of Registration : 2021-03-17

Registration Number : 217MF11111
Registrant's Address : 1-25 Kanda Nishikicho, Chiyoda-ku, Tokyo
Initial Date of Registration : 2005-12-05
Latest Date of Registration : 2023-09-13

Registration Number : 305MF10044
Registrant's Address : No. 1, Lutai Road, High-tech District, Zibo, Shandong, P. R. China
Initial Date of Registration : 2023-04-05
Latest Date of Registration : 2023-04-05

Registration Number : 217MF10180
Registrant's Address : 1-23-3 Nishi-Shinjuku, Shinjuku-ku, Tokyo
Initial Date of Registration : 2005-06-06
Latest Date of Registration : 2007-03-05

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Registrant Name : Aekyung Biopharm Co., Ltd.
Registration Date : 2024-06-28
Registration Number : 20240628-211-J-1661
Manufacturer Name : Aekyung Biopharm Co., Ltd.
Manufacturer Address : 81 Sanggae-ro, Nam-gu, Ulsan (80, 80A)

Registrant Name : Chemport Co., Ltd.
Registration Date : 2022-08-22
Registration Number : 20220822-211-J-1348
Manufacturer Name : Chemport Co., Ltd.
Manufacturer Address : Laboratory: 30-51, Dongsunonggongdanji-gil, Naju-si, Jeollanam-do; 2nd Floor, Researc...

Registrant Name : Aging Life Science Co., Ltd.
Registration Date : 2024-02-23
Registration Number : 20240223-211-J-1613
Manufacturer Name : KD Pharma Bexbach GmbH @ KD ...
Manufacturer Address : Am Kraftwerk 6 - 8 66450 Bexbach Germany@Fabrikstraße 51 66424 Homburg Germany

Registrant Name : Poonglim Pharmaceutical Co., Ltd.
Registration Date : 2025-12-29
Registration Number : 20250415-211-J-1850(1)
Manufacturer Name : Shandong Xinhua Pharmaceutic...
Manufacturer Address : Hutian Chemical Industrial Zone, Zibo, Shandong, China

Registrant Name : MPK Korea Co., Ltd.
Registration Date : 2025-04-15
Registration Number : 20250415-211-J-1850
Manufacturer Name : Shandong Xinhua Pharmaceutic...
Manufacturer Address : Hutian Chemical Industrial Zone, Zibo, Shandong, China

Registrant Name : Daeshin Muyak Co., Ltd.
Registration Date : 2026-04-15
Registration Number : 20260415-211-J-2159
Manufacturer Name : Sichuan Gowell Pharmaceutica...
Manufacturer Address : New Economic Development Zone, Meishan City, Sichuan Province, China

Registrant Name : Ace Biopharm Co., Ltd.
Registration Date : 2025-04-21
Registration Number : 20250421-211-J-1869
Manufacturer Name : Zhejiang Medicine Co., Ltd. ...
Manufacturer Address : 98 East Xinchang Dadao Road, Xinchang, Zhejiang, PR China,312500

Registrant Name : V-On Bio Co., Ltd.
Registration Date : 2025-11-04
Registration Number : 20251104-211-J-2032
Manufacturer Name : Nissui Corporation, Kashima ...
Manufacturer Address : 18-2, HigashiFukashiba, Kamis, Ibaraki 314-0103, Japan

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Registrant Name : V-On Bio Co., Ltd.
Registration Date : 2025-11-04
Registration Number : 20251104-211-J-2033
Manufacturer Name : Nissui Corporation, Tsukuba ...
Manufacturer Address : 7, Okubo, Tsukuba-shi, Ibaraki 300-2611, Japan

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]About the Company : Axplora, created from the merger of Farmabios, Novasep & PharmaZell, is a leading API manufacturing partner to the world’s leading pharma & biotech companies, delivering top-qual...
 Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
About the Company : Chunghwa Chemical Synthesis & Biotech Co. Ltd. (CCSB) offers cost-effective APIs with speed, supported by a skilled team. As the first Southeast Asian company to receive FDA approv...
 Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
Chunghwa provides cost-effective APIs & advanced intermediates with complete DMF or COS, ensuring quality & reliable production.
About the Company : Chunghwa Chemical Synthesis & Biotech Co. Ltd. (CCSB) offers cost-effective APIs with speed, supported by a skilled team. As the first Southeast Asian company to receive FDA approv...
EICOSAPENTAENOIC ACID ETHYL ESTER (EPA-E)
About the Company : We create chemistry for a sustainable future. In line with our corporate purpose, around 112,000 employees contribute to the success of our customers in nearly all sectors and alm...

About the Company : Royal DSM, founded in 1902, is a global, purpose-led health, nutrition, and bioscience company that specializes in human and animal health and nutrition solutions. Its primary goal...

About the Company : Guowei Pharmaceutical was established in 2005 and is headquartered in Chengdu High-tech Zone. Its subsidiaries include Chengdu Guowei Biopharmaceutical Co., Ltd., Sichuan Guowei Ph...

About the Company : Zenfold Sustainable Technologies (ZST) is a specialty and fine chemicals company, established in 2021. Our advanced technological capabilities and state-of-the-art infrastructure a...

About the Company : Zhejiang Medicine Co., Ltd. is a large-scale joint-stock comprehensive pharmaceutical enterprise which was set up in May 1997. Being approved in August 1999 by the China Securities...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Reply
28 Mar 2025
Reply
15 Nov 2023
Reply
16 Sep 2023
Reply
29 Aug 2023
Reply
04 Jul 2023
Reply
22 Jun 2023
Reply
06 Mar 2023
Reply
23 Aug 2022
Reply
28 May 2022
Reply
11 Feb 2022
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
42
PharmaCompass offers a list of Icosapent Ethyl API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Icosapent Ethyl manufacturer or Icosapent Ethyl supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Icosapent Ethyl manufacturer or Icosapent Ethyl supplier.
A Icosapent manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Icosapent, including repackagers and relabelers. The FDA regulates Icosapent manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Icosapent API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Icosapent manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Icosapent supplier is an individual or a company that provides Icosapent active pharmaceutical ingredient (API) or Icosapent finished formulations upon request. The Icosapent suppliers may include Icosapent API manufacturers, exporters, distributors and traders.
click here to find a list of Icosapent suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Icosapent DMF (Drug Master File) is a document detailing the whole manufacturing process of Icosapent active pharmaceutical ingredient (API) in detail. Different forms of Icosapent DMFs exist exist since differing nations have different regulations, such as Icosapent USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Icosapent DMF submitted to regulatory agencies in the US is known as a USDMF. Icosapent USDMF includes data on Icosapent's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Icosapent USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Icosapent suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Icosapent Drug Master File in Japan (Icosapent JDMF) empowers Icosapent API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Icosapent JDMF during the approval evaluation for pharmaceutical products. At the time of Icosapent JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Icosapent suppliers with JDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Icosapent Drug Master File in Korea (Icosapent KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Icosapent. The MFDS reviews the Icosapent KDMF as part of the drug registration process and uses the information provided in the Icosapent KDMF to evaluate the safety and efficacy of the drug.
After submitting a Icosapent KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Icosapent API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Icosapent suppliers with KDMF on PharmaCompass.
Icosapent Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Icosapent GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Icosapent GMP manufacturer or Icosapent GMP API supplier for your needs.
A Icosapent CoA (Certificate of Analysis) is a formal document that attests to Icosapent's compliance with Icosapent specifications and serves as a tool for batch-level quality control.
Icosapent CoA mostly includes findings from lab analyses of a specific batch. For each Icosapent CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Icosapent may be tested according to a variety of international standards, such as European Pharmacopoeia (Icosapent EP), Icosapent JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Icosapent USP).

